General Chemistry Assistant professor Mervat Mohamed Hosny 6
















































































- Slides: 83

General Chemistry Assistant professor • Mervat Mohamed Hosny •

6 -Quantum mechanical atom (Schroedinger)

1 -Democritus: He theorized that all matter is composed of • small indivisible particles called atoms •

2 -Dalton’s atomic theory: *each element is composed of minute • indivisible particles called atoms • *all atoms of a given element are chemically • identical to each other , atoms of one element are different from the atoms of all other element

3 - during ordinary chemical reactions atoms of one element cannot be changed into atoms of different element. 4 - atoms are not created or destroyed • 5 - compound is formed when atoms of more • than one element combine

3 -J. J. Thomson-CRT *he discovered the electron • * in thomson‘s model , electrons are • embedded in a positive sphere of matter •

4 -Rutherford gold foil experiment: *he established that the positive charged • alpha particles emitted by certain radioactive elements (helium). * he used these alpha particles to establish the • nuclear nature of atoms. *in these experiments , he directed a stream of • positive charged helium ions (alpha particles)at a very thin sheet of gold foil

conclusion *most of the mass and all of the positive • charge of the atom are contained in a small space called the nucleus • *most of the volume of the atom is empty • space occupied by tiny negatively charged electrons

*negative charged electrons outside nucleus =positive charge inside nucleus *the atom is electrically neutral • *protons: +vely charged subatomic particles • found in nucleus *neutrons : neutral (uncharged)subatomic • particles found in nucleui *electrons very small – vely charged • subatomic particles

5 - The Bohr model: *electrons in an atom exist in specific regions • at various distances from the nucleus. *The electrons are rotating in orbits around • the nucleus like planets rotating around the sun.

*he describe hydrogen atom as a single • electron rotating in an orbit about a relatively heavy nucleus. • *he applied the concept of energy quanta, • proposed by the German physicist Planck

Planck stated that : *energy is never emitted in a • continuous stream but only in small discrete packets called quanta •

Bohr theorized that : *There are several possible orbits for • electrons at different distances from the nucleus *but electron had to be in one specific orbit or • another.

*It could not exist between orbits *when a hydrogen atom adsorbed one or • more quanta of energy , its electron jumped to another orbit a greater distance from the nucleus. *when the electron fell back to lower orbits , it • emitted quanta of energy as light , giving rise to the spectrum of hydrogen.

*each orbit is at a different energy level *an electron in the orbit closest to the nucleus • • is in the 1 st energy level , at greater distances it • • may be in the second , 3 rd or fourth energy • level

6 -Quantum mechanical atom (Schroedinger) *They found that Bohr’s assumptions have to • be modified *Difficulty arise in applying theory to • atoms containing many electrons *Bohr’s concept was replaced by quantum • mechanics theory

One of the chief difference between the 2 theories is that: In the quantum mechanics theory electrons • are not considered to be revolving around the • nucleus in orbits but to occupy orbitals cloud • like regions surrounding the nucleus and • corresponding to energy levels •


Erwin Schrodinger introduced his famous wave equation Quantum mechanics or wave mechanics He describe an electron as simultaneously • having properties of: 1 -a wave (like light) • 2 -and a particle (have mass) •

The solution of the Schrodinger equation is complex but as aconclusion : There is four quantum numbers which • define the location and properties of • electrons in atoms: n, l, m, s •

n is the principle quantum no indicate the energy levels of the electron relative to their distance from the nucleus n=1, 2, 3, ……… • But always 1 -7 • • ﺑﻌﺪ ﺍﻻﻟﻜﺘﺮﻭﻥ ﻋﻦ ﺍﻟﻨﻮﺍﺓ

L=2 nd quantum no explain the shape of orbital Electron exist in orbitals having specific shapes • S P d f •

3 -m magnetic quantum no Orientation in space • *electron orbitals have specific orientation in • Space • *This quantum number accounts for the • number of s, p, d, f orbitals that can be present in the principal energy level

4 -Spine quantum no (s) *an electron spins about its own axis in either • a clockwise or counter clockwise direction *S relates to the direction of spin of an • electron *when 2 electrons occupy the same • orbital, they must have opposite spins

