Chemistry and Chemical Reactivity 6 th Edition 1



































- Slides: 60

Chemistry and Chemical Reactivity 6 th Edition 1 John C. Kotz Paul M. Treichel Gabriela C. Weaver CHAPTER 3 Molecules, Ions and Their Compounds Lectures written by John Kotz © 2006 Brooks/Cole Thomson © 2006 Brooks/Cole - Thomson

Molecules, Ions & Their Compounds Na. Cl, salt Ethanol, C 2 H 6 O Buckyball, C 60 © 2006 Brooks/Cole - Thomson 2

3 Compounds & Molecules • COMPOUNDS are a combination of 2 or more elements in definite ratios by mass. • The character of each element is lost when forming a compound. • MOLECULES are the smallest unit of a compound that retains the characteristics of the compound. © 2006 Brooks/Cole - Thomson

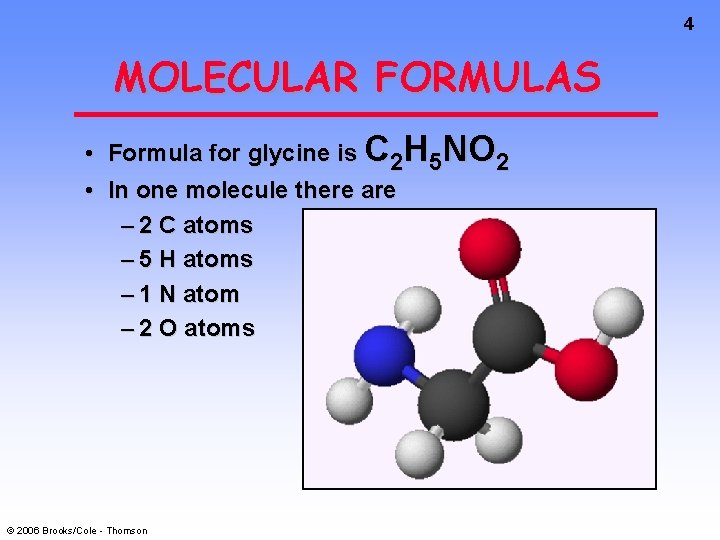
4 MOLECULAR FORMULAS • Formula for glycine is C 2 H 5 NO 2 • In one molecule there are – 2 C atoms – 5 H atoms – 1 N atom – 2 O atoms © 2006 Brooks/Cole - Thomson

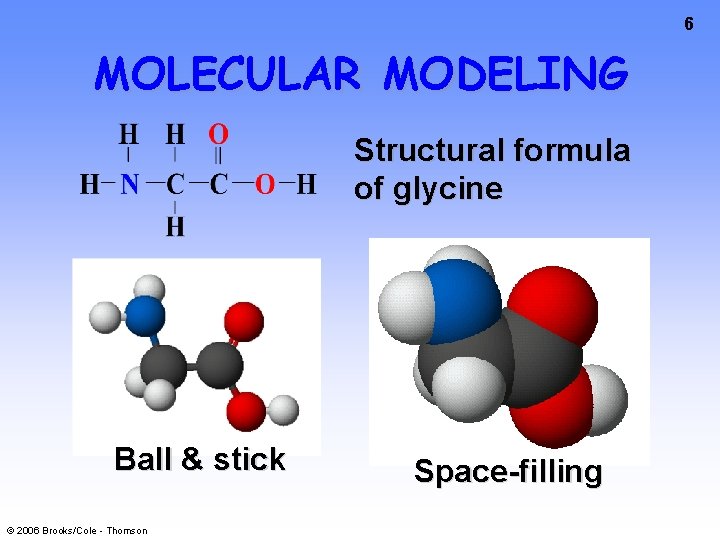
5 WRITING FORMULAS • Can also write glycine formula as –H 2 NCH 2 COOH to show atom ordering • or in the form of a structural © 2006 Brooks/Cole - Thomson formula

6 MOLECULAR MODELING Structural formula of glycine Ball & stick © 2006 Brooks/Cole - Thomson Space-filling

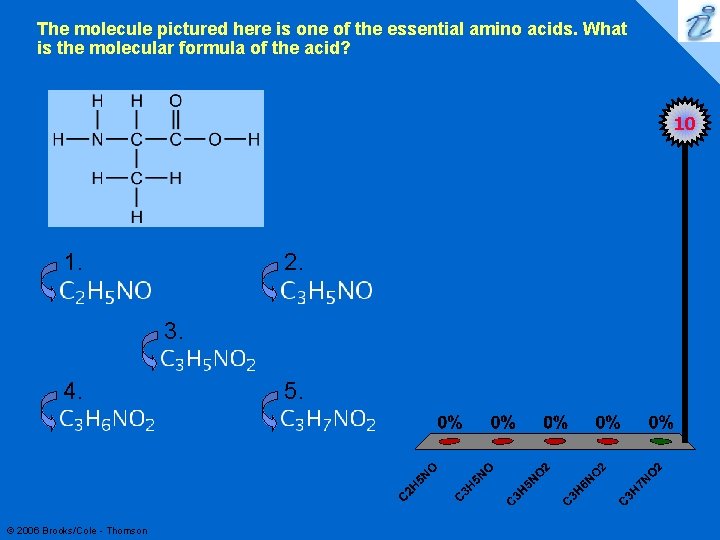
The molecule pictured here is one of the essential amino acids. What is the molecular formula of the acid? 7 10 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson 5.

