AP Chemistry Chemical Equilibrium Chemical equilibrium is reached

AP Chemistry Chemical Equilibrium
Chemical equilibrium is reached when the concentrations of reactants and products cease changing with time. reactants products these RATES are equal; NOT amt. of R = amt. of P -- system must be closed -- equilibrium is a dynamic process (although it might look static)

For the reaction A B: -- Eq. can be reached starting with… either A (i. e. , A B) or with B (i. e. , A B). Eventually, A B is reached.
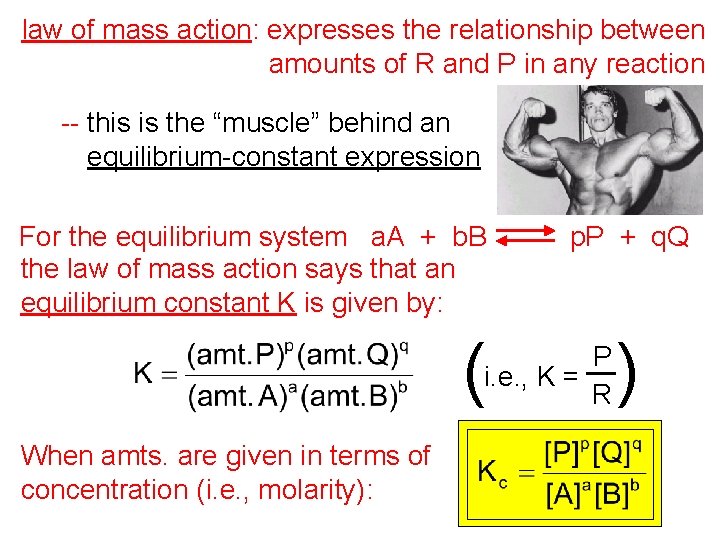
law of mass action: expresses the relationship between amounts of R and P in any reaction -- this is the “muscle” behind an equilibrium-constant expression For the equilibrium system a. A + b. B the law of mass action says that an equilibrium constant K is given by: ( p. P + q. Q P i. e. , K = R When amts. are given in terms of concentration (i. e. , molarity): )

For a gaseous system: where PX is the partial pressure of X at PX values must eq. , and Kp is the pressure eq. constant be in atm. (!) The relationship between Kc and Kp is given by. . . Kp = Kc(RT)Dn Dn = SPcoeff – SRcoeff R = universal gas const. 0. 08206 L-atm/mol-K T = absolute temp.

For the gaseous reaction 2 NO + O 2 2 NO 2 (1) write the Kc and Kp expressions (2) find Kp at T = 750 o. C, if Kc = 2. 19 x 10– 3. [NO 2]2 (1) Kc = [NO]2 [O 2] (2) Kp = When a flask filled with colorless NO is opened to the air, O 2 enters, resulting in brown NO 2. (PNO 2)2 (PNO)2 (PO 2) T = 1023 K Kp = Kc(RT)Dn = 2. 19 x 10– 3 [0. 08206(1023)]– 1 = 2. 61 x 10– 5

A few notes about K: -- it depends only on the reaction stoichiometry, not its mechanism -- it is independent of initial concentrations -- it is unaffected by other substances, as long as they don’t react with R or P -- it varies with temperature -- it is written without units -- it NEVER includes pure liquids or pure solids
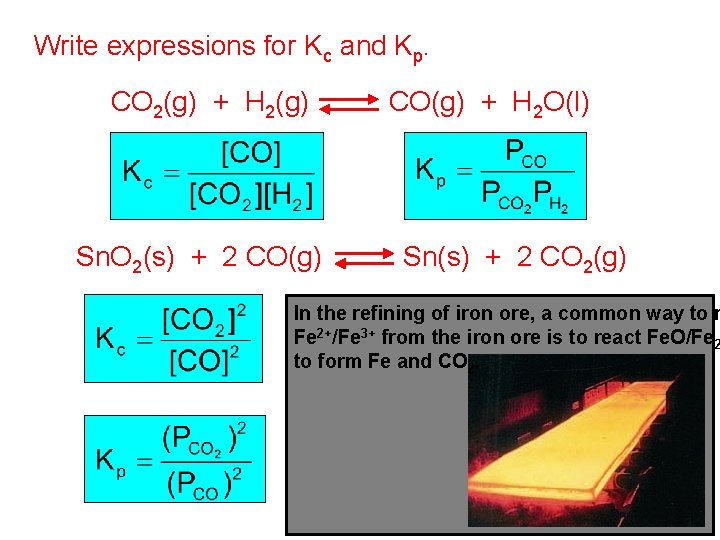
Write expressions for Kc and Kp. CO 2(g) + H 2(g) Sn. O 2(s) + 2 CO(g) + H 2 O(l) Sn(s) + 2 CO 2(g) In the refining of iron ore, a common way to r Fe 2+/Fe 3+ from the iron ore is to react Fe. O/Fe 2 to form Fe and CO 2.