*when an orbital contain 2 electrons the electrons are said to be paired NO ELECTRONS IN AN ATOM CAN HAVE • THE SAME 4 QUANTUM NUMBERS •

7 - Energy levels of electrons *all the electrons in an atom are not located • the same distance from the nucleus *as said in Bohr theory and quantum • mechanics the probability of finding the electrons is greatest at certain specified distance s called energy levels, from the nucleus

*energy levels are also referred to as electron shell and may contain only a limited number of electrons * energy levels are numbered startly with • n=1 to n=7 • Or K, L, M, N, O, P, Q • Where K=1 st energy level, L=2 nd energy level •

*the maximum number of electrons that can occupy a specific energy level =2(nxn) n= number of the principle energy level • E. g. for shell k or energy level 1=2 x(1 x 1)=2 • E. g. for shell l or energy level 2 =2 x(2 x 2)=8 •

8 - energy sublevels of electrons: *the principle energy levels contain sublevels designated by the letters s, p, d, f *s sublevels consists of 1 orbital *p sublevels consists of 3 orbitals * d sublevels consists of 5 orbitals * f sublevels consists of 7 orbitals • • •

The maximum no of electrons that can exist in these sublevels is : S sublevel P sublevel 10 14 2 electrons • 6 • •

al *No more than 2 electrons can *an electron will occupy the lowest • possible sublevel

9 -The atomic number of the element : 1 - the elements are numbered consecutively • from 1 to 106 coinciding with the number of protons in the nucleus 2 -H element number 1 has 1 proton in nucleus • 3 - helium number 2 has 2 protons • Mg 12 protons • The atomic number of an element is the same as • the number of protons in the nucleus , the same as positive charge and also number of electrons in neutral atom.

1 -Hydrogen atomic number of the elements: 1 - The H atom consisting of a nucleus containing • one proton and an electron 2 - orbital containing one electron , is the simplest • known atom 3 - The electron occupies an S orbital in the 1 st • energy level 4 -the electron doesn’t move in any definite path • orbital but rather in a random motion within its forming an electron cloud about the nucleus •

11 - Isotpes of the elements *atoms of an element having the same atomic • number but different atomic masses are called at element • *atoms of the isotopes of an element • , therefore have the same number of protons and elements but different numbers of neutrons

12 -atomic structure of the first twenty elements : * the structure of the atoms of the 1 st 20 • elements , arranged in the order of increasing atomic number (number of protons) *the atoms of each succeeding element contain • one more proton and one more electron than the atoms of the proceeding element. *the number of neutrons in an atom also • increases as we progress from the simpler elements to the more complex one Periodic table page 23 •

Chemical bonding 2 - bonding and molecular structure • • *chemical bond : • The attractive force that hold atoms together • in compounds are called chemical bonding

Bonding types • •

Bonding types Ionic, covalent and metallic bonding • 1 -ionic bond : term given to the electrostatic (charge-based)attractive forces which Hold oppositely charged ions together • • • 2 -Covalent bond : the sharing of electrons • between two atoms that act s to hold the atoms together

*metallic bond : is found in metals. Atoms of • the metal are bound to several neighbors, holding the atoms together but allowing electrons to move freely

Ionic bonding *The ionic bond is the electrostatic force • which attracts particles with opposite electrical charges The formation of ions : • *Atoms can gain or lose electrons to become • charged particles called ions Cations: Are positively charged ions formed • rons

Anions: are negatively charged ions formed when an atom gain electrons An ion is formed when an atom gain or losses • one or more electron M → X + e- → M+ + e– • x- •

*If electron lost by M is gained by x , the overall reaction will be: M + M+ + X- X →M + X - + X- • The ions attracted to each other because they have • opposite charges , the attraction is called an ionic bond or electrovalent bond. •

Lewis structure : *Lewis discover a Lewis structure in which the • chemical symbol for an atom is surrounded by a number of dots corresponding to the number of electrons in the valence shell of the atom. e. g Na atom has one valence –shell-electron • so its Lewis structure is Na. •