8 MOLECULAR WEIGHT AND MOLAR MASS Molecular weight = sum of the atomic weights of all atoms in the molecule. Molar mass = molecular weight in grams © 2006 Brooks/Cole - Thomson

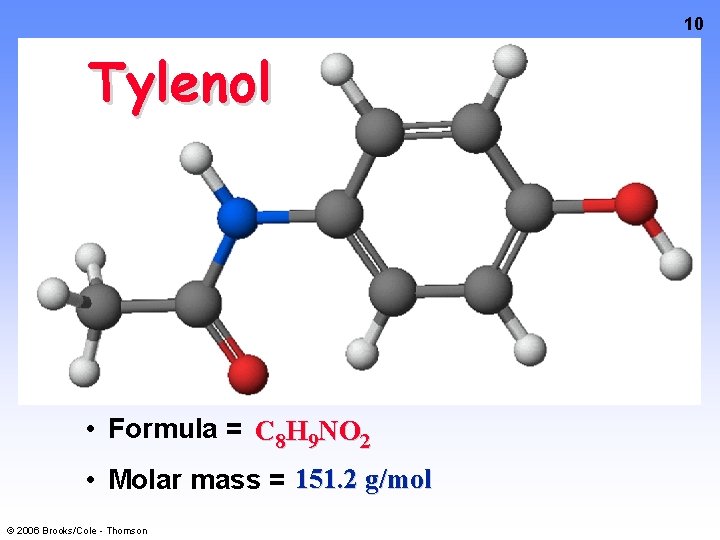
10 Tylenol • Formula = C 8 H 9 NO 2 • Molar mass = 151. 2 g/mol © 2006 Brooks/Cole - Thomson

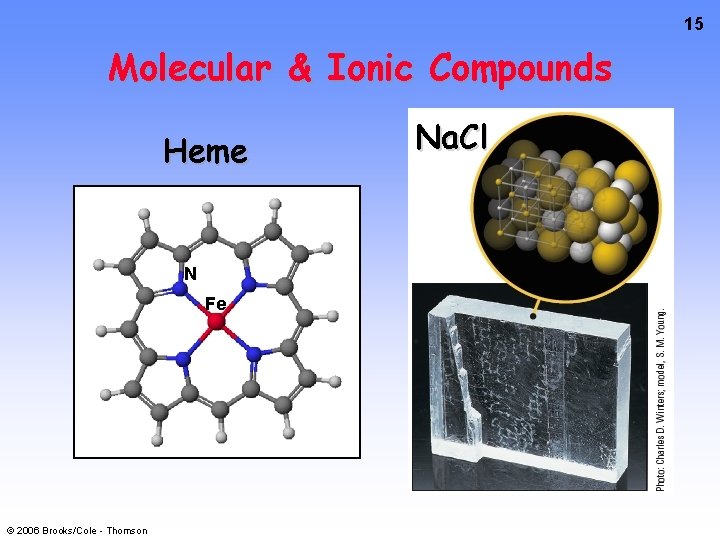
15 Molecular & Ionic Compounds Heme N Fe © 2006 Brooks/Cole - Thomson Na. Cl

16 ELEMENTS THAT EXIST AS MOLECULES Allotropes of C © 2006 Brooks/Cole - Thomson

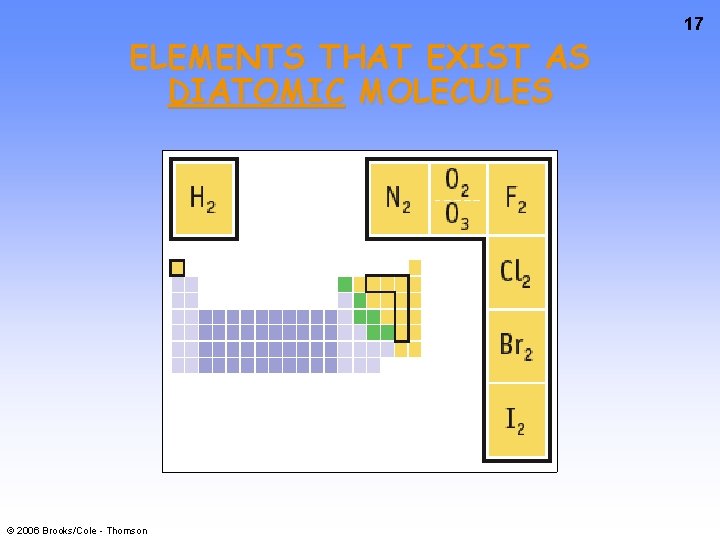
ELEMENTS THAT EXIST AS DIATOMIC MOLECULES © 2006 Brooks/Cole - Thomson 17

ELEMENTS THAT EXIST AS POLYATOMIC MOLECULES 18 S 8 sulfur molecules White P 4 and polymeric red phosphorus © 2006 Brooks/Cole - Thomson

19 IONS AND IONIC COMPOUNDS see Screen 3. 5 • IONS are atoms or groups of atoms with a positive or negative charge. • Taking away an electron from an atom gives a CATION with a positive charge • Adding an electron to an atom gives an ANION with a negative charge. © 2006 Brooks/Cole - Thomson

