In the time that it takes the music























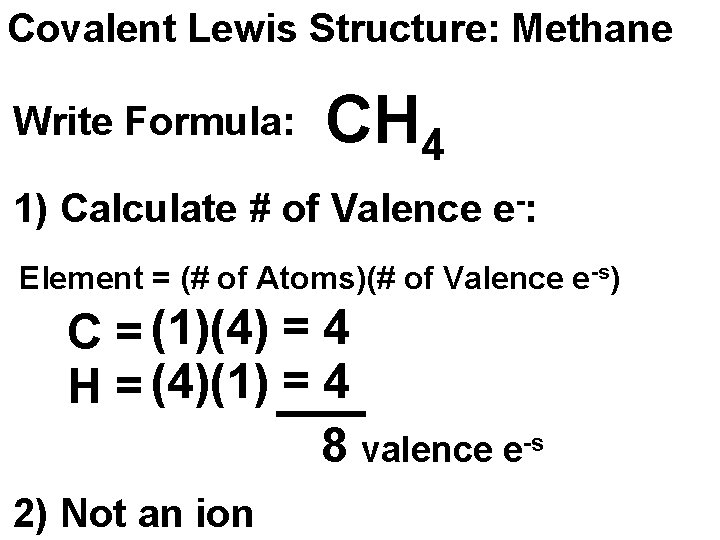







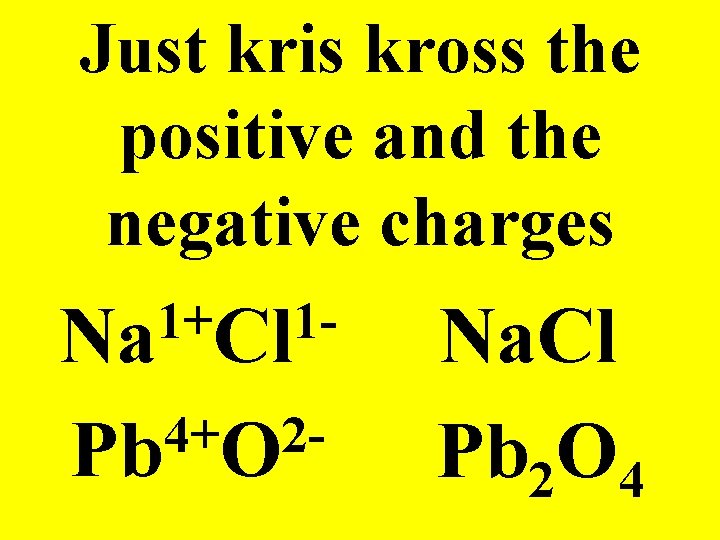



- Slides: 50

In the time that it takes the music to play…write as much as you know about bonds… Compound with the most correct wins!

What determines the type of bond that forms?

ELECTRONEGATIVITY The tendency of an atom to attract electrons to itself when it is bonded to another atom

BOND STRENGTH the energy needed to break the bonds between atoms in a compound

Bond Strength The greater the difference in electronegativities, the greater the bond strength

Misconception Alert! It does not require energy to make bonds. It requires energy to break bonds. “Breaking up is hard to do”

2 Main Bonding Options • By transferring electrons (producing ions) (big difference in EN) • By sharing electrons (small difference in EN)

IONIC BOND bond formed between two ions by the transfer of electrons (difference in EN > 1. 67)

IONIC COMPOUND substance formed when electrons are transferred between 2 or more substances (making ions)

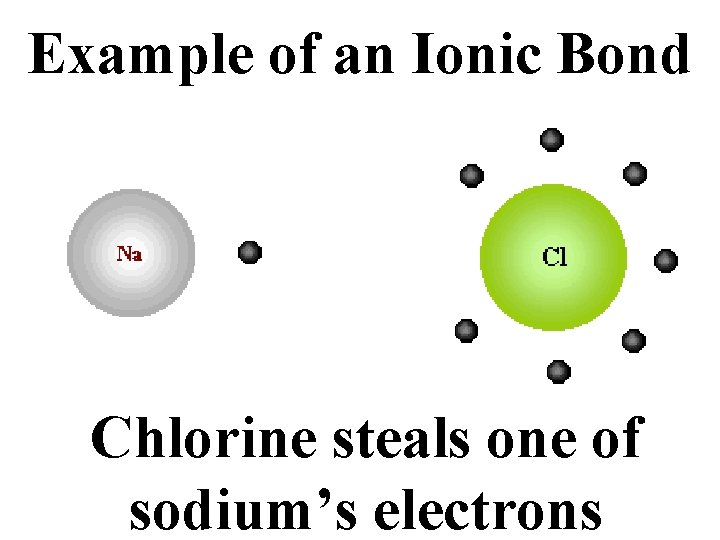
Example of an Ionic Bond Chlorine steals one of sodium’s electrons

