Chapter 21 Nuclear Chemistry Nuclear chemistry is the

















- Slides: 25

Chapter 21 Nuclear Chemistry

• Nuclear chemistry is the study of nuclear reactions, with an emphasis on their uses in chemistry and their effects on biological systems. 2

2. 1 Radioactivity The Nucleus • Review Chap 2. 3 for basic atomic structure. • Remember that the nucleus is comprised of two subatomic particles (nucleons) protons and neutrons. • The number of protons is the atomic number. • The number of protons and neutrons together is effectively the mass of the atom. 3

Isotopes • Not all atoms of the same element have the same mass due to different numbers of neutrons in those atoms. • There are three naturally occurring isotopes of uranium: ØUranium-234 ØUranium-235 ØUranium-238 4

• A nuclide is a nucleus with a specific number of protons and neutrons. • It is not uncommon for some nuclides of an element to be unstable, or radioactive. • We refer to these as radionuclides. • There are several ways radionuclides can decay into a different nuclide. 5

Nuclear Equations • Radionuclides are unstable and spontaneously emit particles and electromagnetic radiation. • Emission of radiation is a way for an unstable nucleus to be transformed into a more stable one with less energy. • When a nucleus spontaneously decomposes in this way it is said to have decayed. 6

Types of Radioactive Decay (see tables 21. 1 and 21. 2) 1. Alpha Decay: Loss of an -particle (a helium nucleus) 4 2 238 92 U He 234 90 Th + 4 2 He 7

2. Beta Decay: Loss of a -particle (a high energy electron) 0 − 1 131 53 I 0 or − 1 131 54 e Xe + 0 − 1 e 8

3. Gamma Emission: • Loss of a -ray -- high-energy photon that almost always accompanies the loss of a nuclear particle. 0 0 • Generally, not shown in a nuclear equation. 9

4. Positron Emission: • Loss of a positron -- a particle that has the same mass as but opposite charge than an electron. 0 1 11 6 C e 11 5 B + 0 1 e 10

5. Electron Capture • The addition of an electron to a proton in the nucleus. • As a result, a proton is transformed into a neutron. 1 1 p + 0 − 1 e 1 0 n 11

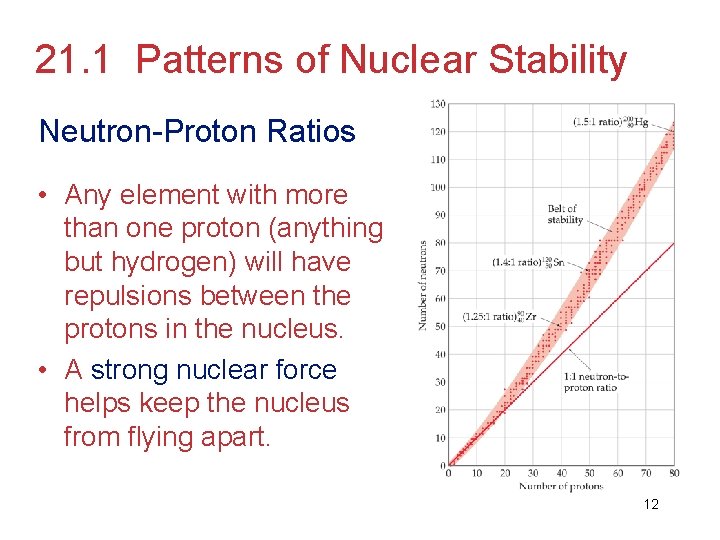
21. 1 Patterns of Nuclear Stability Neutron-Proton Ratios • Any element with more than one proton (anything but hydrogen) will have repulsions between the protons in the nucleus. • A strong nuclear force helps keep the nucleus from flying apart. 12

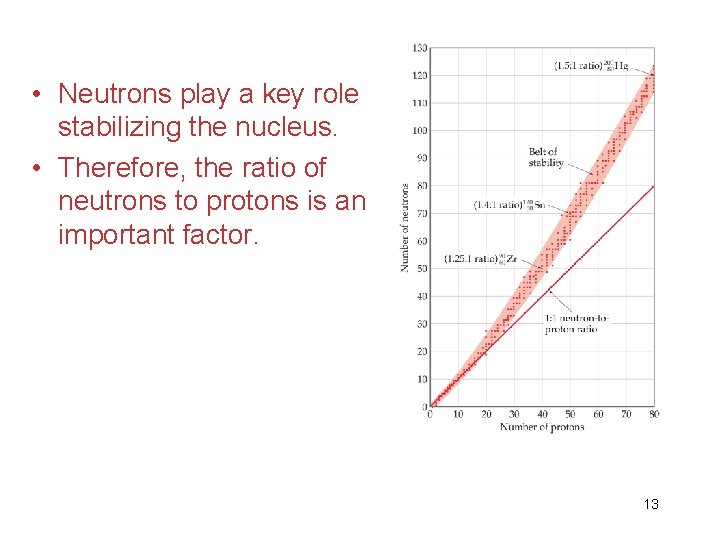
• Neutrons play a key role stabilizing the nucleus. • Therefore, the ratio of neutrons to protons is an important factor. 13