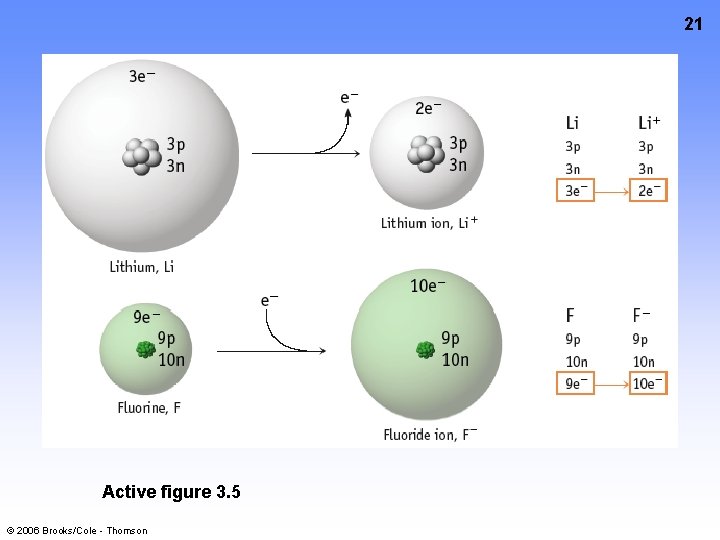
20 Forming Cations & Anions A CATION forms when an atom loses one or more electrons. An ANION forms when an atom gains one or more electrons Mg 2+ F + e- --> F- Mg --> © 2006 Brooks/Cole - Thomson + 2 e-

21 Active figure 3. 5 © 2006 Brooks/Cole - Thomson

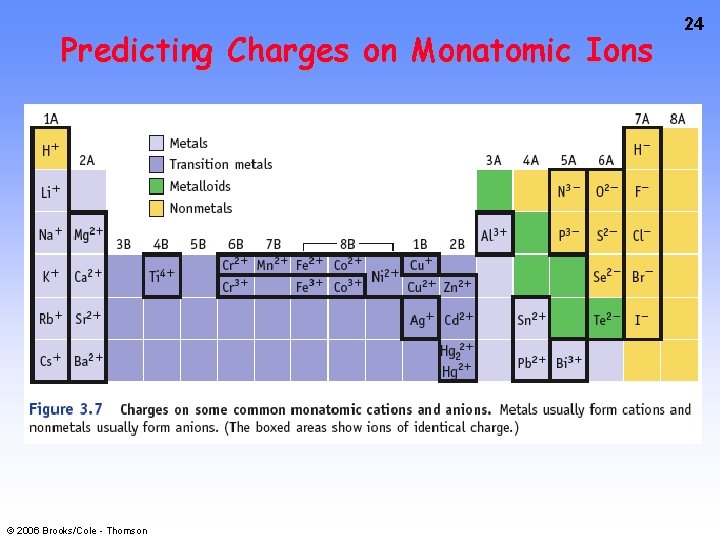
22 PREDICTING ION CHARGES • See CD-ROM Screen 3. 5 and book Figure 3. 7 In general • metals (Mg) lose electrons ---> cations • nonmetals (F) gain electrons ---> anions © 2006 Brooks/Cole - Thomson

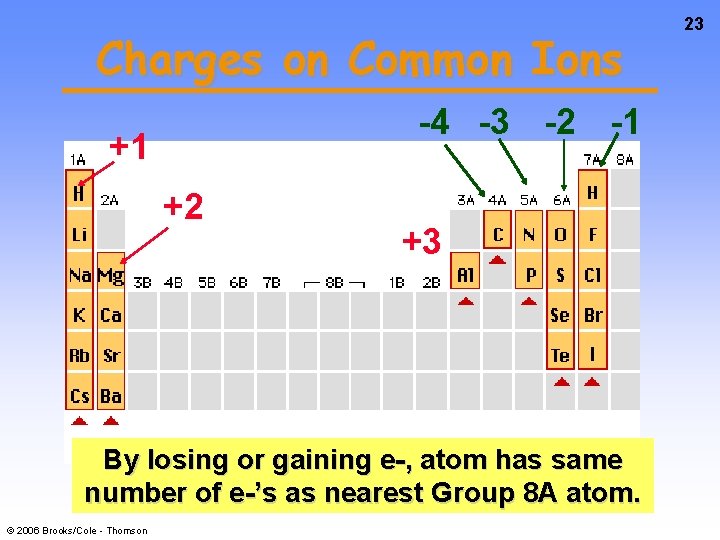
Charges on Common Ions -4 -3 -2 -1 +1 +2 +3 By losing or gaining e-, atom has same number of e-’s as nearest Group 8 A atom. © 2006 Brooks/Cole - Thomson 23

Predicting Charges on Monatomic Ions © 2006 Brooks/Cole - Thomson 24

Which ion in the following list is NOT likely to form? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson 5. 25

26 1. 2. 3. 4. 5. © 2006 Brooks/Cole - Thomson 25 protons and 25 electrons. 25 protons and 26 electrons. 25 protons and 24 electrons. 26 protons and 25 electrons. 26 protons and 26 electrons.

METALS M ---> n e- + Mn+ where n = periodic group Na+ sodium ion Mg 2+ magnesium ion Al 3+ aluminum ion Transition metals --> M 2+ or M 3+ are common Fe 2+ iron(II) ion Fe 3+ iron(III) ion © 2006 Brooks/Cole - Thomson 27

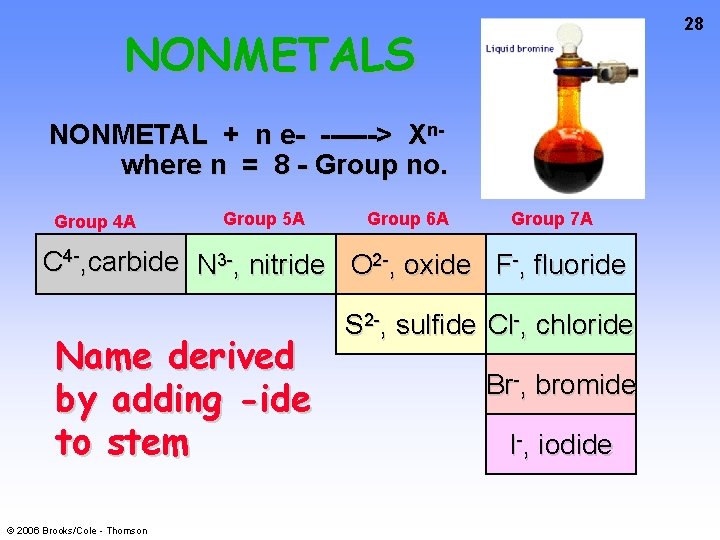
28 NONMETALS NONMETAL + n e- ------> Xnwhere n = 8 - Group no. Group 4 A Group 5 A Group 6 A Group 7 A C 4 -, carbide N 3 -, nitride O 2 -, oxide F-, fluoride Name derived by adding -ide to stem © 2006 Brooks/Cole - Thomson S 2 -, sulfide Cl-, chloride Br-, bromide I-, iodide

