Chapter 12 Basics of Chemistry Learning Objectives List

Chapter 12 Basics of Chemistry

Learning Objectives • List the difference between organic and inorganic chemistry. • Categorize and give examples of different substances for each of the different states of matter: solid, liquid, and gas. • Summarize, in your own words, oxidation– reduction (redox) reactions. (continues)

Learning Objectives (continued) • Define the differences between pure substances and physical mixtures. • Evaluate the differences among solutions, suspensions, and emulsions. • Explain what p. H is and how the p. H scale works.

Introduction • Most cosmetology services depend on the use of chemicals. • Studying the basics of chemistry means that you will have the knowledge you need to understand the products that you are using in the salon to give your clients the professional services they deserve.


Recognize How the Science of Chemistry Influences Cosmetology Chemistry • The science that deals with the composition, structures, and properties of matter and how matter changes under different conditions

Organic Chemistry • The study of substances that contain carbon. • All living or formerly living things contain carbon. • All hair color products, chemical texturizers, shampoos, conditioners, styling aids, nail enhancements, and skin care products are organic chemicals.

Inorganic Chemistry • The study of substances that do not contain the element carbon but may contain the element hydrogen • Metals, minerals, glass, water, air, pure water, oxygen, hydrogen peroxide, hydroxide hair relaxers, titanium dioxide, and zinc oxide in sun protection creams are inorganic substances.

Define Matter • Anything that occupies space • Has physical chemical properties • Exists in the form of a solid, liquid, or gas

Elements • Simplest form of matter • Cannot be reduced without loss of identity • At least 98 naturally occurring • Identified by a letter symbol

Atoms and Molecules • Atom – the basic unit of matter • Molecules – the chemical combination of two or more atoms – Elemental molecule – Compound molecule

States of Matter

Physical and Chemical Properties of Matter • Physical properties – determined without a chemical reaction (color, odor, weight, density) • Chemical properties – determined with a chemical reaction (change identity — rusting iron, burning wood)
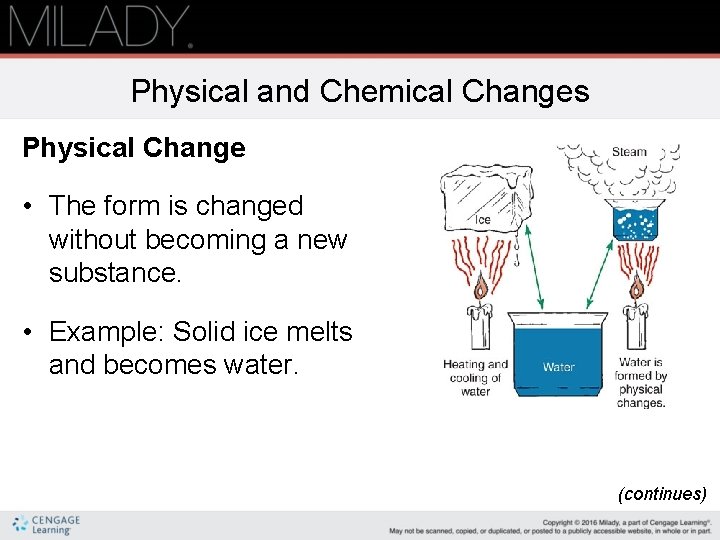
Physical and Chemical Changes Physical Change • The form is changed without becoming a new substance. • Example: Solid ice melts and becomes water. (continues)

Physical and Chemical Changes (continued) Chemical Change • A change in the chemical composition or makeup of a substance • Example: Burning wood turns into ashes

Oxidation • A chemical reaction that combines a substance with oxygen to produce an oxide • Example: Wood turns into charcoal after it has burned.

Oxidation-Reduction • Also known as redox • A chemical reaction in which the oxidizing agent is reduced (by losing oxygen) and the reducing agent is oxidized (by gaining oxygen)

Oxidizing Agent • A substance that releases oxygen • Hydrogen peroxide (an oxidizing agent) can be thought of as water with an extra atom of oxygen.

Reducing Agent • A substance that adds hydrogen to a chemical compound or subtracts oxygen from the compound

Exothermic Reaction and Combustion • Exothermic reaction – a chemical reaction when heat is released • Combustion – rapid oxidation of a substance accompanied by the production of heat and light

Pure Substances and Physical Mixtures • Pure substances – distinct properties • Physical mixtures – elements combined physically

Solutions, Suspensions, and Emulsions • Solution – a blend of two or more liquids or a solid dissolved in a liquid • Solute – the dissolved substance in a solution • Solvent – the substance that dissolves another substance to form a solution with no change in chemical composition

Solutions and Suspensions • Miscible liquids – mutually soluble • Immiscible liquids – not capable of being mixed • Suspension – unstable physical mixtures of undissolved particles in a liquid

Emulsions and Emulsifiers • Emulsion – mixture of two or more immiscible substances plus a special ingredient called an emulsifier • Emulsifier – ingredient that brings two normally incompatible materials together and binds them into a uniform and fairly stable blend

Surfactants • Hydrophilic – water loving • Lipophilic – oil loving • Surfactant – joins water and oil • Oil-in-water emulsion • Water-in-oil emulsion

Differences Among Solutions, Suspensions, and Emulsions


Other Physical Mixtures • Ointments – semisolids made with any combination of petrolatum, oil, and wax • Powders – physical mixture of two solids

Common Chemical Product Ingredients • • • Volatile alcohols Alkanolamines Ammonia Glycerin Silicones Volatile organic compounds

