Scanning Probe Investigations of Physisorption and Chemical Reactivity

Scanning Probe Investigations of Physisorption and Chemical Reactivity

Tapping Mode AFM Studies of PAMAM Dendrimers T. Müller, D. Yablon, M. Kleinman, R. Karchner, H. Fang, and G. Flynn & collaborators: S. Jockusch and N. Turro, K. Rahman, and C. Durning
![Range over which tunneling probability is non-zero is 10 Å [Resolution 0. 5 Å] Range over which tunneling probability is non-zero is 10 Å [Resolution 0. 5 Å]](http://slidetodoc.com/presentation_image_h/77e955c71e402150d5f58d6bec959921/image-3.jpg)
Range over which tunneling probability is non-zero is 10 Å [Resolution 0. 5 Å]
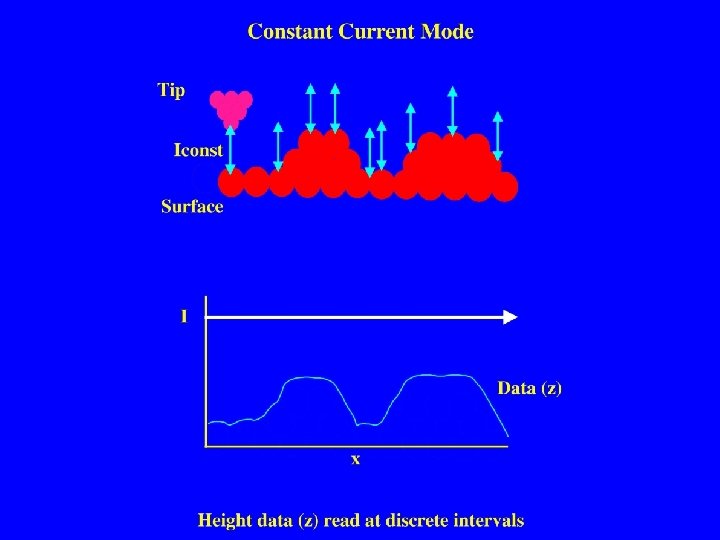
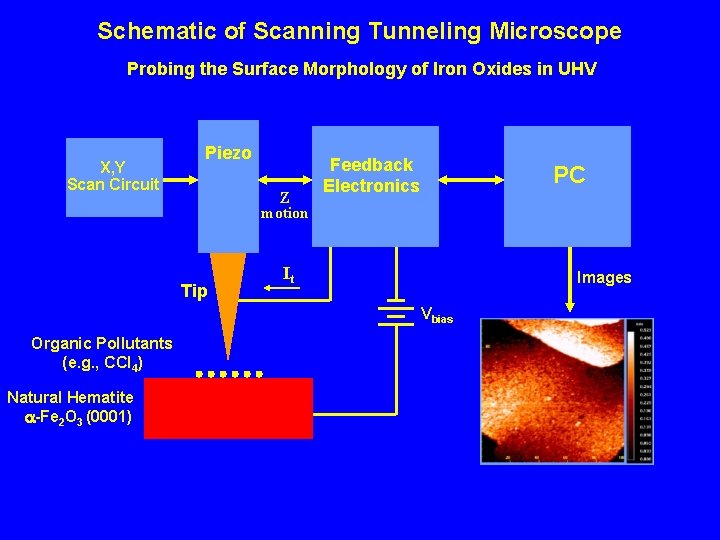
Schematic of Scanning Tunneling Microscope Probing the Surface Morphology of Iron Oxides in UHV X, Y Scan Circuit Piezo Z motion Tip Feedback Electronics PC It Images Vbias Organic Pollutants (e. g. , CCl 4) Natural Hematite -Fe 2 O 3 (0001)
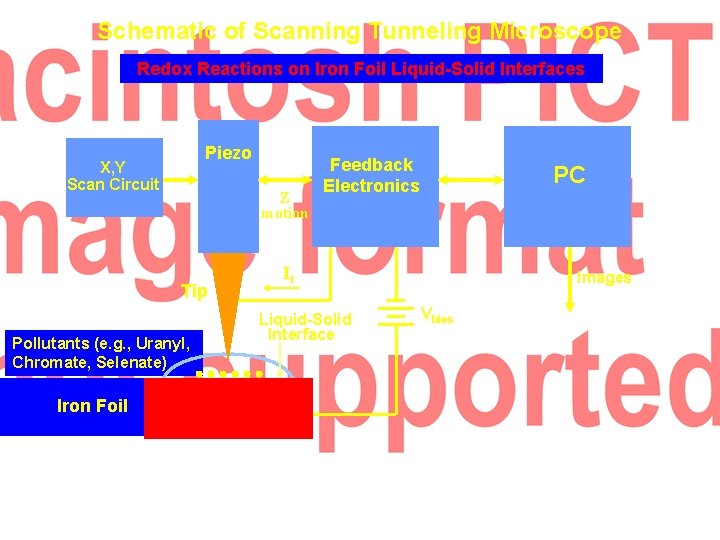
Schematic of Scanning Tunneling Microscope Redox Reactions on Iron Foil Liquid-Solid Interfaces Piezo X, Y Scan Circuit Z motion Tip Pollutants (e. g. , Uranyl, Chromate, Selenate) Iron Foil Feedback Electronics PC It Liquid-Solid Interface Images Vbias


