Chemistry and Chemical Reactivity 6 th Edition 1


































- Slides: 88

Chemistry and Chemical Reactivity 6 th Edition 1 John C. Kotz Paul M. Treichel Gabriela C. Weaver CHAPTER 9 Bonding and Molecular Structure: Fundamental Concepts Lectures written by John Kotz © 2006 Brooks/Cole Thomson © 2006 Brooks/Cole - Thomson

CHEMICAL BONDING Cocaine © 2006 Brooks/Cole - Thomson 2

Chemical Bonding Problems and questions — How is a molecule or polyatomic ion held together? Why are atoms distributed at strange angles? Why are molecules not flat? Can we predict the structure? How is structure related to chemical and physical properties? © 2006 Brooks/Cole - Thomson 3

Forms of Chemical Bonds • There are 2 extreme forms of connecting or bonding atoms: • Ionic—complete transfer of 1 or more electrons from one atom to another • Covalent—some valence electrons shared between atoms • Most bonds are somewhere in between. © 2006 Brooks/Cole - Thomson 4

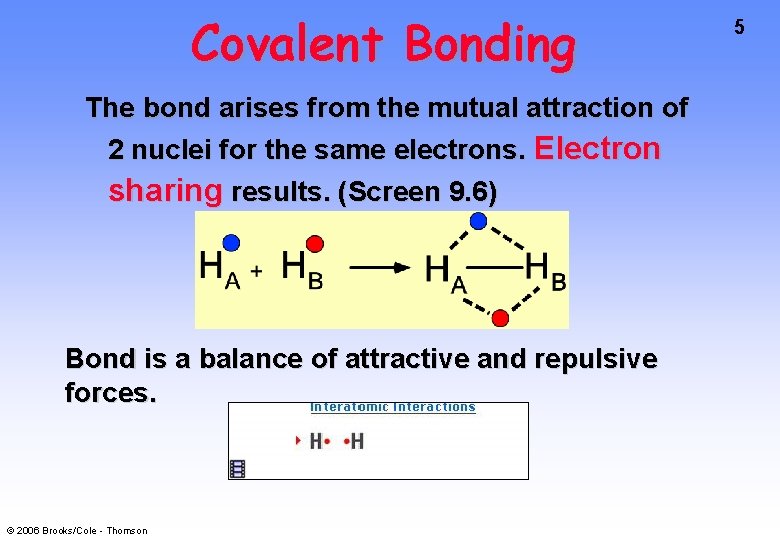
Covalent Bonding The bond arises from the mutual attraction of 2 nuclei for the same electrons. Electron sharing results. (Screen 9. 6) Bond is a balance of attractive and repulsive forces. © 2006 Brooks/Cole - Thomson 5

Chemical Bonding: Objectives are to understand: 1. valence e- distribution in molecules and ions. 2. molecular structures 3. bond properties and their effect on molecular properties. © 2006 Brooks/Cole - Thomson 6

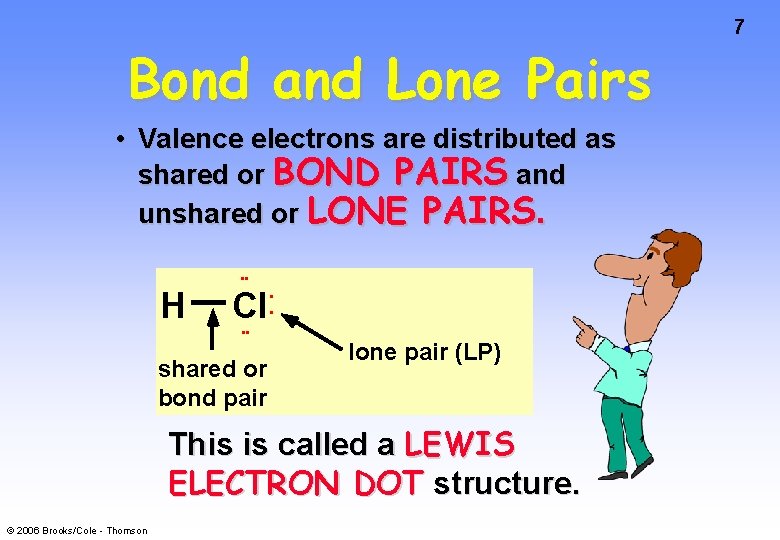
Bond and Lone Pairs • Valence electrons are distributed as shared or BOND PAIRS and unshared or LONE PAIRS. • • H Cl • • shared or bond pair • • lone pair (LP) This is called a LEWIS ELECTRON DOT structure. © 2006 Brooks/Cole - Thomson 7