Characteristics of Ionic Substances • Electrons are transferred • Bonds between metals and non-metals • Formulas are in simple ratios (Na. Cl)

More Characteristics of Ionic Substances • Crystalline structure at room temperature • High melting points • Good conductors of electricity when molten or dissolved

COVALENT BOND bond formed by the sharing of electrons (difference in EN < 1. 67)

MOLECULE a neutral group of atoms held together by covalent bonds (ex: H 2 O)

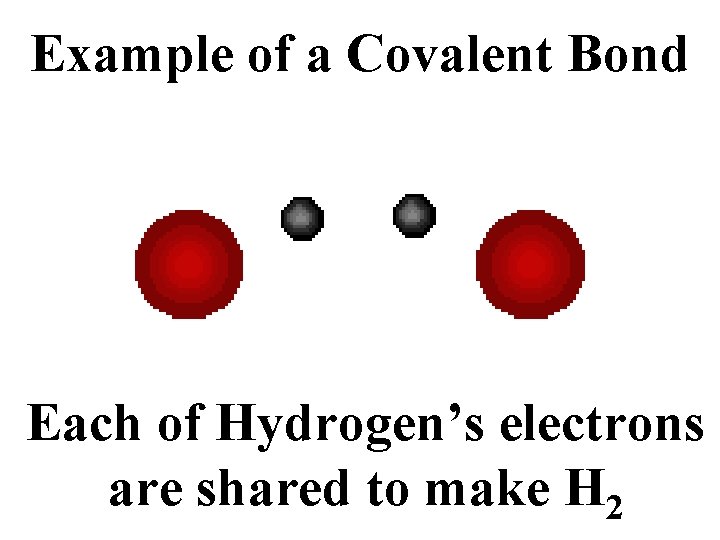
Example of a Covalent Bond Each of Hydrogen’s electrons are shared to make H 2

Characteristics of Covalent Substances • Electrons are shared • Bonds between non-metals and other non-metals • Formulas are in true ratio of atoms (C 6 H 12 O 6)

More Characteristics of Ionic Substances • Substances may exist in any state of matter at room temperature • Low melting points • Nonconductors of electricity

Bonds in all the polyatomic ions and diatomics are all covalent bonds

CONDUCTIVITY The ability to conduct an electrical current

Can we use conductivity to determine if a substance is ionic or covalent?

WHICH IS STRONGER? Ionic bonds are stronger than covalent

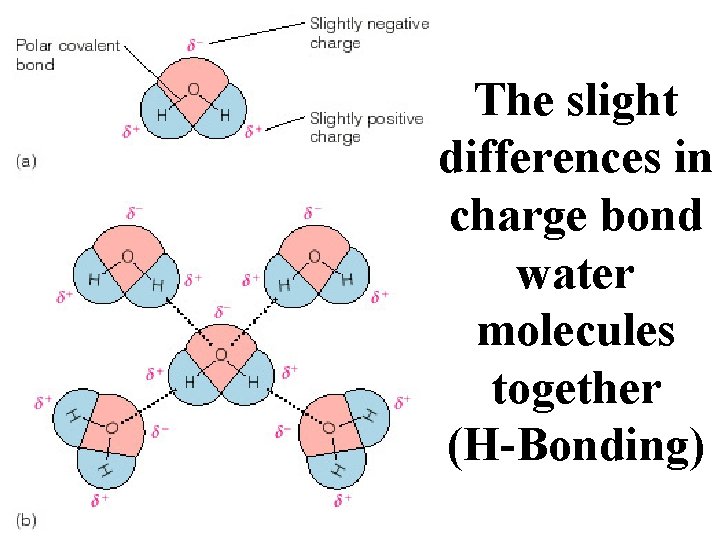
POLAR COVALENT BONDS when electrons are shared but shared unequally H 2 O