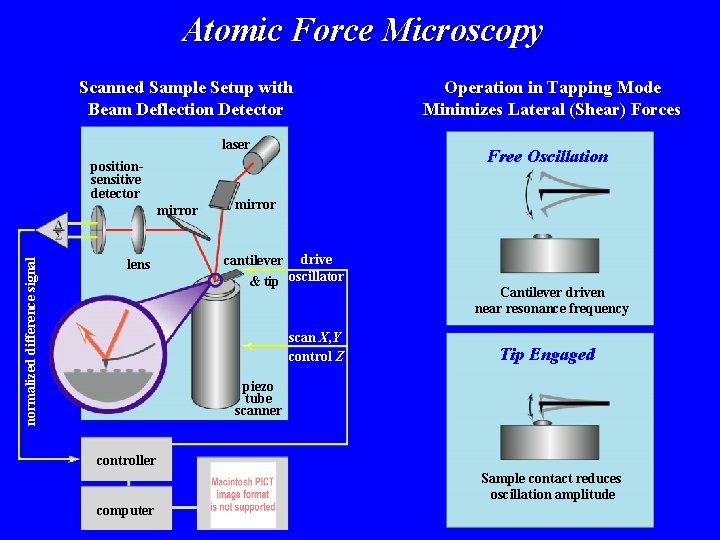
Atomic Force Microscopy Scanned Sample Setup with Beam Deflection Detector laser positionsensitive detector normalized difference signal mirror lens Operation in Tapping Mode Minimizes Lateral (Shear) Forces Free Oscillation mirror cantilever drive & tip oscillator scan X, Y control Z Cantilever driven near resonance frequency Tip Engaged piezo tube scanner controller Sample contact reduces oscillation amplitude computer
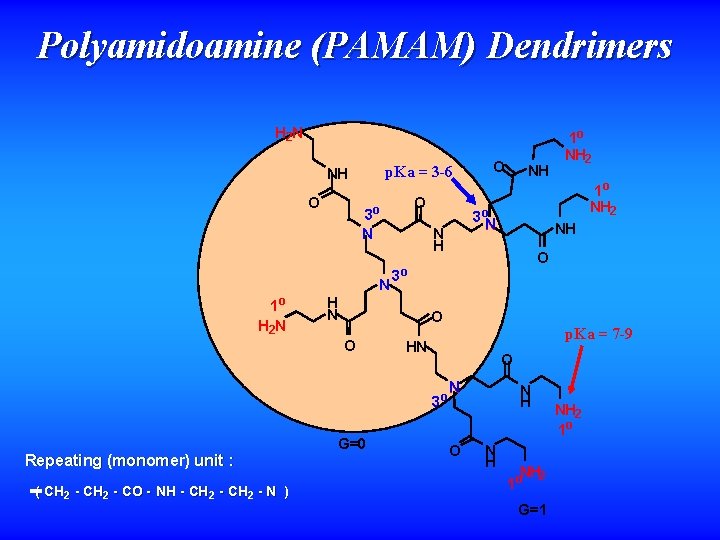
Polyamidoamine (PAMAM) Dendrimers H 2 N p. Ka = 3 -6 NH O N 1 o H 2 N O 3 o N O O G=0 1 o NH 2 NH 3 o p. Ka = 7 -9 HN O 3 o ( CH 2 - C O - NH - CH 2 - N ) NH 3 o. N N H H N O Repeating (monomer) unit : O 1 o NH 2 N O N H NH 2 1 o G=1 NH 2 1 o
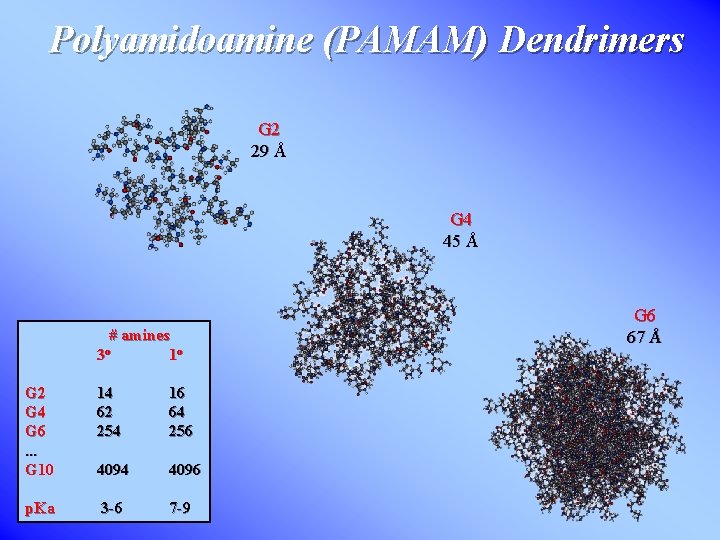
Polyamidoamine (PAMAM) Dendrimers G 2 29 Å G 4 45 Å # amines 1 o 3 o G 2 G 4 G 6. . . G 10 14 62 254 16 64 256 4094 4096 p. Ka 3 -6 7 -9 G 6 67 Å