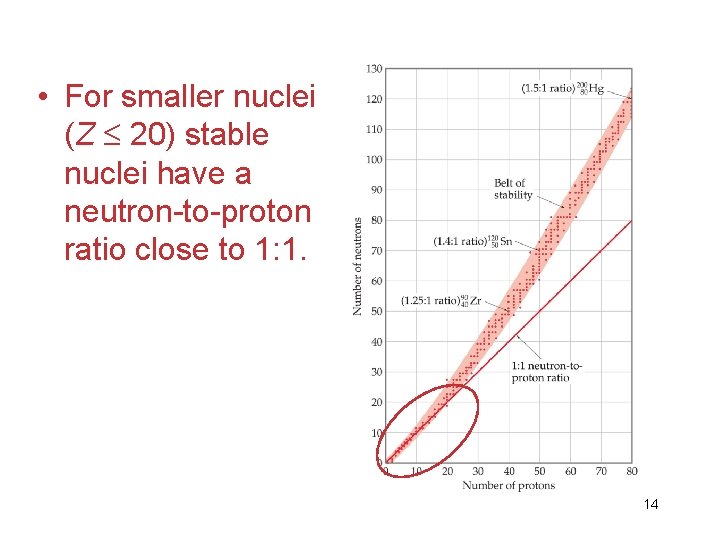
• For smaller nuclei (Z 20) stable nuclei have a neutron-to-proton ratio close to 1: 1. 14

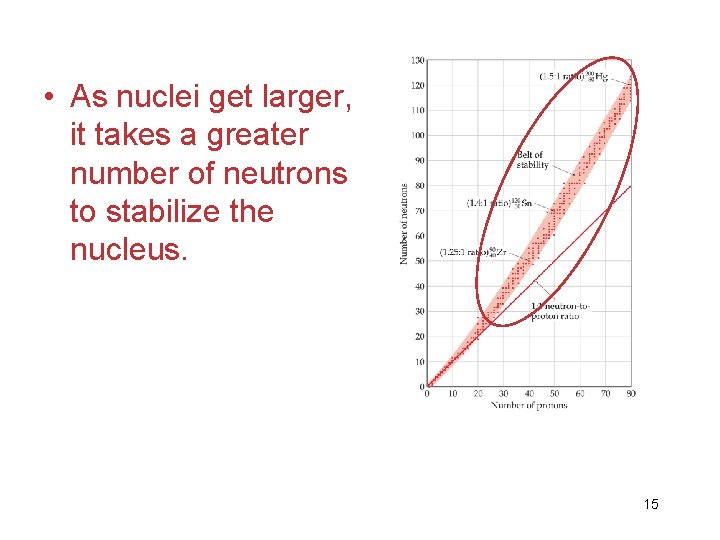
• As nuclei get larger, it takes a greater number of neutrons to stabilize the nucleus. 15

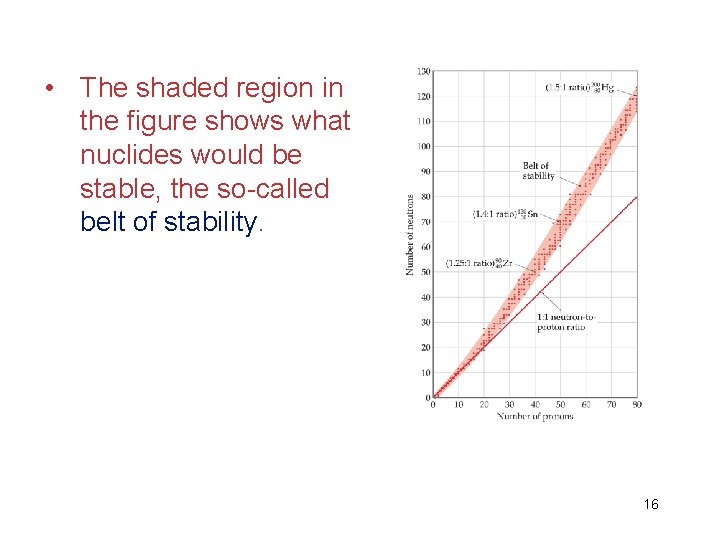
• The shaded region in the figure shows what nuclides would be stable, the so-called belt of stability. 16

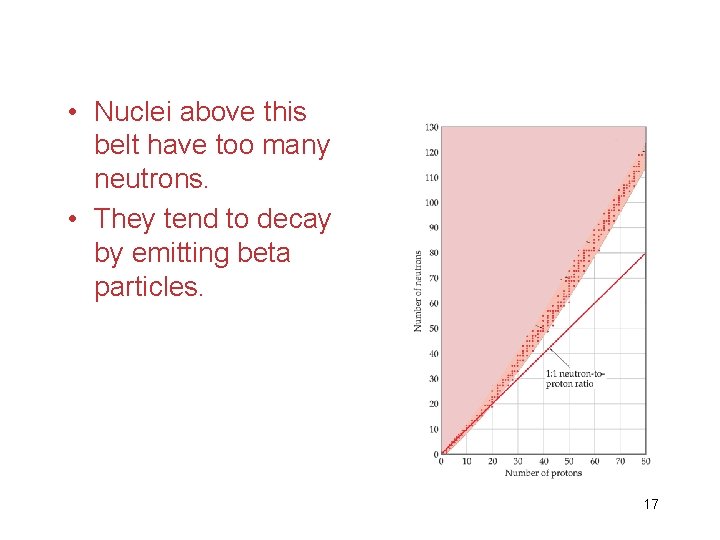
• Nuclei above this belt have too many neutrons. • They tend to decay by emitting beta particles. 17