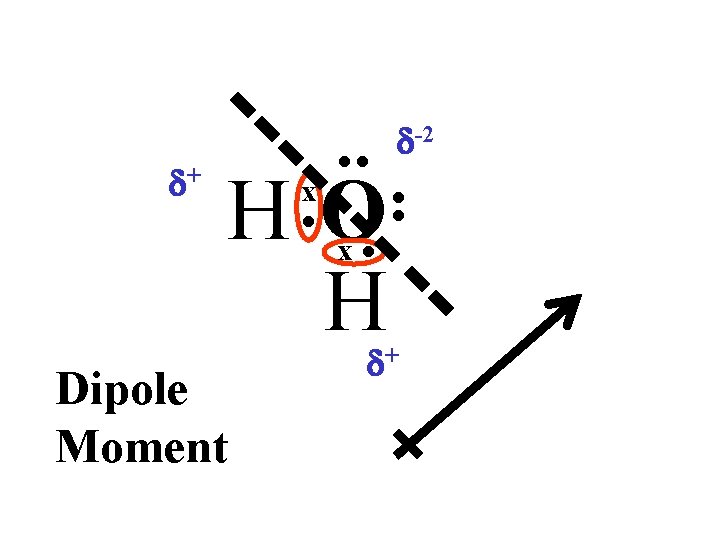
Example of a Polar Covalent Bond A dipole caused by the polar covalent bond of the water molecule. An unequal sharing of hydrogen’s electrons

Unequal sharing creates areas of positive and negative charge on a polar compound.

d+ • • H • O • H x x Dipole Moment d-2 • • d+

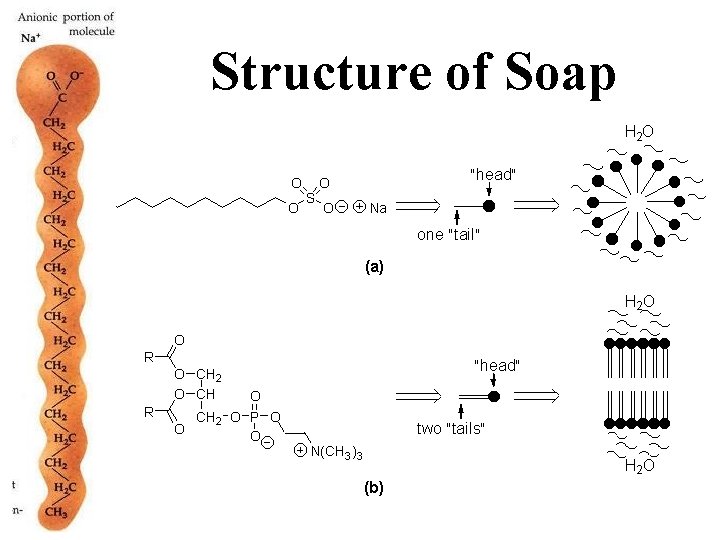
Structure of Soap

The slight differences in charge bond water molecules together (H-Bonding)