Valence Electrons are divided between core and valence electrons B atomic #5 has a total of 5 electrons. Two are in the Core, [He] ; the remaining 3 are valence = 2 s 2 2 p 1 These valence electrons are available for bonding; we show them as “dots. ” Br [Ar] 3 d 10 4 s 2 4 p 5 Core = [Ar] 3 d 10 , valence = 4 s 2 4 p 5 © 2006 Brooks/Cole - Thomson 8

Rules of the Game No. of valence electrons of a main group atom = Group number I II IV V VI VIII 1 2 3 5 6 7 8 © 2006 Brooks/Cole - Thomson 4 9

10 How many valence electrons are in … • H 2 2(1)= 2 valence electrons PCl 3 5+3(7)= 26 valence elctrons CH 2 Cl 2 2(4)+2(1)+2(7)= 24 valence electrons © 2006 Brooks/Cole - Thomson

Lewis Structures • Learn to find total number of valence electrons when given a chemical formula. • Find which group each element belongs to. • Group number = number of valence electrons. • Include any charge in the total electron count Each element in a Lewis structure has at least 8 electrons around it. Except H, which has only 2. This observation is called the OCTET RULE © 2006 Brooks/Cole - Thomson 11

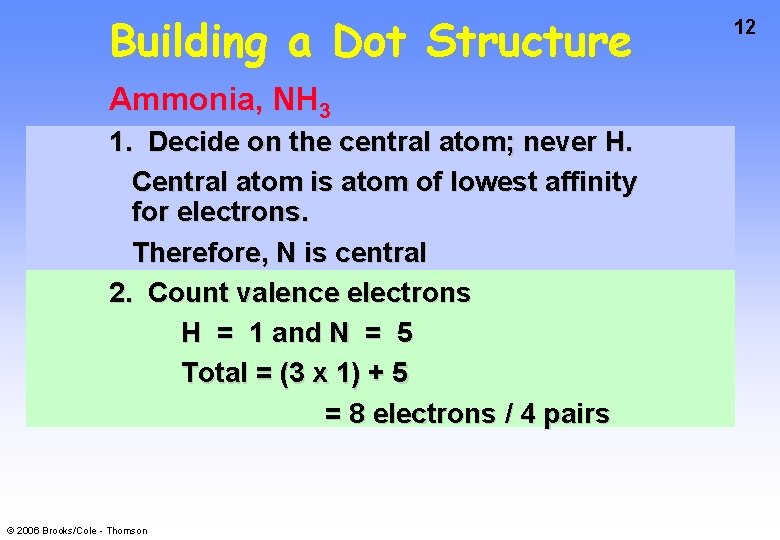
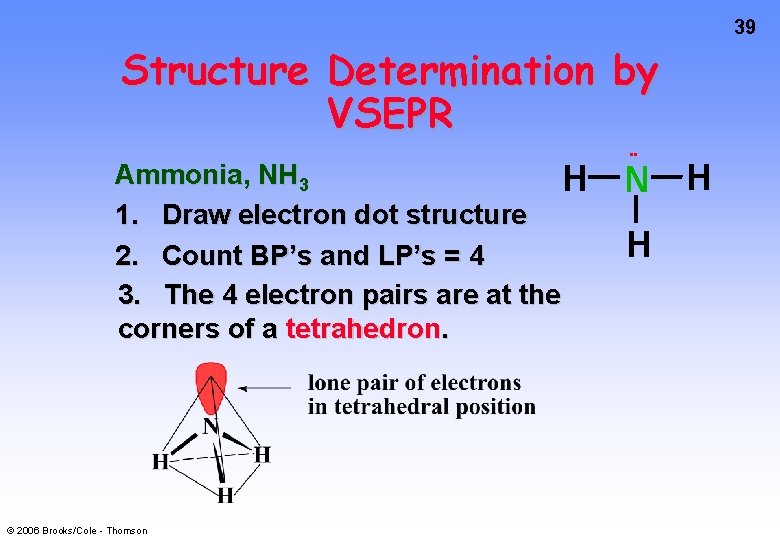
Building a Dot Structure Ammonia, NH 3 1. Decide on the central atom; never H. Central atom is atom of lowest affinity for electrons. Therefore, N is central 2. Count valence electrons H = 1 and N = 5 Total = (3 x 1) + 5 = 8 electrons / 4 pairs © 2006 Brooks/Cole - Thomson 12

