Nuclear Chemistry Decay Particles Decay Reactions RealLife Examples









- Slides: 17

Nuclear Chemistry • Decay Particles • Decay Reactions • Real-Life Examples

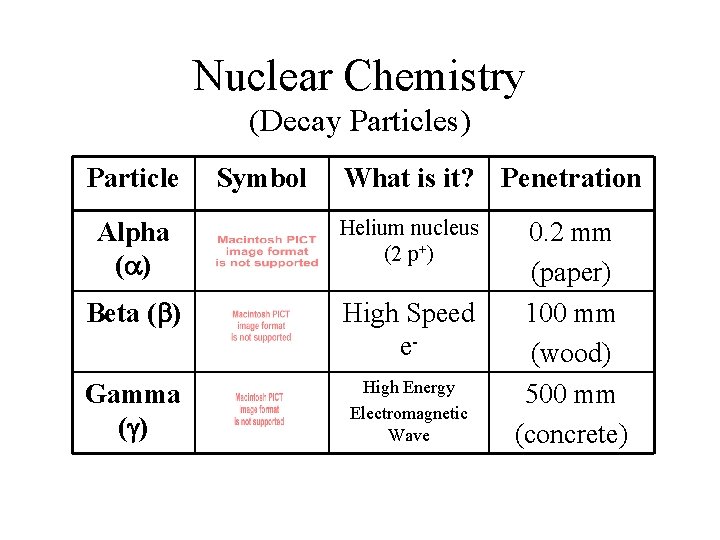
Nuclear Chemistry (Decay Particles) Particle Symbol What is it? Penetration Alpha (a) Helium nucleus (2 p+) Beta (b) High Speed e- Gamma (g) High Energy Electromagnetic Wave 0. 2 mm (paper) 100 mm (wood) 500 mm (concrete)

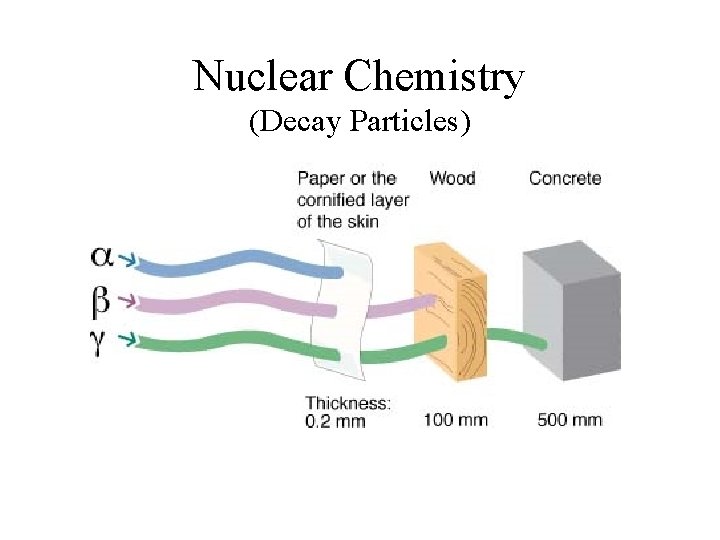
Nuclear Chemistry (Decay Particles)