Write the Kc expression for each reaction. ** substances w/only 1 or 2 nonmetals: best guess = gases N 2 + 3 H 2 2 SO 3 2 NH 3 2 SO 2 + Fritz Haber (1868– 1934) discovered to generate ammonia from O 2 a way hydrogen and nitrogen at high pressure. The ammonia was needed for Germany’s munitions industry, which was cut off from the nitrate sources of South America by the British blockade during WWI.

Based on Ca. CO 3(s) Ca. O(s) + CO 2(g), which of the following can attain equilibrium? Start with. . . (a). . . pure Ca. CO 3 “YEP. ” (Kp = PCO 2) (it breaks down, forming products until the amt. of CO 2 is (b). . . Ca. O and some CO 2 at P > Kp “YEP. ” (too much CO 2; it reacts w/available Ca. O until amt. of CO (c). . . Ca. CO 3 and some CO 2 at P > Kp “NOPE. ” (too much CO 2, but no Ca. O for it to react with in order to “right” amt. of CO 2) (d). . . Ca. CO 3 and Ca. O “YEP. ”

The Magnitude of the Equilibrium Constant If K >> 1. . . products are favored. Eq. “lies to the right. ” If K << 1. . . reactants are favored. Eq. “lies to the left. ” The K for the forward and reverse reactions are NOT the same. -- they are reciprocals -- You must write out the equation and specify the temperature when reporting a K.

Calculating Equilibrium Constants (1) If the concentrations of all substances at equilibrium are known, plug and chug. Find Kc @ 472 o. C for N 2(g) + 3 H 2(g) 2 NH 3(g) At equilibruim @ 472 o. C…[NH 3] = 0. 00272 M [N 2] = 0. 0402 M [H 2] = 0. 1207 M. = 0. 105

(2) If you know the concentrations of only some substances at equilibrium, make a chart and use reaction stoichiometry to figure out the other concentrations at equilibrium. THEN plug and chug. “Ice, ice, baby…” I = “initial” C = “change” E = “equilibrium”

At 1000 K, the amounts shown below are known. Find Kc @ 1000 K. initial 2 SO 3(g) 6. 09 x 10– 3 M D – 3. 65 x 10– 3 M at eq. 2. 44 x 10– 3 M 2 SO 2(g) 0 M +3. 65 x 10– 3 M +1. 825 x Put “at eq. ” #s into Kc expression… = 4. 08 x 10– 3

