Radioactivity Radioactivity Radioactivity The process by which an

Radioactivity

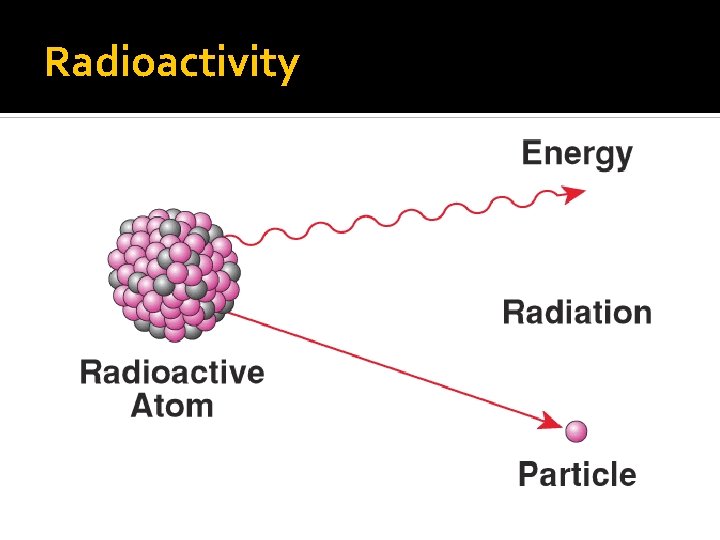
Radioactivity Radioactivity: The process by which an unstable nucleus emits one or more particles or energy in order to become stable

Nuclear Decay Some isotopes of common elements are unstable Too many or too few neutrons They decay into a stable atom The nucleus will release at least 1 form of nuclear radiation as it decays (gamma, alpha, or beta)
Radioactivity

Nuclear Radiation - 3 Types Alpha Particle - helium nucleus 2 protons 2 neutrons Beta Particle - electron Gamma Ray - high energy light wave no mass

Nuclear Power This is how Nuclear Energy works. These radioactive materials produce heat energy as they decay which is used to produce steam to turn a turbine.

Rates of Decay – HALF LIFE Half-Life: How long it takes for a radioactive element to decay into half of its original mass. The original element is made out of Parent Atoms. These atoms are unstable and radioactive. At each half-life, half of the existing parent atoms decay into the stable Daughter Atoms. These Parent atoms are not “lost”, rather they turn into something else – Daughter atoms.

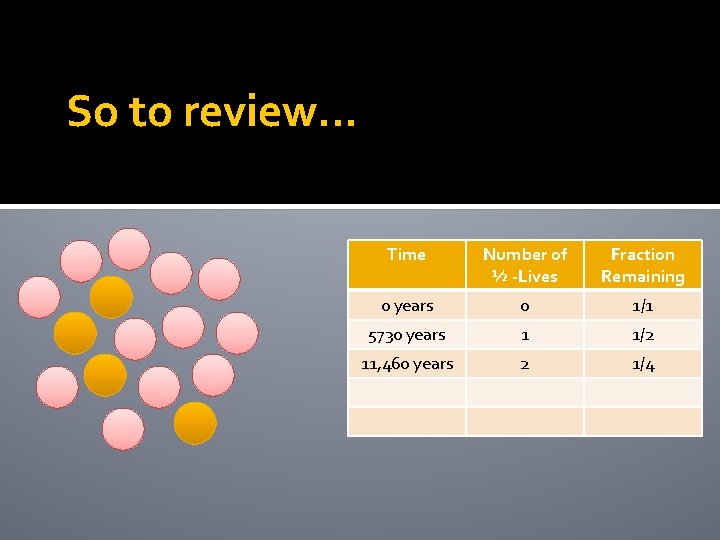
Example The half life of carbon-14 is 5730 years These circles represent 16 atoms of carbon-14. How many carbon -14 atoms will remain after 5730 years (one half life)?

Example The half life of carbon-14 is 5730 years 8 The 8 pink atoms are the new daughter atoms and are stable. How many of the carbon-14 atoms will decay after another 5730 years?

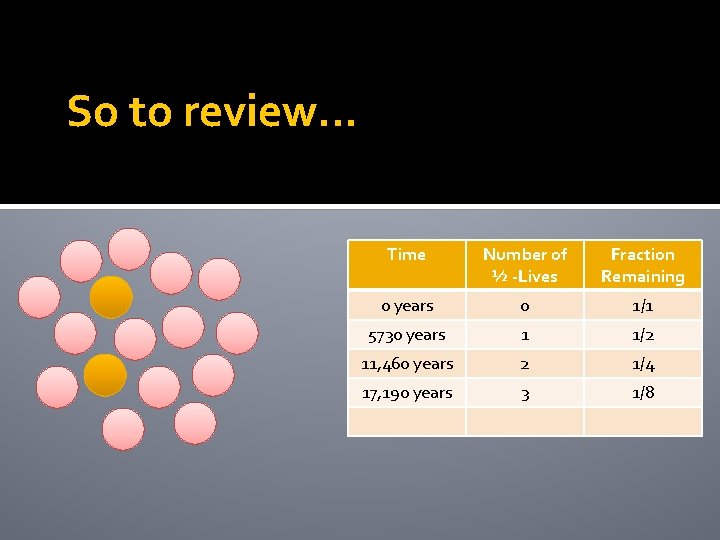
Example The half life of carbon-14 is 5730 years 4 The 12 pink atoms are the new daughter atoms and are stable. How many of the carbon-14 atoms will decay after another 5730 years?

Example The half life of carbon-14 is 5730 years 2 The 14 pink atoms are the new daughter atoms and are stable. How many of the carbon-14 atoms will decay after another 5730 years?