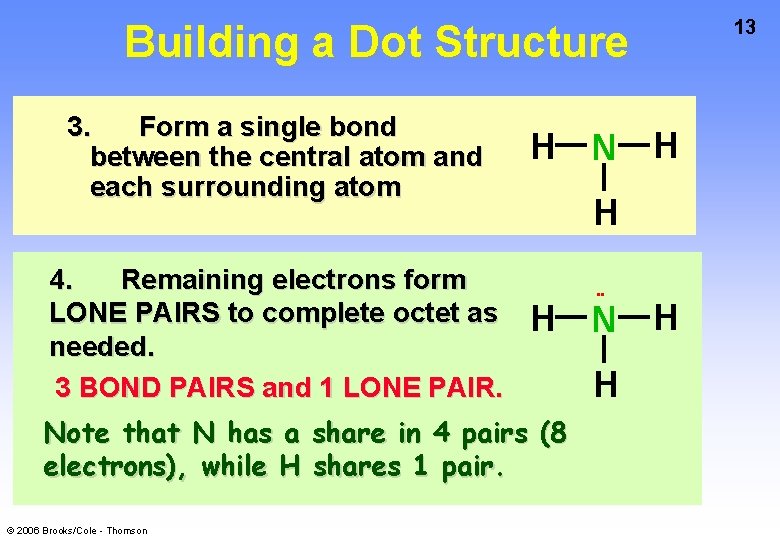
Building a Dot Structure 3. Form a single bond between the central atom and each surrounding atom 4. Remaining electrons form LONE PAIRS to complete octet as needed. 3 BOND PAIRS and 1 LONE PAIR. H N H H H Note that N has a share in 4 pairs (8 electrons), while H shares 1 pair. © 2006 Brooks/Cole - Thomson • • H 13

Sulfite ion, SO 32 How many valence electrons? 6+3(6) +2 = 26 Draw the skeleton with S in the middle. Draw in all single bonds as lines 10 pairs of electrons are now left. © 2006 Brooks/Cole - Thomson 14

Sulfite ion, SO 32 Step 1. Central atom = S Step 2. Count valence electrons S= 6 3 x O = 3 x 6 = 18 Negative charge = 2 TOTAL = 26 e- or 13 pairs Step 3. Form bonds 10 pairs of electrons are now left. © 2006 Brooks/Cole - Thomson 15

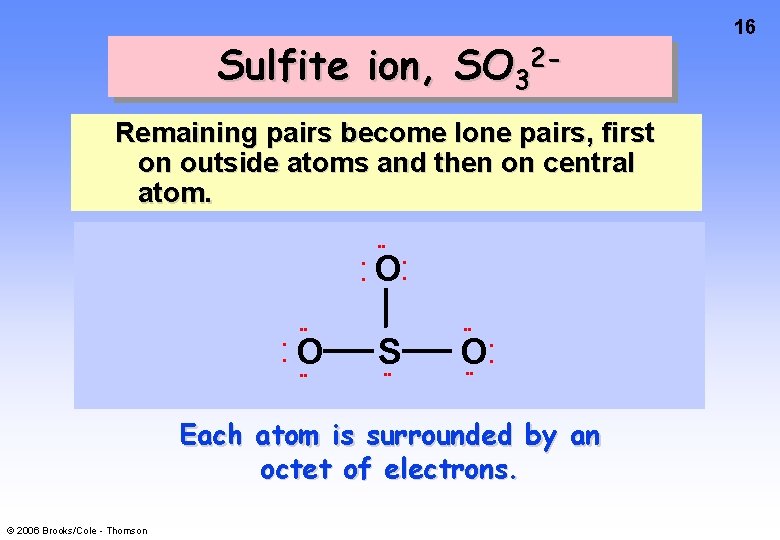
Sulfite ion, SO 32 Remaining pairs become lone pairs, first on outside atoms and then on central atom. • • • • O • • O S • • O • • • • Each atom is surrounded by an octet of electrons. © 2006 Brooks/Cole - Thomson 16