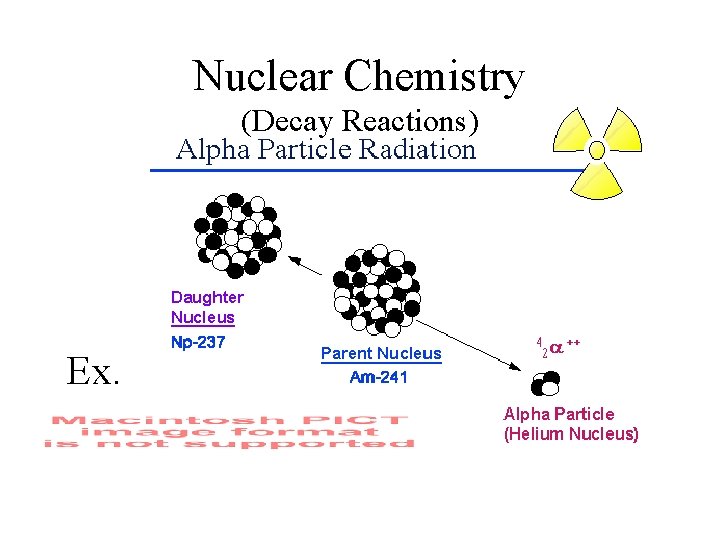
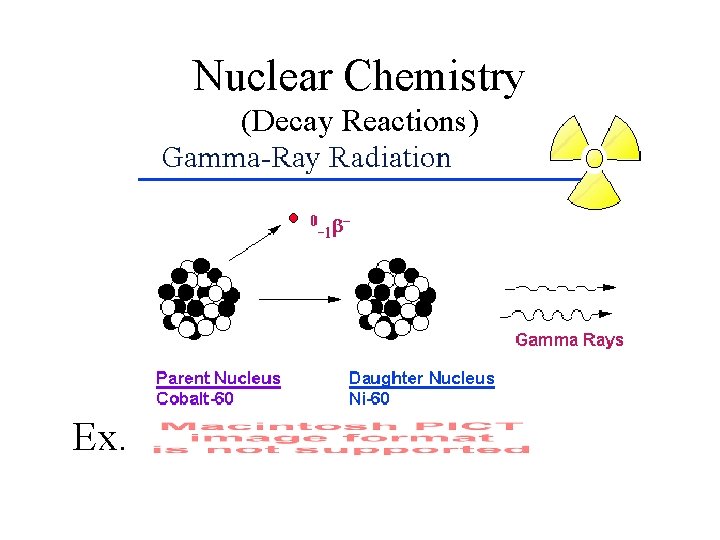
Nuclear Chemistry (Decay Reactions) Ex.

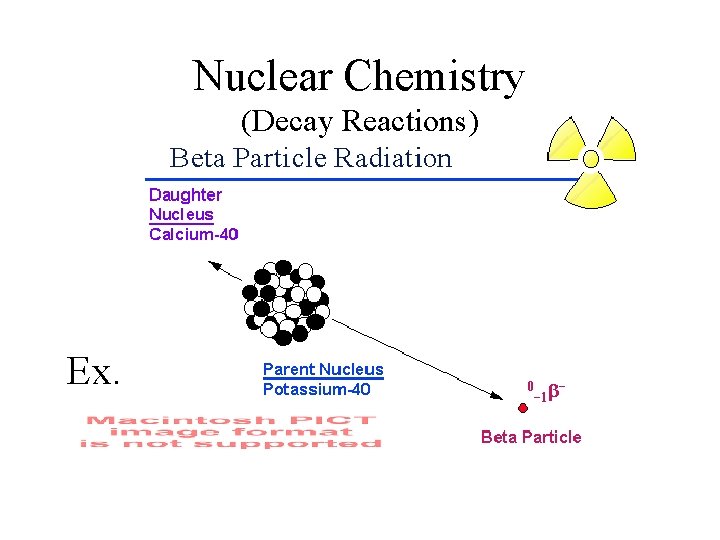
Nuclear Chemistry (Decay Reactions) Ex.

Nuclear Chemistry (Decay Reactions) Ex.