29 Ion Formation Reaction of aluminum and bromine © 2006 Brooks/Cole - Thomson

COMPOUNDS FORMED FROM IONS 30 CATION + ANION ---> COMPOUND Na+ + Cl- --> Na. Cl A neutral compd. requires equal number of + and - charges. © 2006 Brooks/Cole - Thomson

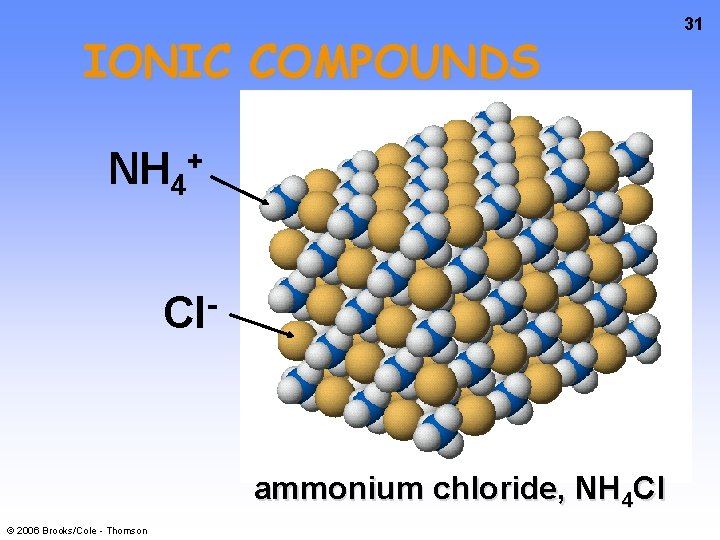
IONIC COMPOUNDS NH 4 + Cl ammonium chloride, NH 4 Cl © 2006 Brooks/Cole - Thomson 31

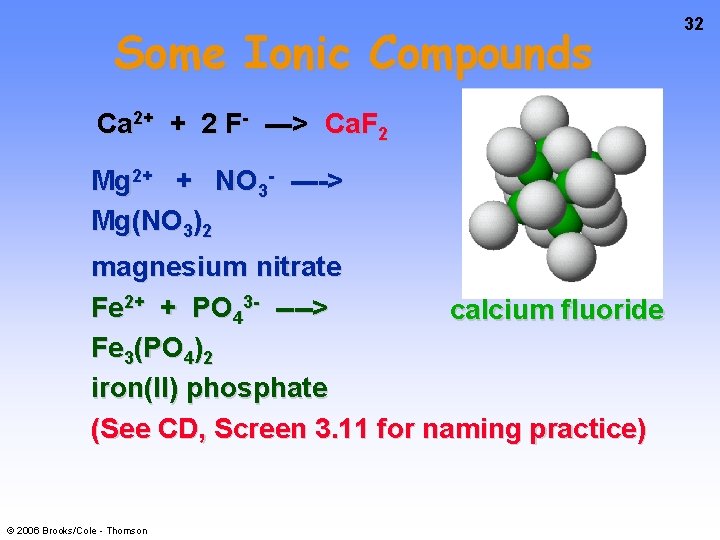
Some Ionic Compounds Ca 2+ + 2 F- ---> Ca. F 2 Mg 2+ + NO 3 - ----> Mg(NO 3)2 magnesium nitrate Fe 2+ + PO 43 - ----> calcium fluoride Fe 3(PO 4)2 iron(II) phosphate (See CD, Screen 3. 11 for naming practice) © 2006 Brooks/Cole - Thomson 32

Properties of Ionic Compounds Forming Na. Cl from Na and Cl 2 • A metal atom can transfer an electron to a nonmetal. • The resulting cation and anion are attracted to each other by electrostatic forces. © 2006 Brooks/Cole - Thomson 33

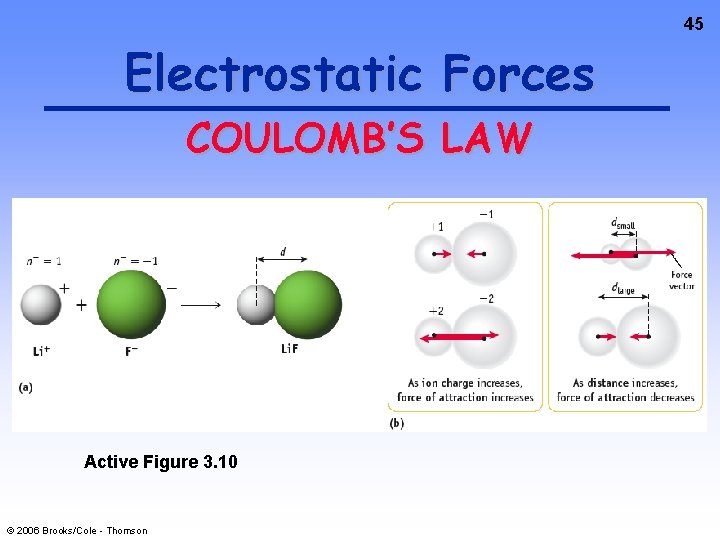
34 Electrostatic Forces The oppositely charged ions in ionic compounds are attracted to one another by ELECTROSTATIC FORCES. These forces are governed by COULOMB’S LAW. © 2006 Brooks/Cole - Thomson