Structure & Applications high density of functional groups branched structure, spherical shape for gen. ≥ 5 empty “container” space (micelle mimic) size: diameter ~ 10 nm for G 9 G 7 PAMAM Dendrimer many transport applications, catalysis / reaction vessels, molecular antennae Ordered Dendrimer Film Self-assembly at interface useful for chemical sensing devices modifies size & shape of dendrimers

Previous Studies • • Focus: dried adsorbate on hydrophilic surfaces Amine-terminated dendrimers readily adsorb Observed single dendrimers and smooth films Compression along surface normal & lateral spreading (G 5: d = 15 nm, h = 1 nm / G 10: d = 25 nm, h = 5 nm) • Evolution of conditions during drying process ? • Influence of residual water (& capillary forces) ? • Influence of charge interactions between dendrimer (+) and surface (-) ?

Dried Films on Hydrophobic Surfaces

PAMAM Dendrimers on dry HOPG G 9, 10 mg/ml, p. H~8 G 9, 1 mg/ml, p. H~7 • Adsorption onto (hydrophobic) HOPG from dilute (0. 001% w/w) solution • Few surface contact-minimizing aggregates • AFM-induced lateral motion avoidable
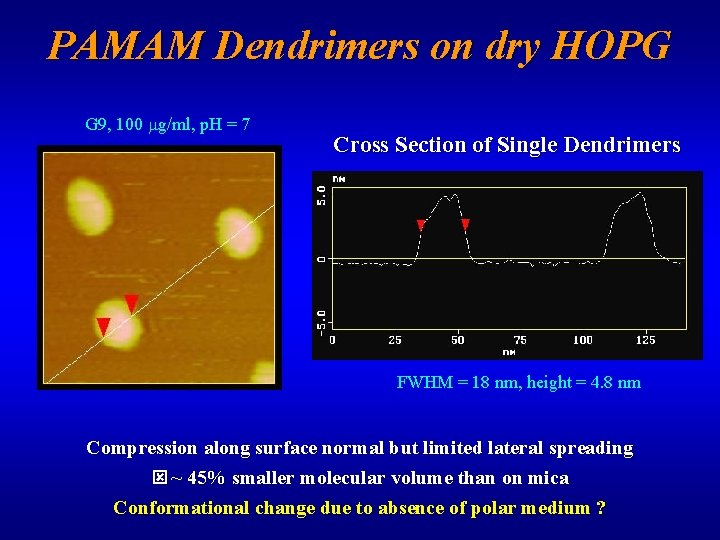
PAMAM Dendrimers on dry HOPG G 9, 100 mg/ml, p. H = 7 Cross Section of Single Dendrimers FWHM = 18 nm, height = 4. 8 nm Compression along surface normal but limited lateral spreading ~ 45% smaller molecular volume than on mica Conformational change due to absence of polar medium ?