17 © 2006 Brooks/Cole - Thomson

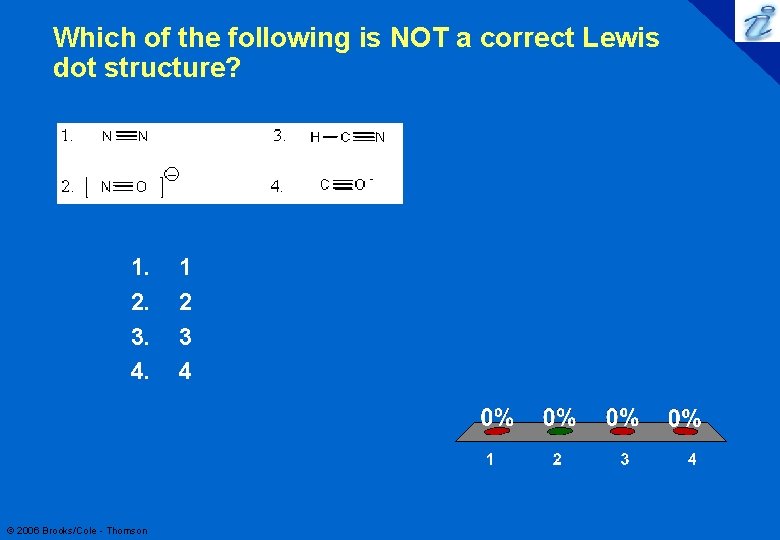
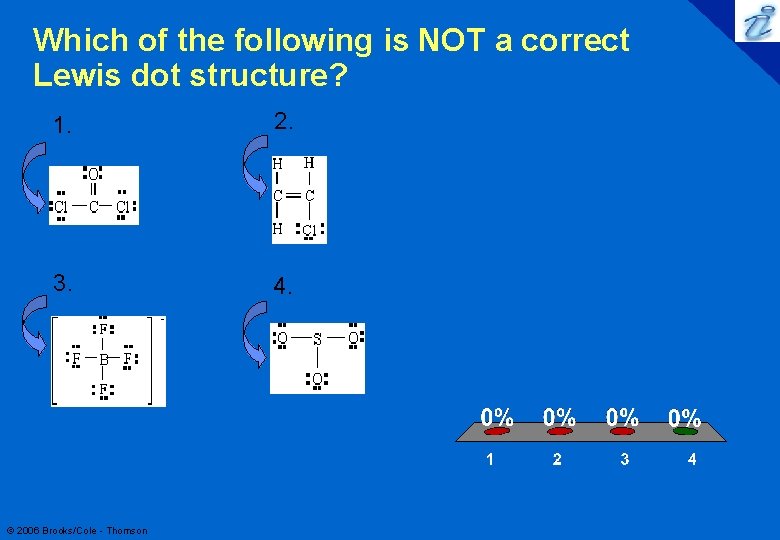
Which of the following is NOT a correct Lewis dot structure? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson 1 2 3 4 18

Which of the following is NOT a correct Lewis dot structure? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson 19

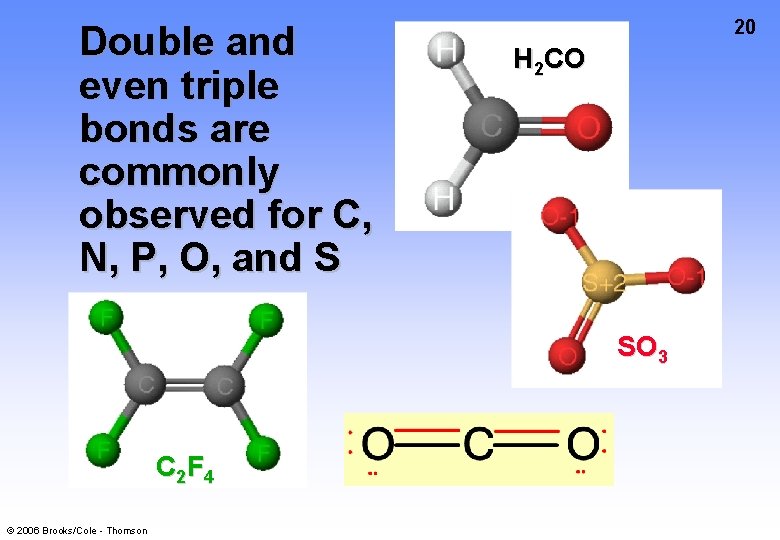
Double and even triple bonds are commonly observed for C, N, P, O, and S 20 H 2 CO SO 3 C 2 F 4 © 2006 Brooks/Cole - Thomson