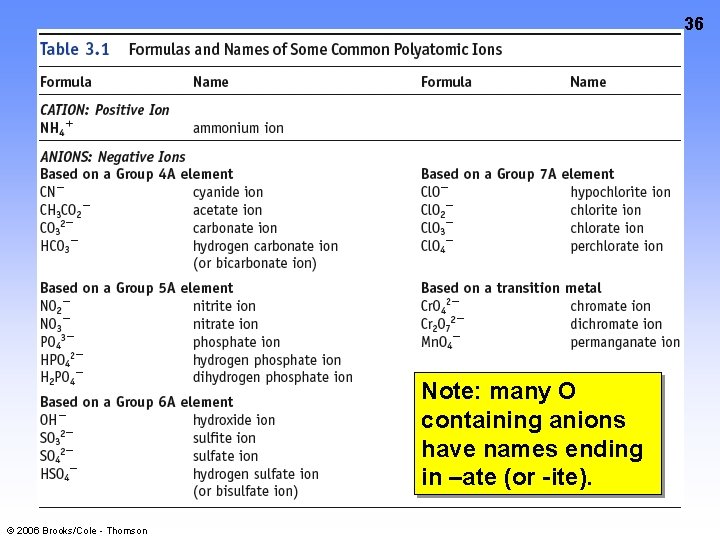
35 POLYATOMIC IONS CD Screen 3. 6 Groups of atoms with a charge. MEMORIZE the names and formulas in Table 3. 1, page 107. © 2006 Brooks/Cole - Thomson

36 Note: many O containing anions have names ending in –ate (or -ite). © 2006 Brooks/Cole - Thomson

Which compound in the following list is NOT possible? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson 5. 37

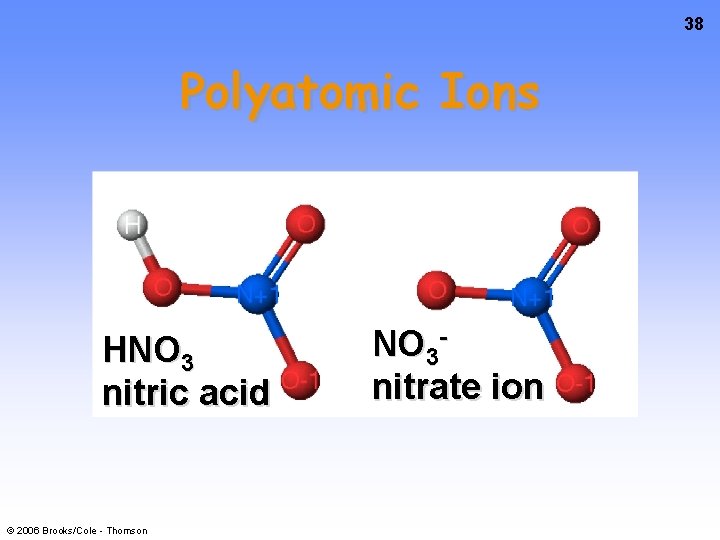
38 Polyatomic Ions HNO 3 nitric acid © 2006 Brooks/Cole - Thomson NO 3 nitrate ion

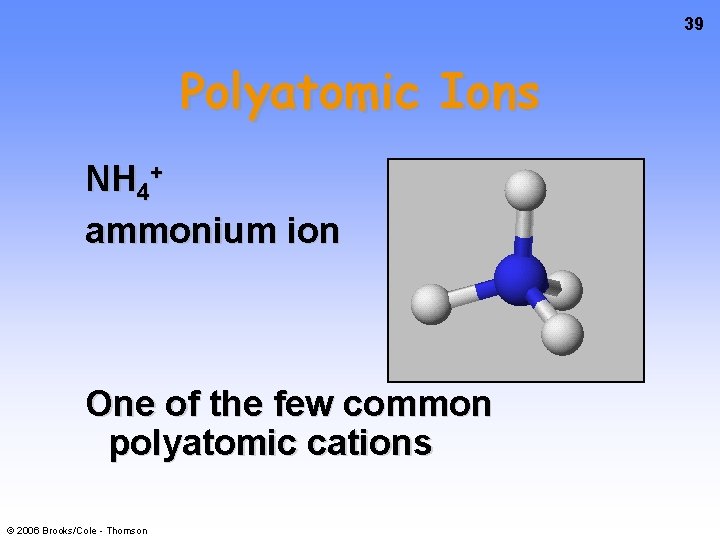
39 Polyatomic Ions NH 4+ ammonium ion One of the few common polyatomic cations © 2006 Brooks/Cole - Thomson

40 Polyatomic Ions CO 32 carbonate ion HCO 3 bicarbonate ion hydrogen carbonate © 2006 Brooks/Cole - Thomson

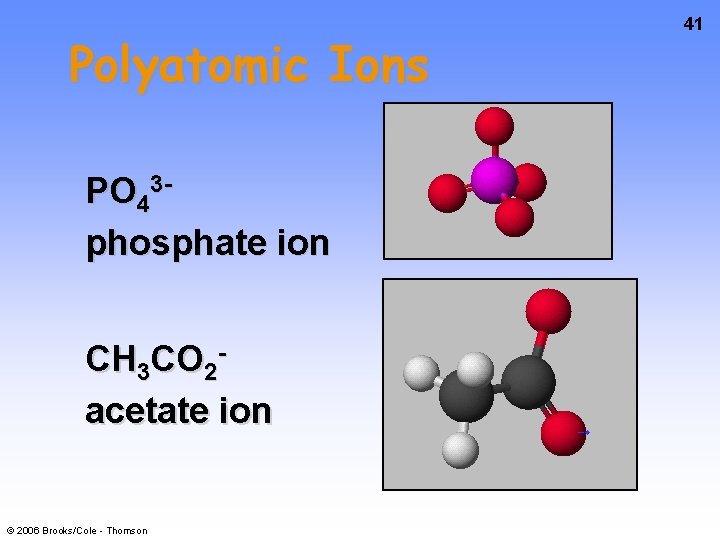
Polyatomic Ions PO 43 phosphate ion CH 3 CO 2 acetate ion © 2006 Brooks/Cole - Thomson 41