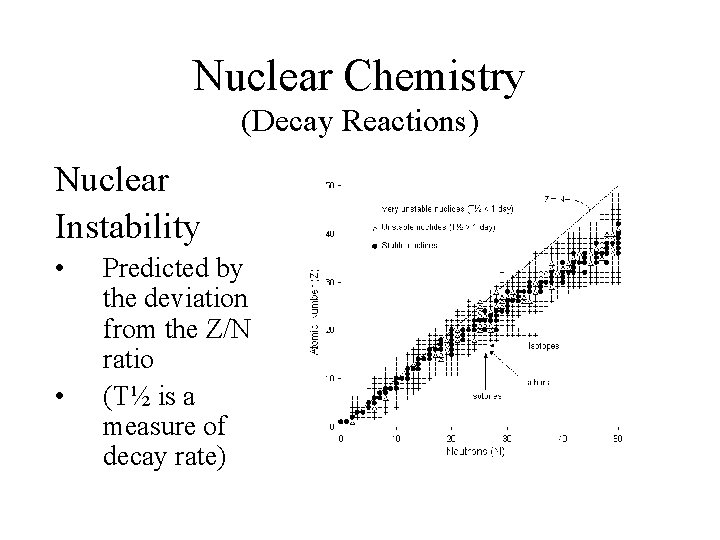
Nuclear Chemistry (Decay Reactions) Nuclear Instability • • Predicted by the deviation from the Z/N ratio (T½ is a measure of decay rate)

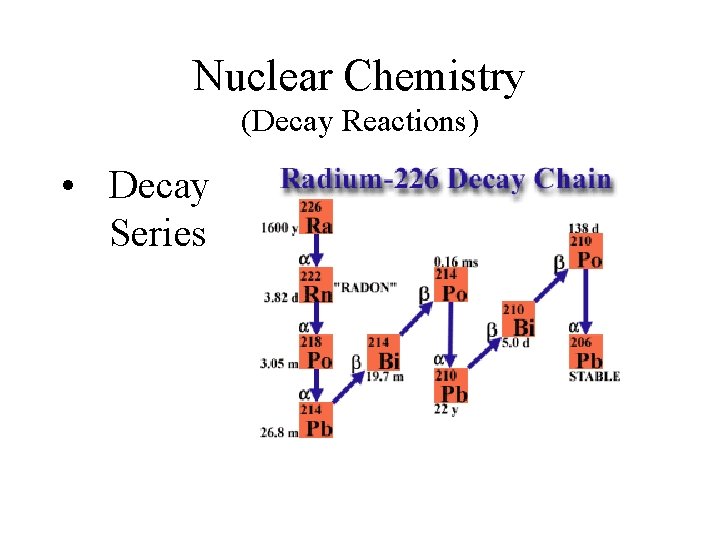
Nuclear Chemistry (Decay Reactions) • Decay Series

Nuclear Chemistry (Decay Reactions) Carbon-14 undergoes beta decay, what is the product? • We know it is Beta decay, so … • Balance the mass and atomic #s. • Answer:

Nuclear Chemistry (Real Life Examples) Nuclear Fission • Carbon-14 Dating • Atomic Fission (the bomb, nuclear power) • Radon • Chain Reactions

Nuclear Chemistry (Real Life Examples) Nuclear Fusion • The Sun! • Cold-Fusion

Nuclear Chemistry (Summary) • Decay Particles – Alpha, Beta, Gamma • Decay Reactions – Alpha decay, Beta decay, Gamma Decay, Decay Series • Real-Life Examples – Fission, Fusion

Nuclear Chemistry (Challenges/Misconceptions) • • Students have preconceived negative connotations of “nuclear” chemistry. Students have had little exposure to the facts (textbooks tend to put it at the end).

Nuclear Chemistry (Teaching Hints) • • Make use of the vast opinion articles and documentary videos on the subject. Students should get plenty of practice on balancing nuclear reactions (i. e. , carbon 14 undergoes beta decay, what is the product? ).

Average Atomic Mass To Determine Average Atomic Mass: • Step #1: Mass each isotope and multiply by # present • Step #2: Add these products • Step #3: Divide this sum by the total # present
Average Atomic Mass (Challenges/Misconceptions) • • Basically a math exercise, can hang up students. Students often forget what the total # present really is.

Average Atomic Mass (Teaching Hints) • • Introduce with a weighted average example from exam scores. Mix up with numbers and % of isotopes.