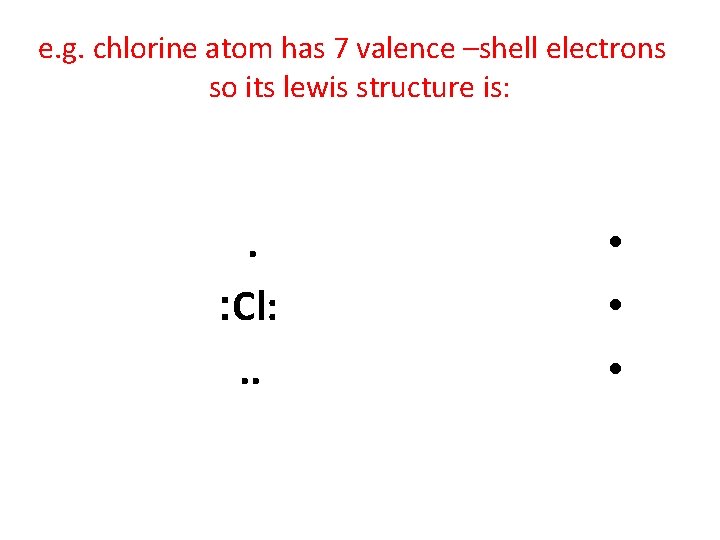
e. g. chlorine atom has 7 valence –shell electrons so its lewis structure is: . : Cl: . . • • •

The symbol = the nucleus plus all the inner shell electrons , It called the core E. g Al=13 • Electronic configration is 1 S 2 2 P 6 3 S 2 3 P 1 • Lewis structure . . Al. Here the valences are shown as a pair (the 2 • e electron (the 3 P) • •

Octet rule : * The octet rule is a statement of the stability • Of the n. S 2 –n. P 6 valence-shell configuration. • Atoms which can achieve this configuration by • the addition of only a few electrons that is, tend to complete the octet. In adding electrons the atom becomes a negative ion. Thus the chloride ion is formed when one electron adds to a chlorine atom.

. . . Cl : . . + e - . . →[ : Cl : ]. . *Here the negative sign is written • because the resulting particle is anion

In positive ions , when has few valence electrons and has an octet in the second shell from the out side , it tend to lose it Valence electrons thereby exposing the octet. • In this way the resulting positive ion ends up • with an octet in what is now its outer shell. Thus the sodium ion tends to lose its valence • Electron to form a sodium ion : • Na(1 S 2 2 P 6 3 S 1)→Na+ (1 S 2 2 P 6 )+e - • Na. → Na + + e- •

Lewis structure and ionic compounds * To write the Lewis structure for an ionic • compound , we write structures for the individual ions. Thus the Lewis structure for Na. Cl is: . . • Na+ [ : Cl : ] - • . . •

Note that: * The octet rule help us to predict • stoichiometry that is , atomic combining ratio in ionic compounds. In the Na. Cl example one electron was transferred from one Na atom to one Cl atom • . . • Na. +. Cl: → Na+ [ : Cl: ] - • . . •

In sodium oxide Oxgyen has only six valence electrons so need • to complete its octet. . : O. . + 2 e - → . . • [ : O : ]2 – • . . •

Because Na atom has only one valence electron to lose so 2 Na atom are requried to furnish two electrons to a single electron Na. . . ↘: O. } → { Na+ [ : O: ] 2 Na. ↗. Na +. . the Lewis structure for sodium oxide can. . be written as 2 Na+ [: O: ] 2. . • •

Write the Lewis structure for calcium chloride Ca in group IIA of the periodic table, has 2 valence electrons, while chlorine in group VIIA And has seven. A calcium atom can by losing its 2 valences electrons, convert 2 Cl atoms to ions. . Ca 2+ 2[ : Cl: ]. . • • •

In this type of bonds One atom has a low ionization energy the • affinity other has a high electron So one or two electron transfer from the first • to the second. to form an ionic bond

Covalent bonding *Covalent bonding occurs when 2 atoms are • more nearly alike in their tendencies to gain and lose electrons. • So outright transfer of electron doesn’t occur. • Instead, electrons are shared between the atoms

Formation of covalent bond : *In H 2 molecule the H and H • There are : • Attractive force between electron of one atom • and the nucleus of the other And • Repulsion between the electron of one atom • and the electron of the other atom

As 2 hydrogen atoms approach each other Each electron begins to ‘sense’ • Electrostatically the presence of the nucleus • Of the opposite atom. • In terms of quantum mechanics this results in • an increase in the probability of finding the 1 st atom’s electron near the second atom’s nucleus and vice versa.