HOMEWORK Do problems 3 -4 on page 306 of the text. Pg. 304 #1, 2

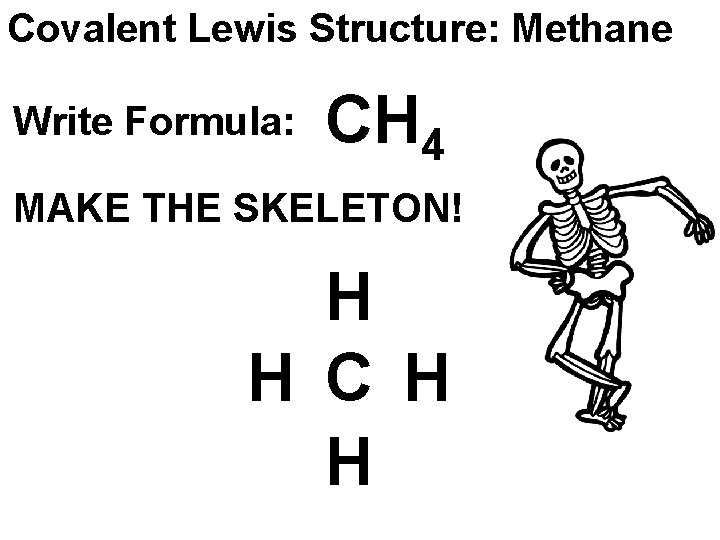
Covalent Lewis Structure: Methane Write Formula: CH 4 MAKE THE SKELETON! H H C H H
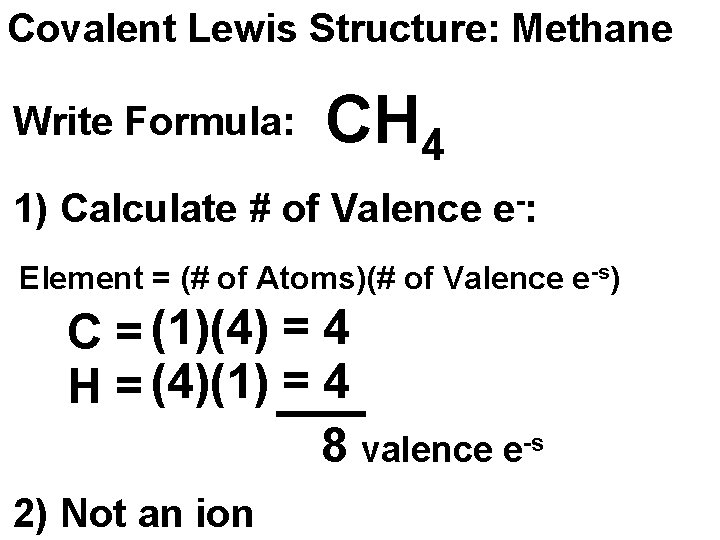
Covalent Lewis Structure: Methane Write Formula: CH 4 1) Calculate # of Valence e-: Element = (# of Atoms)(# of Valence e-s) C = (1)(4) = 4 H = (4)(1) = 4 8 valence e-s 2) Not an ion

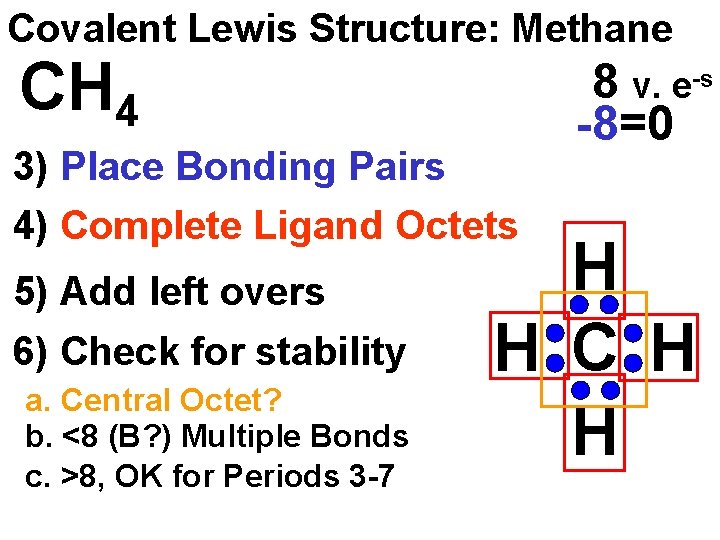
Covalent Lewis Structure: Methane CH 4 3) Place Bonding Pairs 4) Complete Ligand Octets 5) Add left overs 6) Check for stability a. Central Octet? b. <8 (B? ) Multiple Bonds c. >8, OK for Periods 3 -7 8 v. e-s -8=0 H H C H H