21 © 2006 Brooks/Cole - Thomson

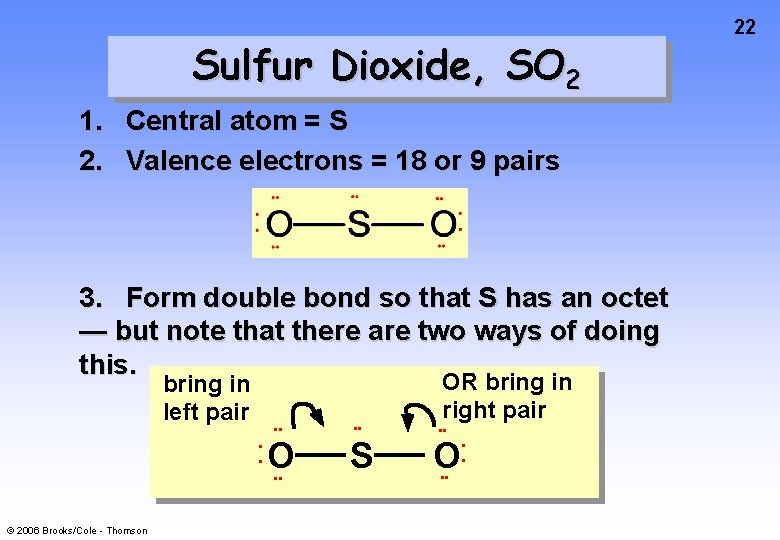
Sulfur Dioxide, SO 2 1. Central atom = S 2. Valence electrons = 18 or 9 pairs 3. Form double bond so that S has an octet — but note that there are two ways of doing this. bring in left pair • • • • O • • © 2006 Brooks/Cole - Thomson • • S OR bring in right pair • • O • • • • 22

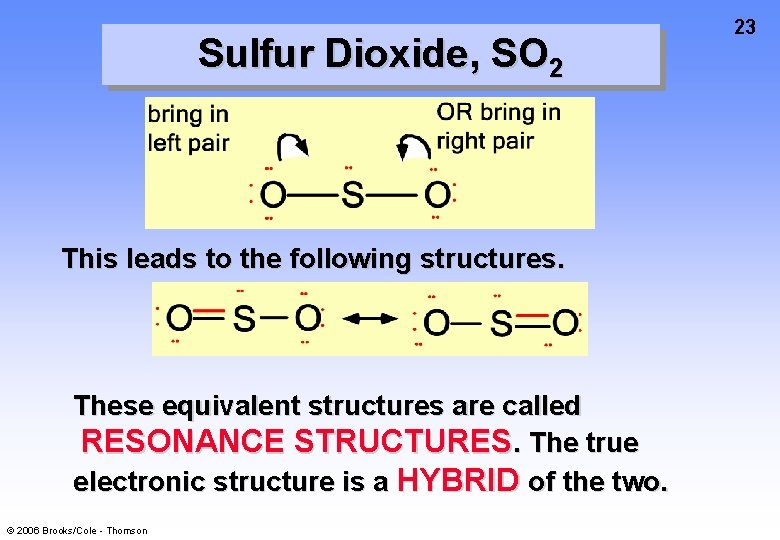
Sulfur Dioxide, SO 2 This leads to the following structures. These equivalent structures are called RESONANCE STRUCTURES. The true electronic structure is a HYBRID of the two. © 2006 Brooks/Cole - Thomson 23

24 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson

25 © 2006 Brooks/Cole - Thomson

Formal Atom Charges • Atoms in molecules often bear a charge (+ or -). • The predominant resonance structure of a molecule is the one with charges as close to 0 as possible. • Formal charge = Group number – (no. of bonds) - (no. of LP electrons) © 2006 Brooks/Cole - Thomson 26

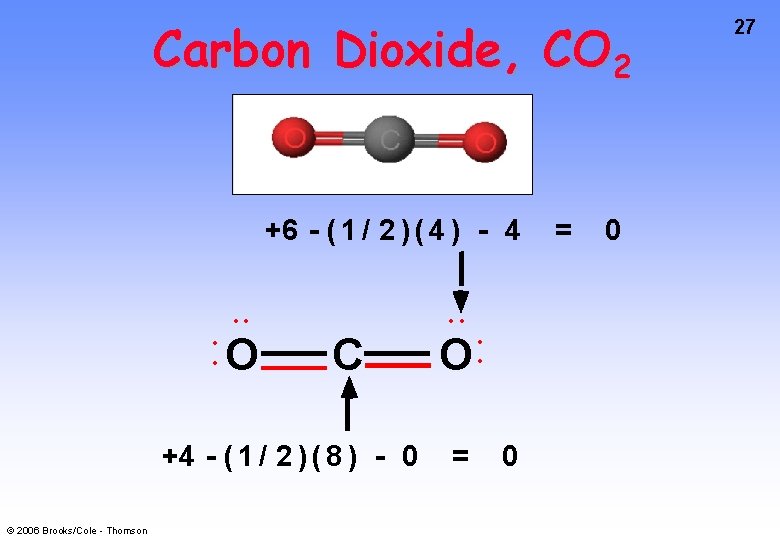
Carbon Dioxide, CO 2 +6 - ( 1 / 2 ) ( 4 ) - 4 • • • • O • • C +4 - ( 1 / 2 ) ( 8 ) - 0 © 2006 Brooks/Cole - Thomson O = • • 0 = 0 27