At 1000 K, the amounts shown below are known. Find Kc @ 1000 K. initial 2 SO 3(g) 6. 09 x 10– 3 M D – 3. 65 x 10– 3 M at eq. 2. 44 x 10– 3 M 2 SO 2(g) 0 M +3. 65 x 10– 3 M +1. 825 x Put “at eq. ” #s into Kc expression… = 4. 08 x 10– 3
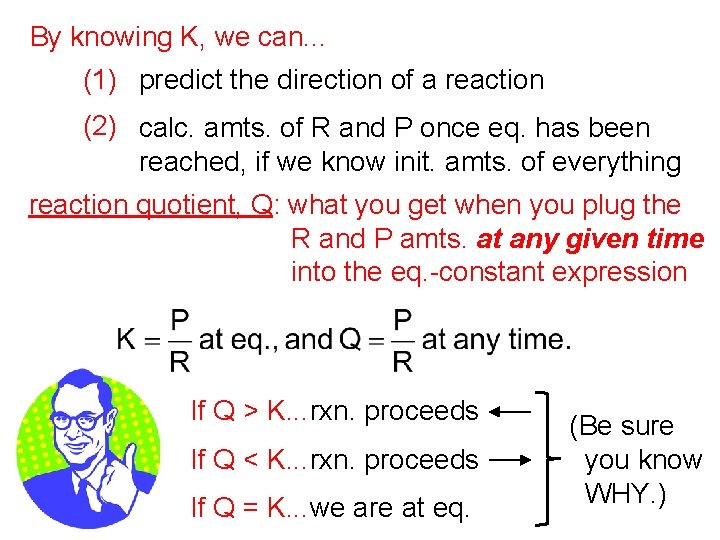
By knowing K, we can. . . (1) predict the direction of a reaction (2) calc. amts. of R and P once eq. has been reached, if we know init. amts. of everything reaction quotient, Q: what you get when you plug the R and P amts. at any given time into the eq. -constant expression If Q > K. . . rxn. proceeds If Q < K. . . rxn. proceeds If Q = K. . . we are at eq. (Be sure you know WHY. )
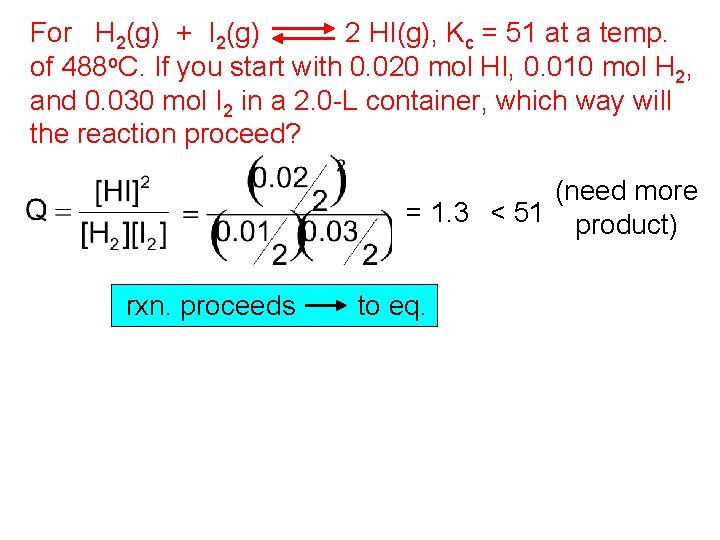
For H 2(g) + I 2(g) 2 HI(g), Kc = 51 at a temp. of 488 o. C. If you start with 0. 020 mol HI, 0. 010 mol H 2, and 0. 030 mol I 2 in a 2. 0 -L container, which way will the reaction proceed? (need more = 1. 3 < 51 product) rxn. proceeds to eq.
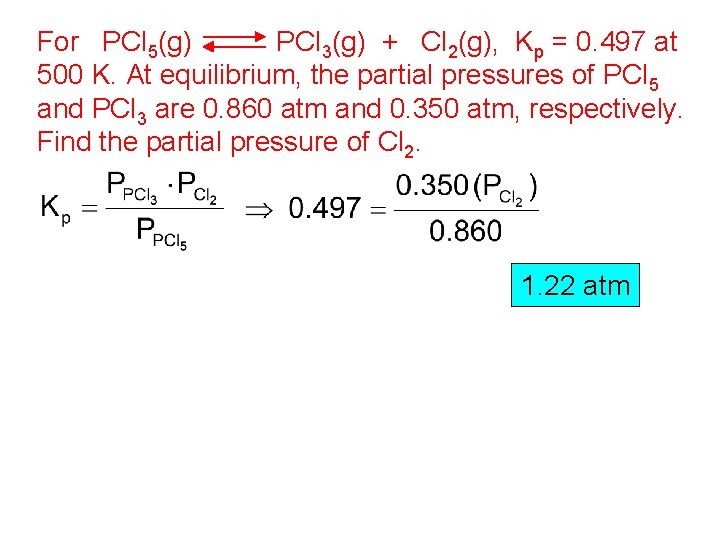
For PCl 5(g) PCl 3(g) + Cl 2(g), Kp = 0. 497 at 500 K. At equilibrium, the partial pressures of PCl 5 and PCl 3 are 0. 860 atm and 0. 350 atm, respectively. Find the partial pressure of Cl 2. 1. 22 atm
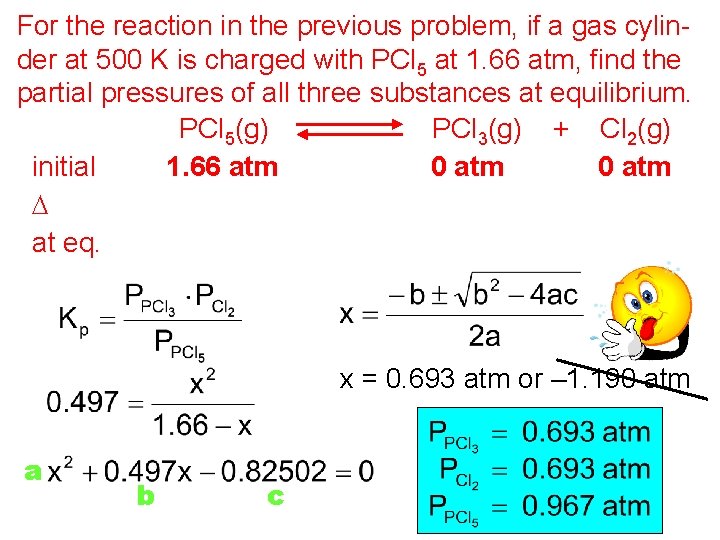
For the reaction in the previous problem, if a gas cylinder at 500 K is charged with PCl 5 at 1. 66 atm, find the partial pressures of all three substances at equilibrium. PCl 5(g) PCl 3(g) + Cl 2(g) initial 1. 66 atm 0 atm D –x x x at eq. 1. 66 – x x = 0. 693 atm or – 1. 190 atm a b c