In Situ Studies of Self-Assembly at the Liquid-Solid Interface

In Situ AFM Studies of PAMAM Dendrimers at the Liquid-Solid Interface Self-Assembly in the Presence of the Supernatant drying Air ? ? Solid

In Situ AFM Studies of PAMAM Dendrimers at the Liquid-Solid Interface Self-Assembly in the Presence of the Supernatant G 9, 1 mg/ml, p. H~2, 8 mm • Formation of Oblate Aggregates G 9, p. H ~ 7: d ~ 200 nm, d/h ~ 10 • EPR & fluorescence probes find no evidence of aggregation in solution [Turro Group] Aggregates form and reside exclusively at interface
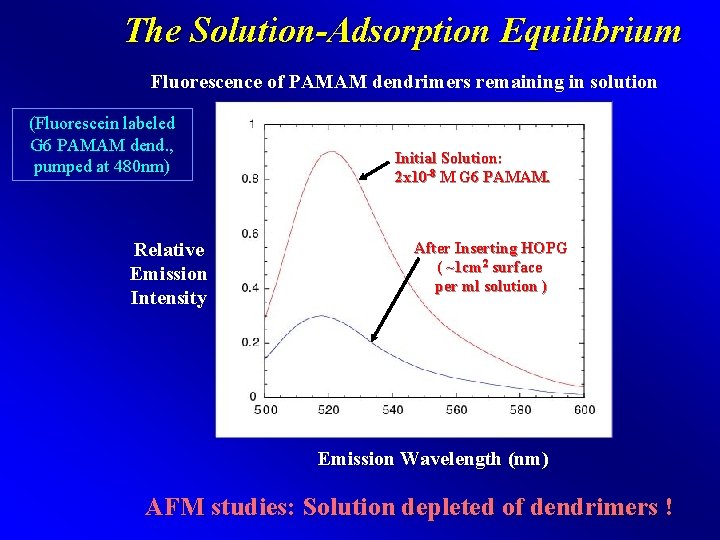
The Solution-Adsorption Equilibrium Fluorescence of PAMAM dendrimers remaining in solution (Fluorescein labeled G 6 PAMAM dend. , pumped at 480 nm) Relative Emission Intensity Initial Solution: 2 x 10 -8 M G 6 PAMAM. After Inserting HOPG ( ~1 cm 2 surface per ml solution ) Emission Wavelength (nm) AFM studies: Solution depleted of dendrimers !
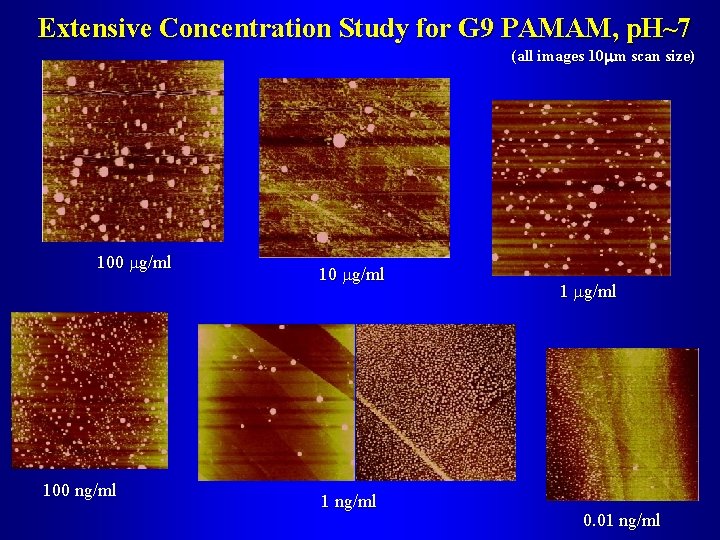
Extensive Concentration Study for G 9 PAMAM, p. H~7 (all images 10 mm scan size) 100 mg/ml 100 ng/ml 10 mg/ml 1 ng/ml 1 mg/ml 0. 01 ng/ml
![Concentration Study for G 9 Parameterization of Aggregate Size Distribution Aggregate FWHM [nm] Concentration Concentration Study for G 9 Parameterization of Aggregate Size Distribution Aggregate FWHM [nm] Concentration](http://slidetodoc.com/presentation_image_h/77e955c71e402150d5f58d6bec959921/image-22.jpg)
Concentration Study for G 9 Parameterization of Aggregate Size Distribution Aggregate FWHM [nm] Concentration in Supernatant [mg/ml]