Formal charges on the formate ion: 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson 0, 0, 0 +1, -1 +1, 0, -1 0, 0, -1 29

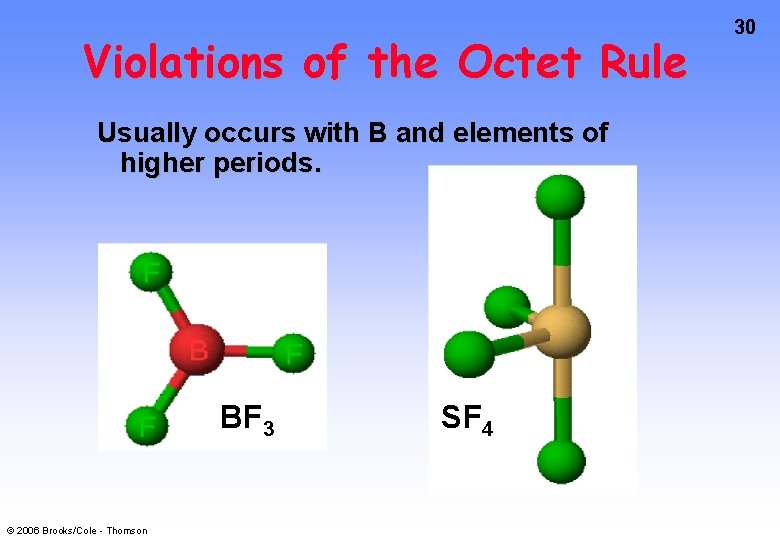
Violations of the Octet Rule Usually occurs with B and elements of higher periods. BF 3 © 2006 Brooks/Cole - Thomson SF 4 30

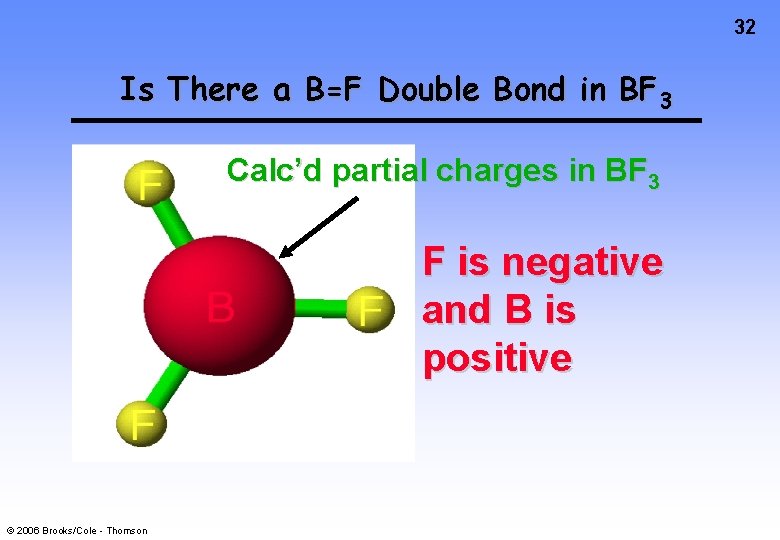
31 Boron Trifluoride, BF 3 F • • • F • • • • -1 B • • F +1 • • • • What if we form a B—F double bond to satisfy the B atom octet? © 2006 Brooks/Cole - Thomson

32 Is There a B=F Double Bond in BF 3 Calc’d partial charges in BF 3 F is negative and B is positive © 2006 Brooks/Cole - Thomson

33 Sulfur Tetrafluoride, SF 4 • Central atom = • Valence electrons = ___ or ___ pairs. • Form sigma bonds and distribute electron pairs. 5 pairs around the S atom. A common occurrence outside the 2 nd period. © 2006 Brooks/Cole - Thomson