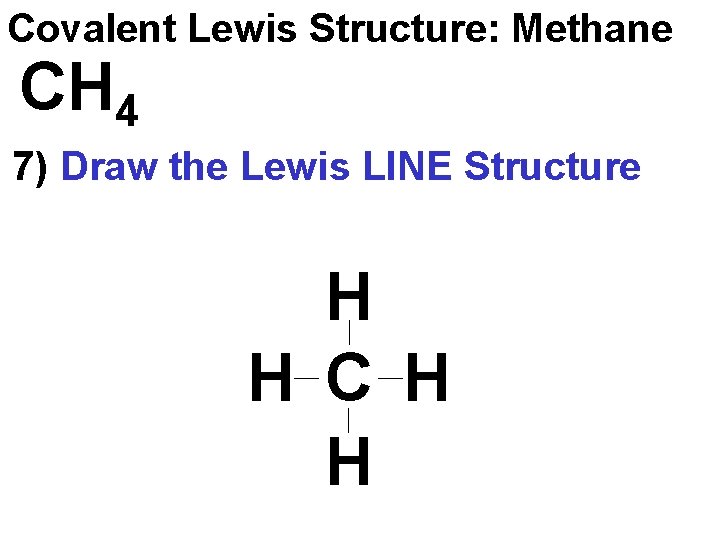
Covalent Lewis Structure: Methane CH 4 7) Draw the Lewis LINE Structure H H C H H

Covalent Lewis Structure: Boron Trifluoride Write Formula: BF 3 MAKE THE SKELETON! F F B F

Covalent Lewis Structure: Boron Trifluoride Write Formula: BF 3 1) Calculate # of Valence e-: Element = (# of Atoms)(# of Valence e-s) B = (1)(3) = 3 F = (3)(7) = 21 24 valence e-s 2) Not an ion

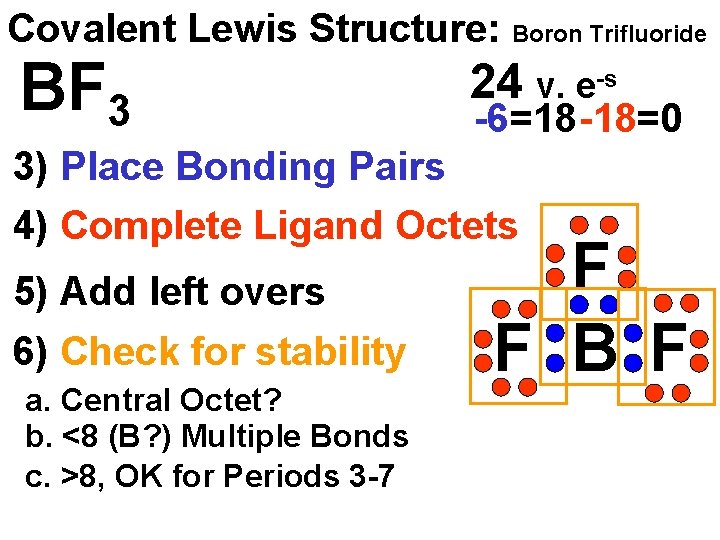
Covalent Lewis Structure: Boron Trifluoride BF 3 24 v. e-s -6=18 -18=0 3) Place Bonding Pairs 4) Complete Ligand Octets 5) Add left overs 6) Check for stability a. Central Octet? b. <8 (B? ) Multiple Bonds c. >8, OK for Periods 3 -7 F F B F

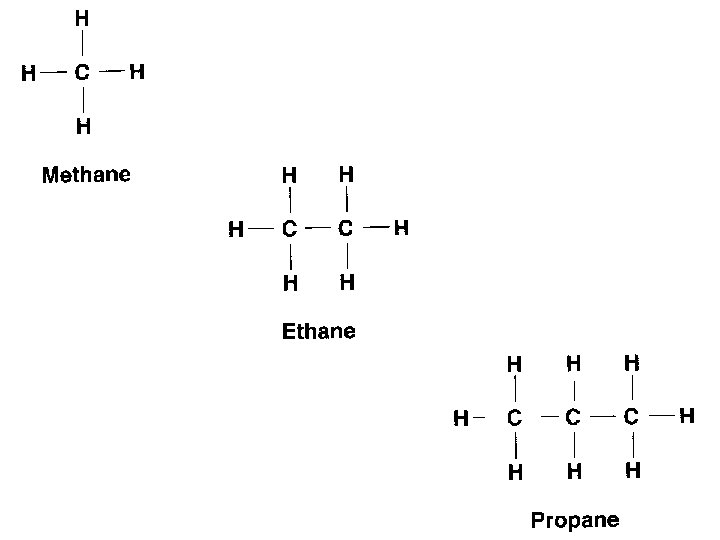
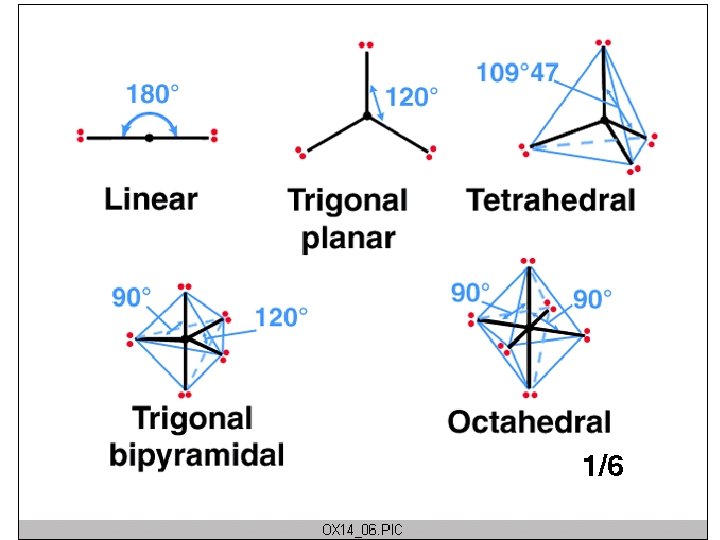
CARBON BONDS Carbon makes LOTS of bonds…but never more than 4