p. H-Dependence of Dendrimer Aggregation on HOPG G 9 PAMAM, 1 mg/ml, 10 mm scan size p. H = 2. 2 p. H = 10. 7

p. H-Dependence of Dendrimer Aggregation on Mica G 9 PAMAM, 1 mg/ml, 5 mm scan size p. H = 3. 1 p. H = 6. 2

Acidification favors increased aggregation Protonation of G 9 PAMAM dendrimers: • 2048 outer (primary) amines with p. Ka ≈ 7 -9 • 2046 inner (tertiary) amines with p. Ka ≈ 3 -6 all within ~ 5 nm radius dramatic changes of charge & H-bonding with p. H 3 p. H 6 p. H 9 … control ionic strength of supernatant ?

Self-Assembly and Ionic Strength G 9 PAMAM on HOPG, 10 mg/ml, 4. 5 mm scan size 0. 001 M Na 2 HPO 4 0. 1 M Na 2 HPO 4
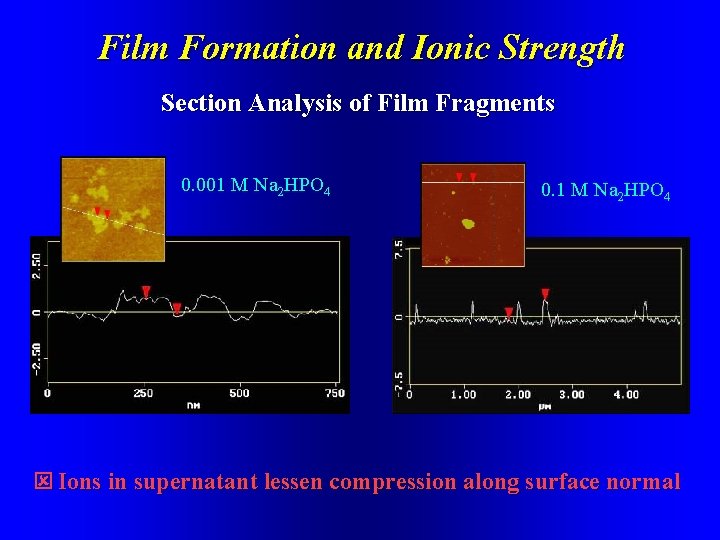
Film Formation and Ionic Strength Section Analysis of Film Fragments 0. 001 M Na 2 HPO 4 0. 1 M Na 2 HPO 4 Ions in supernatant lessen compression along surface normal

Substrate Dependence of Self-Assembly G 5 PAMAM Dendrimers on Si and HOPG p. H = 5. 4 p. H = 9. 2 HOPG 10 mm 5 mm Si Less aggregation on hydrophilic substrates (? )

Summary & Conclusions • • Dendrimers exhibit rich behavior at surfaces & interfaces Adsorption to hydrophobic surfaces despite strong interaction with water Significant compression along surface normal upon physisorption Investigated self-assembly in solution: Near-universal formation of large, oblate aggregates Aggregates form & reside exclusively at interface Weak dependence on solution parameters • Formation of dried films: Drying process breaks up aggregates (isolated dendrimers / film) Important role of residual water (flattening & expansion) • Future Directions: Imaging in nonpolar solvents (e. g. , phenyloctane) Submolecular resolution (low-current STM) Dendrimers with enclosed guest molecules (Fe. Ox nanoparticles ? )
- Slides: 29