Example The half life of carbon-14 is 5730 years 1 The 15 pink atoms are the new daughter atoms and are stable. How many of the carbon-14 atoms will decay after another 5730 years?

Example The half life of carbon-14 is 5730 years 1 The 16 pink atoms are the new daughter atoms and are stable.

So to review… Time Number of ½ -LIves Fraction Remaining 0 years 0 1/1

So to review… Time Number of ½ -LIves Fraction Remaining 0 years 0 1/1 5730 years 1 1/2
So to review… Time Number of ½ -Lives Fraction Remaining 0 years 0 1/1 5730 years 1 1/2 11, 460 years 2 1/4
So to review… Time Number of ½ -Lives Fraction Remaining 0 years 0 1/1 5730 years 1 1/2 11, 460 years 2 1/4 17, 190 years 3 1/8
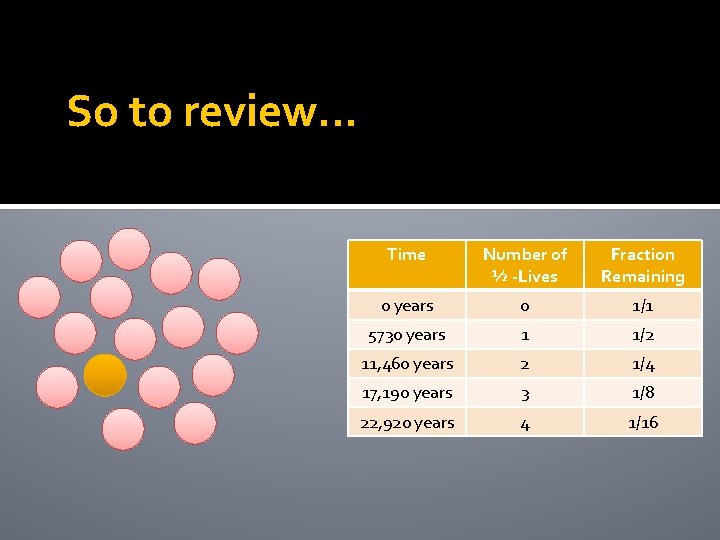
So to review… Time Number of ½ -Lives Fraction Remaining 0 years 0 1/1 5730 years 1 1/2 11, 460 years 2 1/4 17, 190 years 3 1/8 22, 920 years 4 1/16

Rate of Decay: HALF LIFE Carbon-14: (5730 year half-life) It will take 5730 years for 10 g of carbon-14 to decay down to 5 g of carbon-14 and 5 g of nitrogen-14. It will take 5730 years for 1000 atoms of carbon-14 to decay to 500 atoms of carbon-14 and 500 atoms of nitrogen-14.

We Use Half-Lives for Dating Things A sample of cloth has 2 ooo atoms of carbon-14 at formation. Today it has only 1 ooo atoms of carbon 14. How old is the cloth? 2 ooo atoms ÷ 2 = 1 ooo atoms (1 half life) 5730 years (1 half-life = 5730 years) How old is the cloth and how many half-lives have occurred if only 250 atoms of carbon-14 remain? ▪ 2 ooo atoms ÷ 2 = 1000 atoms ▪ 1 ooo atoms ÷ 2 = 500 atoms ▪ 500 atoms ÷ 2 = 250 atoms (1 half life) (2 half lives) (3 half-lives) 3 half lives = 5730 yrs x 3 = 17, 190 yrs
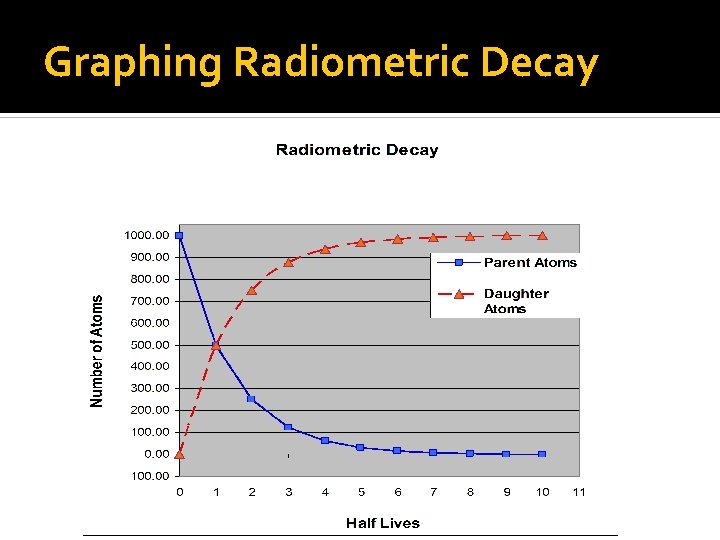
Graphing Radiometric Decay

Carbon Dating When something living organism dies, it stops taking in new carbon. The carbon-14 decays and is not replaced while the carbon-12 remains constant. So the ratio of carbon-12 to carbon-14 tells us how old the formerly living thing is. t = [ ln (Nf/No) / (-0. 693) ] x t 1/2

Radiometric Dating The half-life of carbon-14 is 5, 730 years, it is only reliable for dating objects that were living that are up to about 60, 000 years old. The principle of carbon-14 dating applies to other isotopes as well. The use of various radioisotopes allows the dating of biological and geological samples with a high degree of accuracy. Other Radioactive Elements include: Potassium-40 Uranium -235 Uranium -238 Thorium-232 Rubidium-87 (half-life of 1. 3 billion years) (half-life = 704 million years) (half-life = 4. 5 billion years) (half-life = 14 billion years) (half-life = 49 billion years)

Lab time!!!
- Slides: 24