Eventually *Each electron is equally influenced by the 2 • nuclei , and so the probability of finding each electron is the same at each nucleus. * so the 2 electrons occupy the same region of • space. *in any covalent bond the distance between • the nulei of the bonded atoms is called the bond distance or bond length.

Lewis structures and covalent bonding : Covalent bond: a bond formed between 2 • Atoms by sharing of electrons • Lewis structure for H 2 and Cl 2 • H. +H. → H: H = H-H • . . . • : Cl. + : Cl. → : Cl: =: Cl-Cl: • . . . •

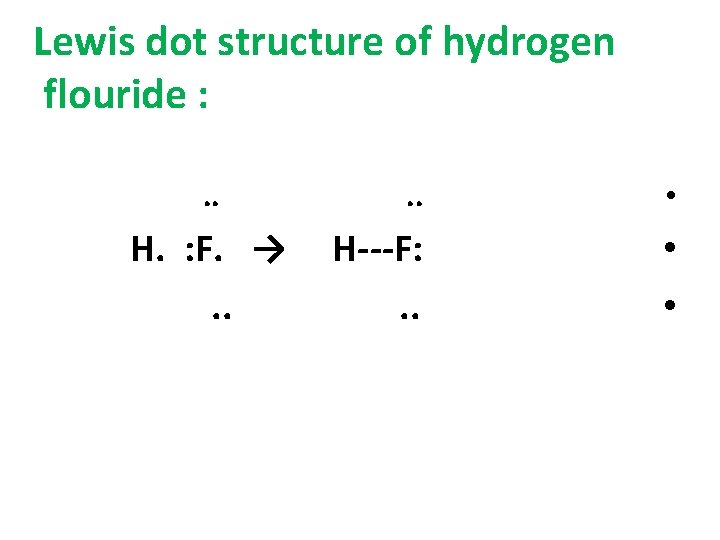
Lewis dot structure of hydrogen flouride : . . H. : F. →. . • H---F: . . • •

Drawing Lewis structure: 1 -Sum the valence electrons from all atoms in • the species 2 - write the atomic symbols for the atoms • involved so as to show which atoms are connected to which , draw a single bond between each pair of bonded atoms 3 - Complete the octets of the atoms bonded • to the central atom

4 - Place leftover electrons on the central atom even if it results in the central atom having more than an octet 5 - If there are not enough electrons to give • the central atom an octet , form multiple bonds by pulling terminal electrons from a peripheral atom and placing them into the bond with the central atom

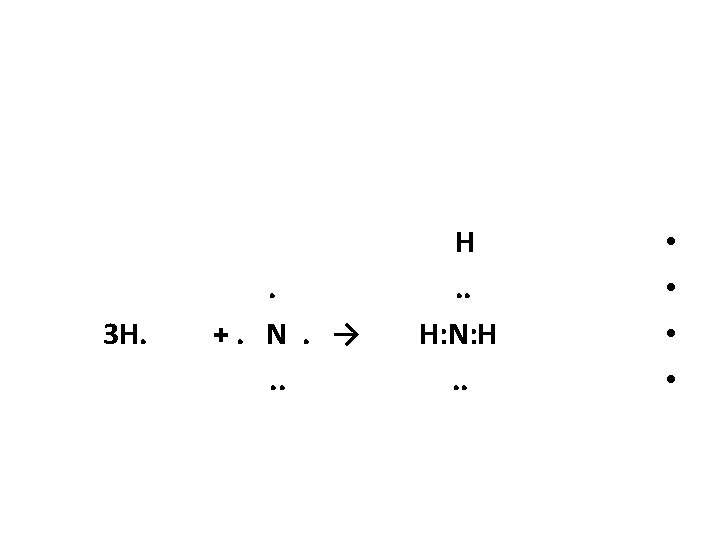
Draw the Lewis structure for ammonia NH 3 * Since each H can form only one covalent • bond, the arrangement of atoms must be : H • HNH • * From the periodic table , N have 5 • valence electrons. These , plus one electron from each H , give a total of 8.