Understand Potential Hydrogen (p. H) and How it Affects Hair, Skin, and Nails • The small p represents a quantity; the capital H represents the hydrogen ion • Ion – atom or molecule that carries an electrical charge • Ionization – temporary separation of a substance into ions • Anion – ion with a negative electrical charge • Cation – ion with a positive electrical charge

Water and p. H • In pure water, some of the water molecules naturally ionize into hydrogen ions and some into hydroxide ions. The p. H scale measures those ions. • Hydrogen ion (H+) is acidic. • Hydroxide ion (OH-) is alkaline.
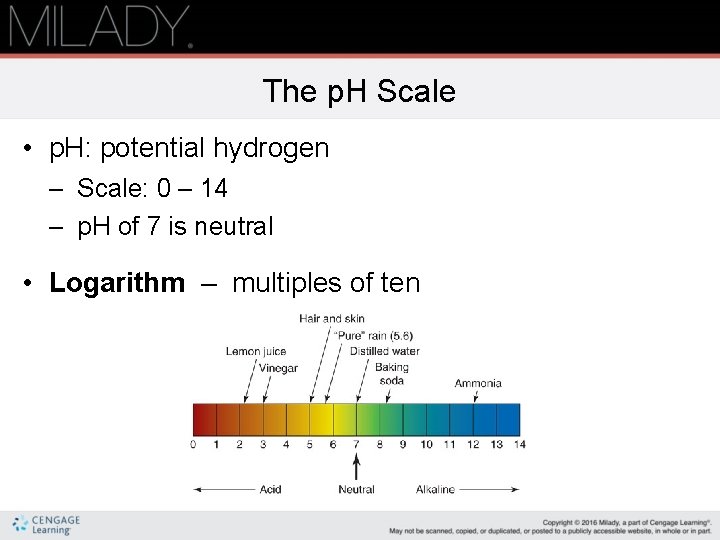
The p. H Scale • p. H: potential hydrogen – Scale: 0 – 14 – p. H of 7 is neutral • Logarithm – multiples of ten

Acids and Alkalis • All acids owe their chemical reactivity to the hydrogen ion. • Acids have a p. H below 7. 0. – Alpha hydroxy acids (AHAs) – Thioglycolic acid – Glycolic acid (continues)

Acids and Alkalis (continued) • Alkalis (base) owe their chemical relativity to the hydroxide ion (OH-). • Alkalis have a p. H above 7. – Feel slippery and soapy on the skin. – Sodium hydroxide also known as lye, is used in chemical hair relaxers, callous softeners, and drain cleaners.

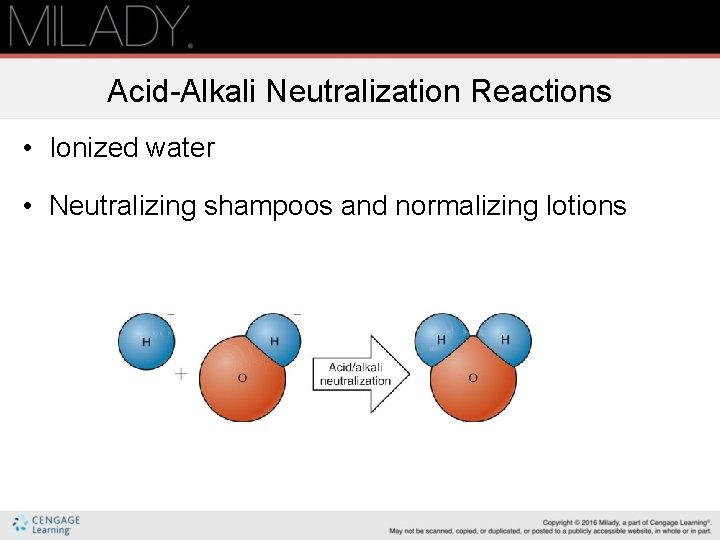
Acid-Alkali Neutralization Reactions • Ionized water • Neutralizing shampoos and normalizing lotions

Summary and Review • The science that deals with the composition, structure, and properties of matter and how matter changes under different chemical conditions is called chemistry. • Organic chemistry deals with all substances in which carbon is present, while inorganic chemistry deals with substances that do not contain carbon. (continues)

Summary and Review (continued) • Matter is anything that occupies space, has physical and chemical properties, and exists as a solid, a liquid, or a gas. Matter exists in the form of elements, compounds, and mixtures. Matter can be changed either physically or chemically. • There are many benefits for the client who takes advantage of the various salon services that use chemical products. While the use of chemical products has great benefits, we must always remember they create a potential for damage or injury as well. (continues)

Summary and Review (continued) • As a professional cosmetologist, your ability to stay informed about new developments and products and how to use them effectively and safely will greatly impact your success.

Chapter Review Questions 1. What is chemistry? 2. Why is a basic understanding of chemistry important to a cosmetologist? 3. What is the difference between organic and inorganic chemistry? (continues)

Chapter Review Questions (continued) 4. What is matter? 5. What is an element? 6. What are atoms? 7. Explain the difference between elemental molecules and compound molecules. Give examples. (continues)

Chapter Review Questions (continued) 8. Name and describe three states of matter. 9. What are the physical and chemical properties of matter? Give examples. 10. What is the difference between physical and chemical change? Give examples. (continues)

Chapter Review Questions (continued) 11. Explain oxidation-reduction (redox). 12. Explain pure substances and physical mixtures. Give examples. 13. What are the differences among solutions, suspensions, and emulsions? Give examples. 14. Define p. H and the p. H scale.
- Slides: 43