MOLECULAR GEOMETRY

MOLECULAR GEOMETRY VSEPR • Valence Shell Electron Pair Repulsion theory. • Most important factor in determining geometry is relative repulsion between electron pairs. Chemistry NOW © 2006 Brooks/Cole - Thomson Molecule adopts the shape that minimizes the electron pair repulsions. 35

36 Rules to determine electronic structure 1. Look at Lewis Structure 2. Count electron domains a. Bonded atoms b. Lone pairs of electrons 3. Add # bonded atoms + # lp 4. Identify shape from the total # e- domains. © 2006 Brooks/Cole - Thomson

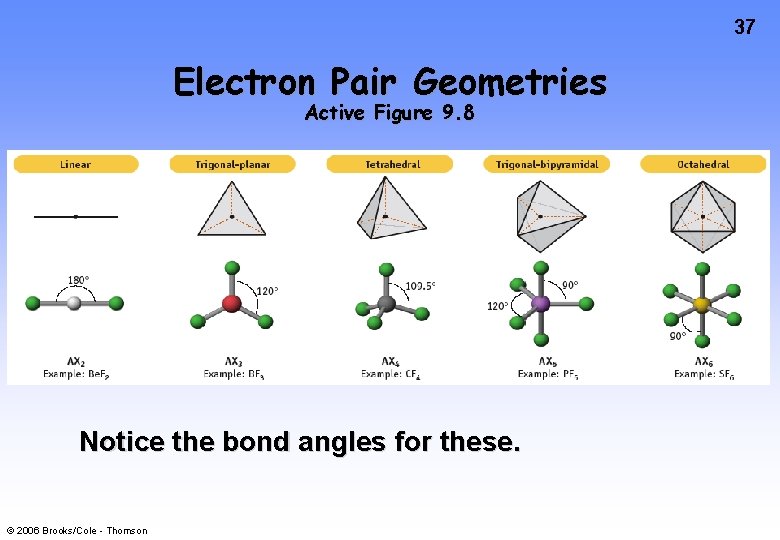
37 Electron Pair Geometries Active Figure 9. 8 Notice the bond angles for these. © 2006 Brooks/Cole - Thomson

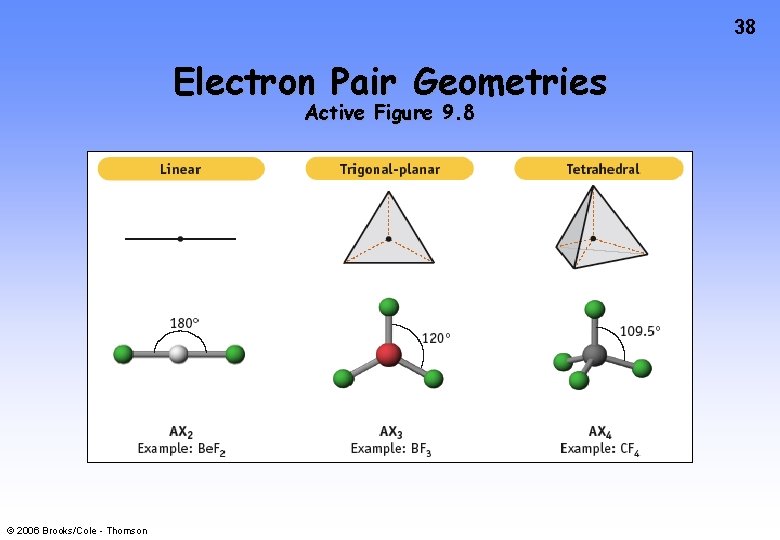
38 Electron Pair Geometries Active Figure 9. 8 © 2006 Brooks/Cole - Thomson