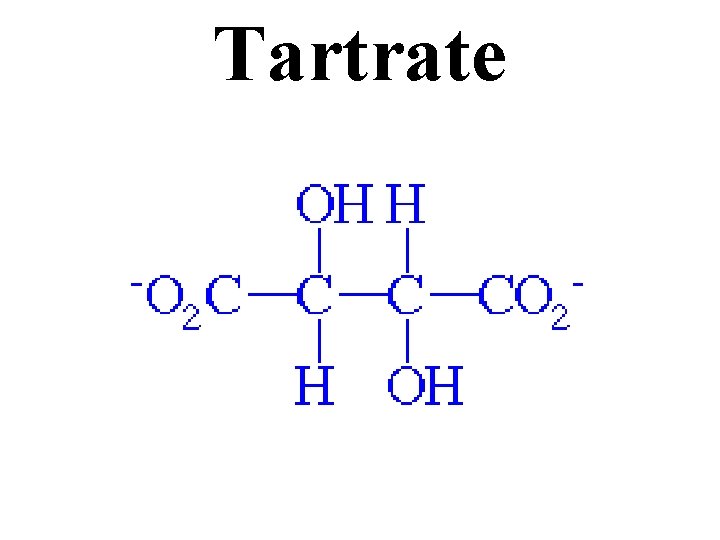
Tartrate


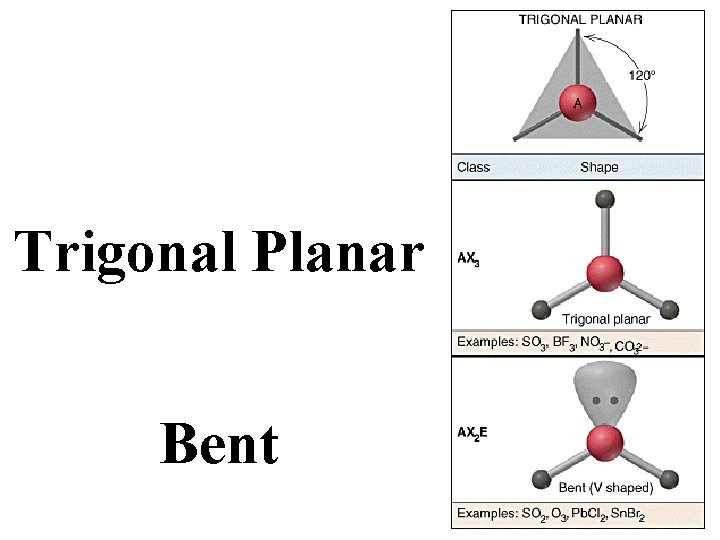
Trigonal Planar Bent

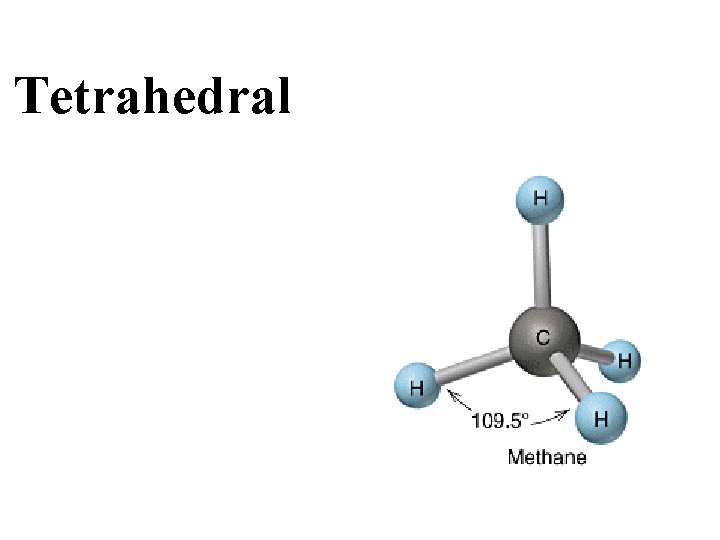
Tetrahedral

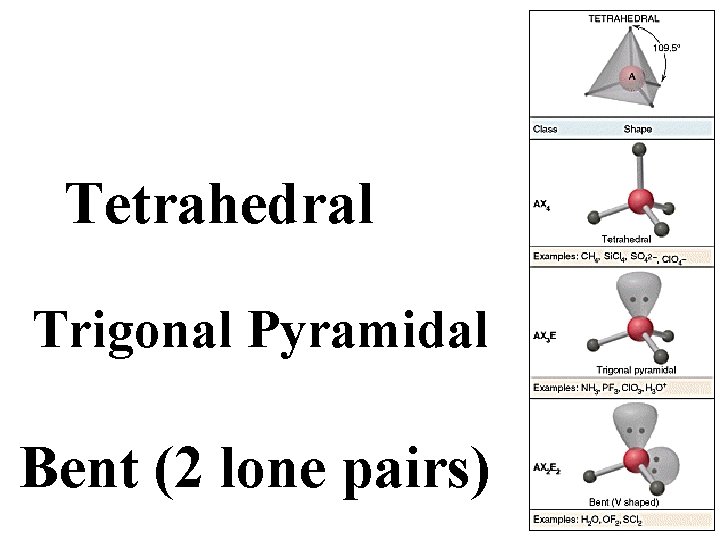
Tetrahedral Trigonal Pyramidal Bent (2 lone pairs)

IONIC COMPOUND substance formed when electrons are transferred between 2 or more substances (making ions)