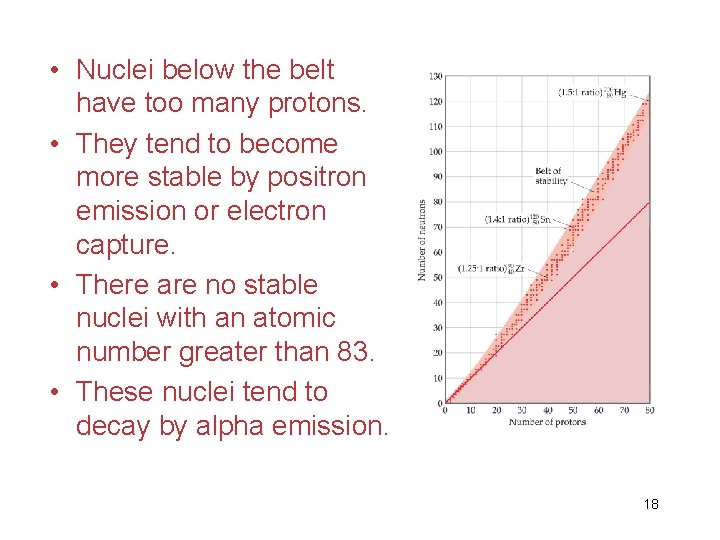
• Nuclei below the belt have too many protons. • They tend to become more stable by positron emission or electron capture. • There are no stable nuclei with an atomic number greater than 83. • These nuclei tend to decay by alpha emission. 18

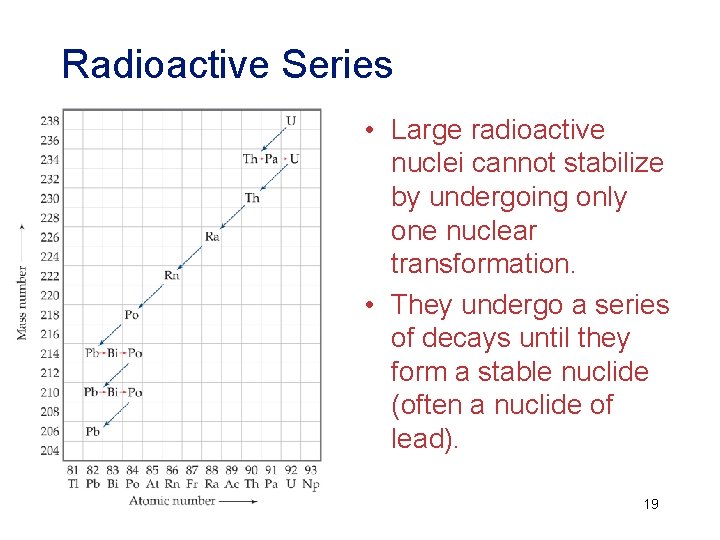
Radioactive Series • Large radioactive nuclei cannot stabilize by undergoing only one nuclear transformation. • They undergo a series of decays until they form a stable nuclide (often a nuclide of lead). 19

Some Trends. . . • Nuclei with 2, 8, 20, 28, 50, or 82 protons or 2, 8, 20, 28, 50, 82, or 126 neutrons tend to be more stable than nuclides with a different number of nucleons. • Nuclei with an even number of protons and neutrons tend to be more stable than nuclides that have odd numbers of these nucleons. 20

21. 3 Nuclear Transmutations • Nuclear transmutations can be induced by accelerating a particle (neutron or another nucleus) and colliding it with the nuclide. • Examples of particle accelerators: 21

• Synthetic isotopes used in medicine and research are made using neutrons as projectiles. • Example: cobalt-60 22

21. 4 Rates Radioactive Decay • Nuclear transmutation is a first-order process. • The kinetics of such a process, you will recall, obey this equation: Nt = kt ln N 0 23

• The half-life of such a process is: 0. 693 = t 1/2 k • Comparing the amount of a radioactive nuclide present at a given point in time with the amount normally present, one can find the age of an object. 24

Calculations Based on Half-life • Because the half-life of any particular nuclide is constant, the half-life serves as a nuclear clock to determine the ages of different objects. (See Table 21. 4) • The rate at which a sample decays is called its activity. • The SI unit for activity is the becquerel (Bq). • Another unit of activity is the curie (Ci), defined as 3. 7 x 1010 disintegrations per second. 25