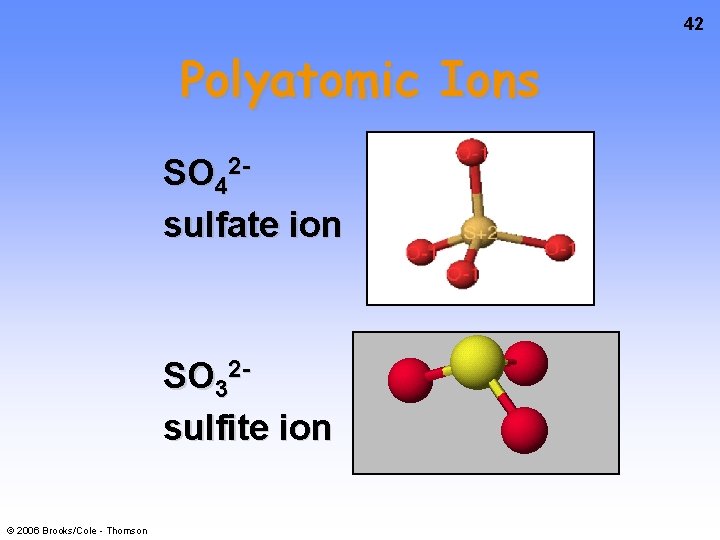
42 Polyatomic Ions SO 42 sulfate ion SO 32 sulfite ion © 2006 Brooks/Cole - Thomson

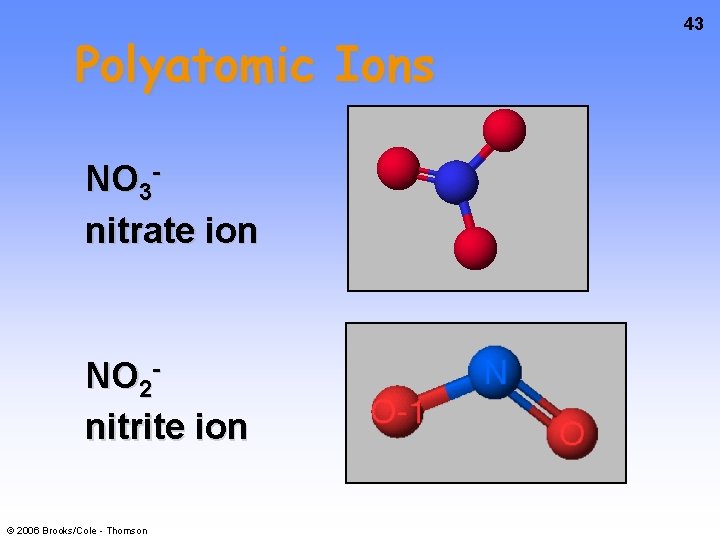
Polyatomic Ions NO 3 nitrate ion NO 2 nitrite ion © 2006 Brooks/Cole - Thomson 43

44 Electrostatic Forces COULOMB’S LAW As ion charge increases, the attractive force ________. As the distance between ions increases, the attractive force ________. This idea is important and will come up many times in future discussions! © 2006 Brooks/Cole - Thomson

45 Electrostatic Forces COULOMB’S LAW Active Figure 3. 10 © 2006 Brooks/Cole - Thomson

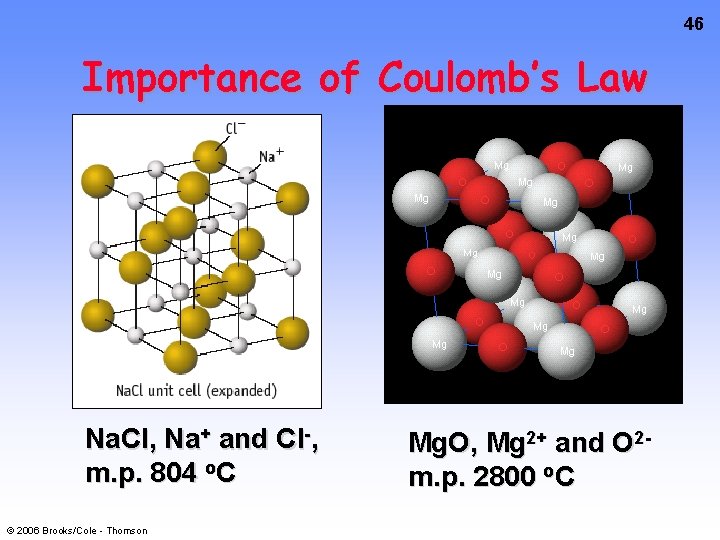
46 Importance of Coulomb’s Law Na. Cl, Na+ and Cl-, m. p. 804 o. C © 2006 Brooks/Cole - Thomson Mg. O, Mg 2+ and O 2 m. p. 2800 o. C