How do you name ionic compounds?

How do you write ionic compounds?

Kris Kross Method! 2+ 3 Ca As Ca 3 As 2
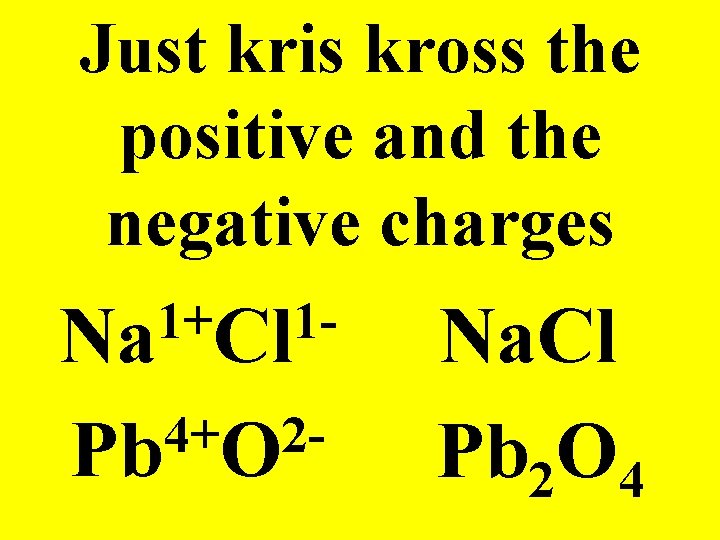
Just kris kross the positive and the negative charges 1+ 1 Na Cl 4+ 2 Pb O Na. Cl Pb 2 O 4

Which charge ALWAYS comes first? POSITIVE!!!

Are you sure?

For Polyatomics it gets a bit more complex 4+ 3 Pb SO 4 1+ 3 NH 4 PO 4 Pb 3(SO 4)4 (NH 4)3 PO 4