Le Chatelier’s Principle: “If a system at equilibrium is disturbed by a change in temperature, pressure, or concentration of a component, the system will shift its equilibrium position to counteract the effect of the disturbance. ” Henri Le Chatelier (1850– 1936)
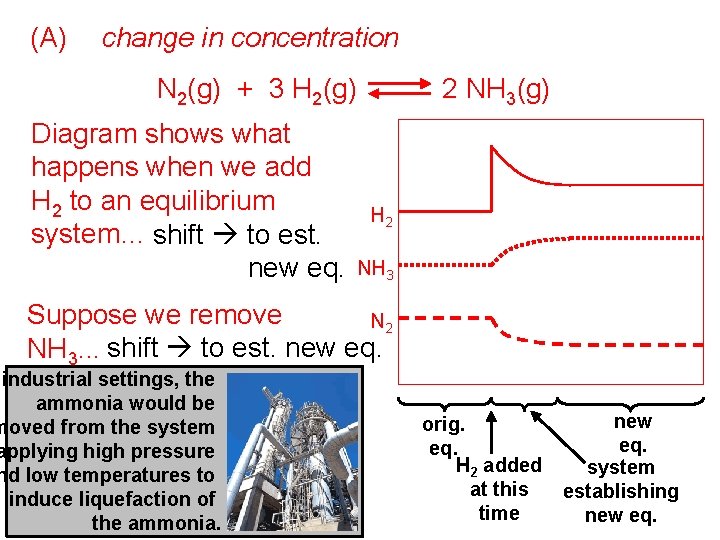
(A) change in concentration N 2(g) + 3 H 2(g) Diagram shows what happens when we add H 2 to an equilibrium system… shift to est. new eq. 2 NH 3(g) H 2 NH 3 Suppose we remove N 2 NH 3. . . shift to est. new eq. industrial settings, the ammonia would be moved from the system applying high pressure nd low temperatures to induce liquefaction of the ammonia. orig. eq. H 2 added at this time new eq. system establishing new eq.