Bonding the atoms in the molecule requires the use of six valence electrons, as : H. . H: N: H The remaining 2 valence electrons are then assigned to N to complete its octet • •


MOLECULAR STRUCTURE 1 -Molecular structure and • covalent bond theories

VALENCE SHELL ELECTRON PAIR REPULSION (VSEPR)THEORY *In a molecule composed of a central atom • bonded covalently to several peripheral atoms the bonding and lone pairs are oriented so that electron-electrons are minimized while electron nucleus attraction are maximized. The method of determining this orientation is called the valence-shell electron-pair Repulsion or VSEPR method. The • assumptionbehind the method are:

1 - electron pairs in the valence shell tend to orient themselves so that their total energy is minimized. This means that they approach the nucleus as • closely as possible, while at the same time • staying as far away from each other as • Possible, thus minimizing interelectronic • repulsions. •

2 -because lone pairs are spread out more broadly than are bonding pairs *repulsions are greatest between two lone • pairs, intermediate between a lone pair and a bonding pair, and weakest between two bonding pairs. Bonding pair bonding pair lone pair → • Increasing repulsion • •

3 -Repulsive forces decrease sharply with increasing interpair angle They are strong at 90 ◦ , much weaker at • 120◦, and very weak at 180◦ •

Steric number and electron –pair orientation: *the first step in the VSEPR method for • determining the shape of a molecule is to draw its Lewis structure in order to find out how many electron pairs are located around the central atom. * consider arsenic trichloride , and sulfur • tetraflouride as example. Their Lewis structures are:


The steric number is defined as : The total number of electron pairs (lone • and bonding) around the central atom. So arsenic has a steric number of 4 in arsenic • trichloride While in sulfur tetraflouride the steric number • of sulfur is 5 (the valence shell of sulfur has been expanded to 10 electrons. )

Special orientation of electrons pairs around a central atom: STERIC NUMBER ORIENTATION ANGLE 2 3 4 5 6 linear 180 120 109. 5 90 -120 90 Triangular planar tetrahedral Trigonal bipyramidal octahedral

Valence – bond theory and orbital overlap: *Two approaches have been used for the • purpose of describing the covalent bond and the electronic structures of molecules. 1 - Valence –bond(VB) theory , consider that • when a pair of atoms forms a bond , the atomic orbitals of each atom remain essentially unchanged and that a pair of electrons occupies an orbital in each of the atoms.

2 -Molecular orbital (MO) *this theory assume that the atomic • orbitals of the original unbonded atoms become replaced by a new set of molecular energy levels called molecular orbitals,

The hydrogen molecule: * the hydrogen molecule formed from 2 • isolated , ground- state hydrogen atoms. *each atom has at start a single electron in its • atomic orbitals. *If we call the two atoms A and B. • *after the covalent bond has been • formed, each electron now exists in the 1 S orbitals of both atoms.


According to valence-bond theory Simultanious occupaucy of orbitals of 2 atoms by a • pair of electrons is possible if the orbitals overlap each other to an appreciable extent. • The orbital overlap produces a region of enhanced • electron probability denisty located directly between the nuclei. *the bond axis (the line connecting the 2 • nuclie)passes through the middle of this region.

the bond in hydrogen is a sigma (Ơ) bond * in which the charge –cloud of the chared • pair is centered on and is symmetrical around • the bond axis. . •

Pi-bonding : When p orbitals overlap sideways , the results • Are different. the resulting side to side overlap • produces enhanced electron probability density in two regions which are on opposite sides of the bond axis. this is characteristic of a pi ( π) bond.

Hybrid orbitals : *Carbon forms countless compounds in which • its atoms bond covalently to 4 other atoms. E. g. methan CH 4 • How can we describe the 4 covalent bonds in • this molecule in terms of orbital overlap?

The ground state electronic configuration of C is • C ↓↑ 1 S ↑↓ ↑ 2 S 2 P ↑ •