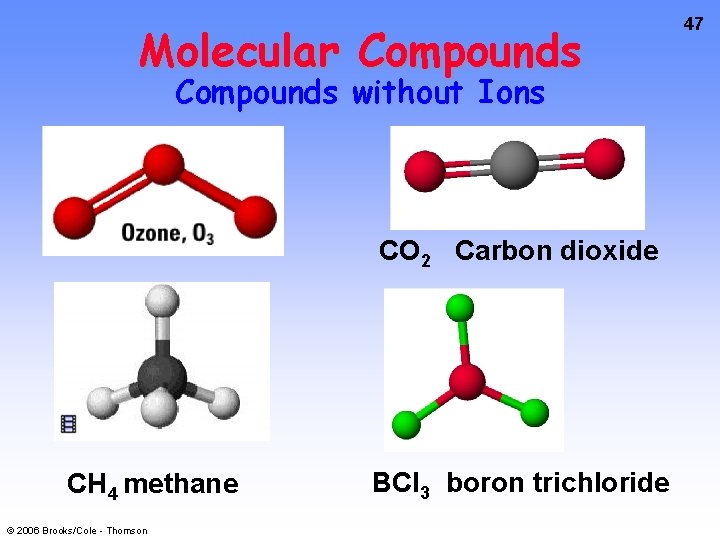
Molecular Compounds without Ions CO 2 Carbon dioxide CH 4 methane © 2006 Brooks/Cole - Thomson BCl 3 boron trichloride 47

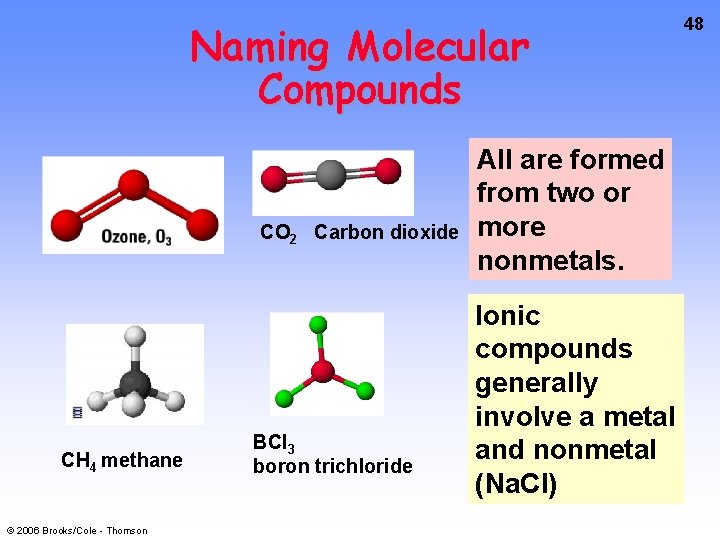
Naming Molecular Compounds CO 2 Carbon dioxide CH 4 methane © 2006 Brooks/Cole - Thomson BCl 3 boron trichloride All are formed from two or more nonmetals. Ionic compounds generally involve a metal and nonmetal (Na. Cl) 48

49 Empirical & Molecular Formulas A pure compound always consists of the same elements combined in the same proportions by weight. Therefore, we can express molecular composition as PERCENT BY WEIGHT Ethanol, C 2 H 6 O 52. 13% C 13. 15% H 34. 72% O © 2006 Brooks/Cole - Thomson

Percent Composition Consider some of the family of nitrogenoxygen compounds: NO 2, nitrogen dioxide and closely related, NO, nitrogen monoxide (or nitric oxide) Chemistry of NO, nitrogen monoxide Structure of NO 2 © 2006 Brooks/Cole - Thomson 50

51 Percent Composition Consider NO 2, Molar mass = ? What is the weight percent of N and of O? What are the weight percentages of N and O in NO? © 2006 Brooks/Cole - Thomson

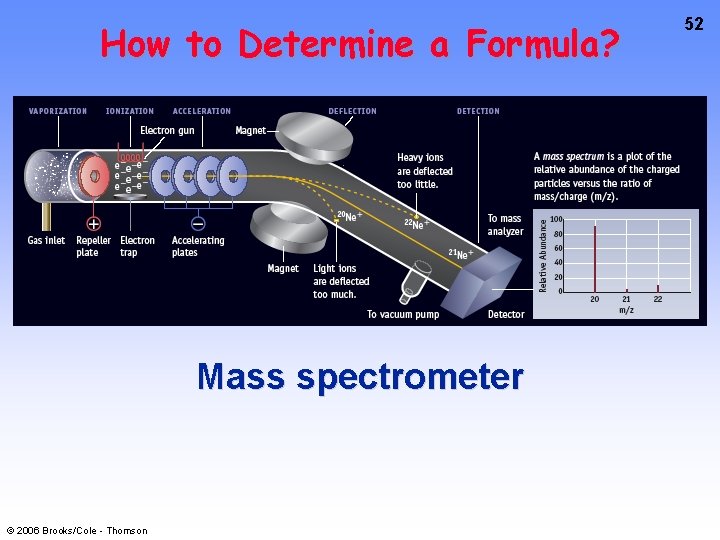
How to Determine a Formula? Mass spectrometer © 2006 Brooks/Cole - Thomson 52