(B) change in pressure – for gaseous equilibrium systems only 2 NO 2(g) N 2 O 4(g) If we increase pressure, the SHIFT system “wants” to. . . get smaller to est. new eq. If we decrease pressure, the system “wants” to. . . get bigger For H 2(g) + I 2(g) 2 HI(g), pressure changes result in. . . NO SHIFT to est. new eq.

More specifically, if the partial pressures are affected, then there will be shifting. Otherwise. . . no. e. g. , 2 SO 2(g) + O 2(g) “Ne!” 2 SO 3(g) e! ” “Ne!” If we add neon into “N “N e! ” “Ne!” reaction vessel. . . Partial pressures of SO 2, and SO 3 are unchanged. NO SHIFT As long as temperature doesn’t change, K is. . . NOT affected by P-V changes.

(C) changes in temperature These always result in. . . shifts in eq. AND changes in K. For exothermic reactions: – P + heat (DH is ____) P + (DH is ____) -- as T increases. . . shift -- as T decreases. . . shift , K … ize or em K! t m IN heat + R TH For endothermic reactions: , K n’ -- as T increases. . . shift -- as T decreases. . . shift Do R
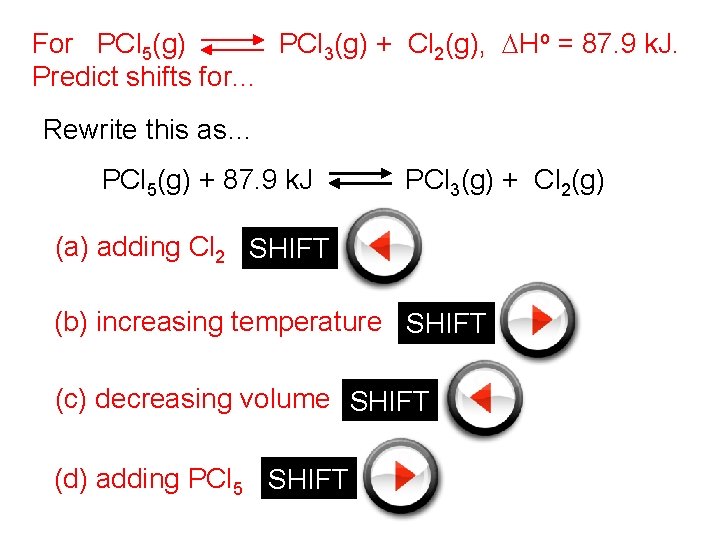
For PCl 5(g) PCl 3(g) + Cl 2(g), DHo = 87. 9 k. J. Predict shifts for. . . Rewrite this as… PCl 5(g) + 87. 9 k. J PCl 3(g) + Cl 2(g) (a) adding Cl 2 SHIFT (b) increasing temperature SHIFT (c) decreasing volume SHIFT (d) adding PCl 5 SHIFT

Using standard molar enthalpies, predict the eq. shifts and changes in K with changes in temperature for 2 POCl 3(g) 2 PCl 3(g) + O 2(g) DHfo (k. J/mol) – 542. 2 – 288. 1 0 Strategy: 1) Use DHfos to estimate DHrxn. 2) Use Le Chatelier to predict shifts. DHrxn = 2(– 288. 1) – 2(– 542. 2) = +508. 2 k. J (endothermic) as T. . . shift , K

Effect of a Catalyst -- the forward and reverse reaction rates are both increased -NO SHIFT -- Catalysts increase the rate at which eq. is achieved, but the final composition of the system is unchanged, as is K. Catalytic converters in car help to quickly convert nitrogen oxides into nitrog and oxygen and carbon monoxide into carbon dioxide.
- Slides: 27