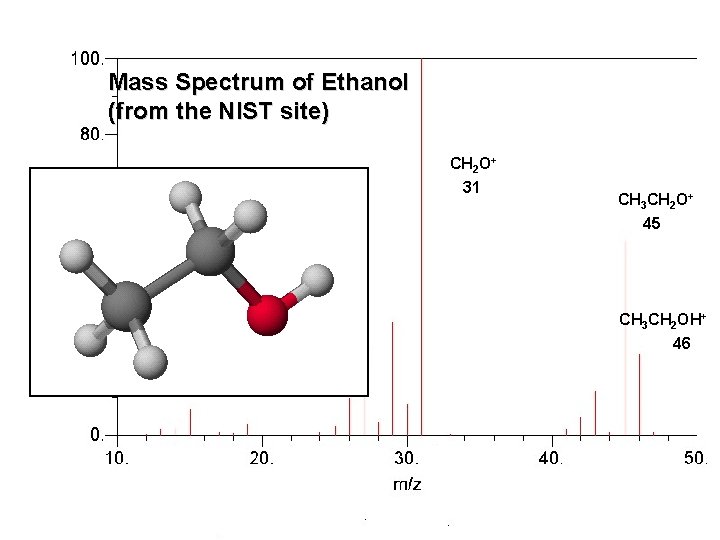
Mass Spectrum of Ethanol 53 (from the NIST site) CH 2 O+ 31 CH 3 CH 2 O+ 45 CH 3 CH 2 OH+ 46 © 2006 Brooks/Cole - Thomson

54 Determining Formulas In chemical analysis we determine the % by weight of each element in a given amount of pure compound and derive the EMPIRICAL or SIMPLEST formula. PROBLEM: A compound of B and H is 81. 10% B. What is its empirical formula? © 2006 Brooks/Cole - Thomson

55 A compound of B and H is 81. 10% B. What is its empirical formula? • Because it contains only B and H, it must contain 18. 90% H. • In 100. 0 g of the compound there are 81. 10 g of B and 18. 90 g of H. • Calculate the number of moles of each constitutent. © 2006 Brooks/Cole - Thomson

56 A compound of B and H is 81. 10% B. What is its empirical formula? Calculate the number of moles of each element in 100. 0 g of sample. © 2006 Brooks/Cole - Thomson

57 A compound of B and H is 81. 10% B. What is its empirical formula? Now, recognize that atoms combine in the ratio of small whole numbers. 1 atom B + 3 atoms H --> 1 molecule BH 3 or 1 mol B atoms + 3 mol H atoms ---> 1 mol BH 3 molecules Find the ratio of moles of elements in the compound. © 2006 Brooks/Cole - Thomson

58 A compound of B and H is 81. 10% B. What is its empirical formula? Take the ratio of moles of B and H. Always divide by the smaller number. But we need a whole number ratio. 2. 5 mol H/1. 0 mol B = 5 mol H to 2 mol B EMPIRICAL FORMULA = B 2 H 5 © 2006 Brooks/Cole - Thomson

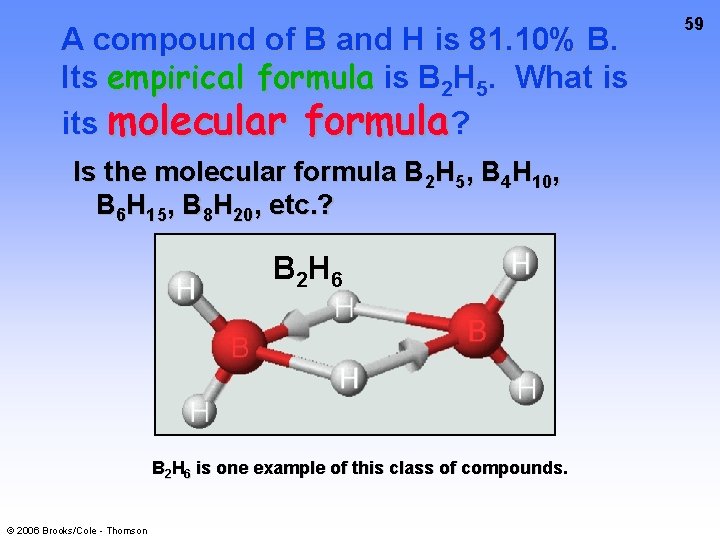
A compound of B and H is 81. 10% B. Its empirical formula is B 2 H 5. What is its molecular formula? Is the molecular formula B 2 H 5, B 4 H 10, B 6 H 15, B 8 H 20, etc. ? B 2 H 6 is one example of this class of compounds. © 2006 Brooks/Cole - Thomson 59

A compound of B and H is 81. 10% B. Its empirical formula is B 2 H 5. What is its molecular formula? We need to do an EXPERIMENT to find the MOLAR MASS. Here experiment gives 53. 3 g/mol Compare with the mass of B 2 H 5 = 26. 66 g/unit Find the ratio of these masses. Molecular formula = B 4 H 10 © 2006 Brooks/Cole - Thomson 60

61 DETERMINE THE FORMULA OF A COMPOUND OF Sn AND I Sn(s) + some I 2(s) ---> Sn. Ix © 2006 Brooks/Cole - Thomson

62 Data to Determine the formula of a Sn—I Compound • Reaction of Sn and I 2 is done using excess Sn. • Mass of Sn in the beginning = 1. 056 g • Mass of iodine (I 2) used = 1. 947 g • Mass of Sn remaining = 0. 601 g • See p. 125 © 2006 Brooks/Cole - Thomson

63 Tin and Iodine Compound Find the mass of Sn that combined with 1. 947 g I 2. Mass of Sn initially = 1. 056 g Mass of Sn recovered = 0. 601 g Mass of Sn used = 0. 455 g Find moles of Sn used: © 2006 Brooks/Cole - Thomson

64 Tin and Iodine Compound Now find the number of moles of I 2 that combined with 3. 83 x 10 -3 mol Sn. Mass of I 2 used was 1. 947 g. How many mol of iodine atoms? = 1. 534 x 10 -2 mol I atoms © 2006 Brooks/Cole - Thomson

65 Tin and Iodine Compound Now find the ratio of number of moles of I and Sn that combined. Empirical formula is © 2006 Brooks/Cole - Thomson Sn. I 4