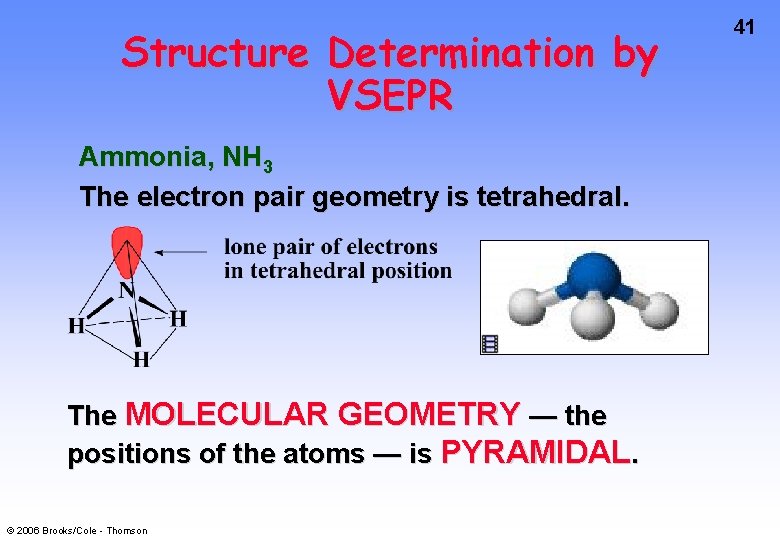
39 Structure Determination by VSEPR Ammonia, NH 3 H 1. Draw electron dot structure 2. Count BP’s and LP’s = 4 3. The 4 electron pairs are at the corners of a tetrahedron. © 2006 Brooks/Cole - Thomson • • N H H

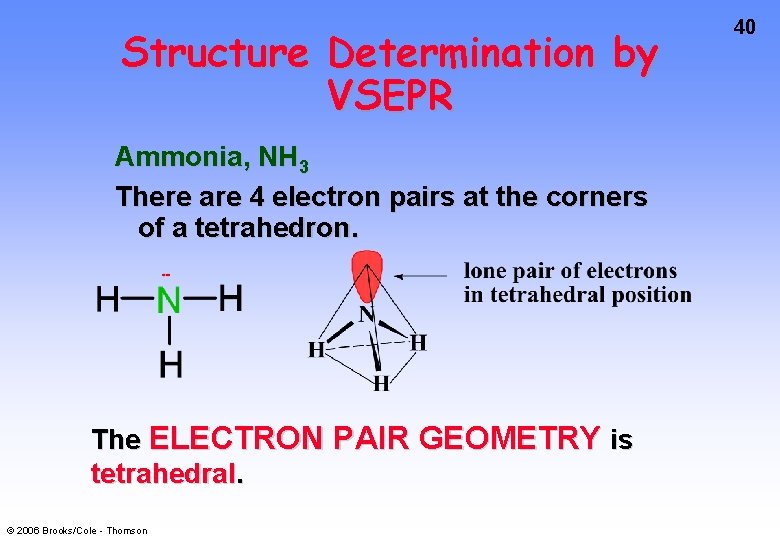
Structure Determination by VSEPR Ammonia, NH 3 There are 4 electron pairs at the corners of a tetrahedron. The ELECTRON PAIR GEOMETRY is tetrahedral. © 2006 Brooks/Cole - Thomson 40

Structure Determination by VSEPR Ammonia, NH 3 The electron pair geometry is tetrahedral. The MOLECULAR GEOMETRY — the positions of the atoms — is PYRAMIDAL. © 2006 Brooks/Cole - Thomson 41

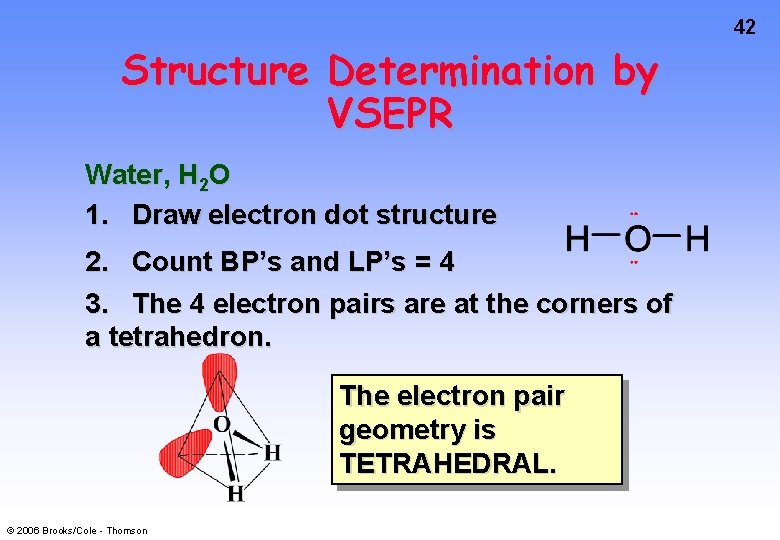
42 Structure Determination by VSEPR Water, H 2 O 1. Draw electron dot structure 2. Count BP’s and LP’s = 4 3. The 4 electron pairs are at the corners of a tetrahedron. The electron pair geometry is TETRAHEDRAL. © 2006 Brooks/Cole - Thomson

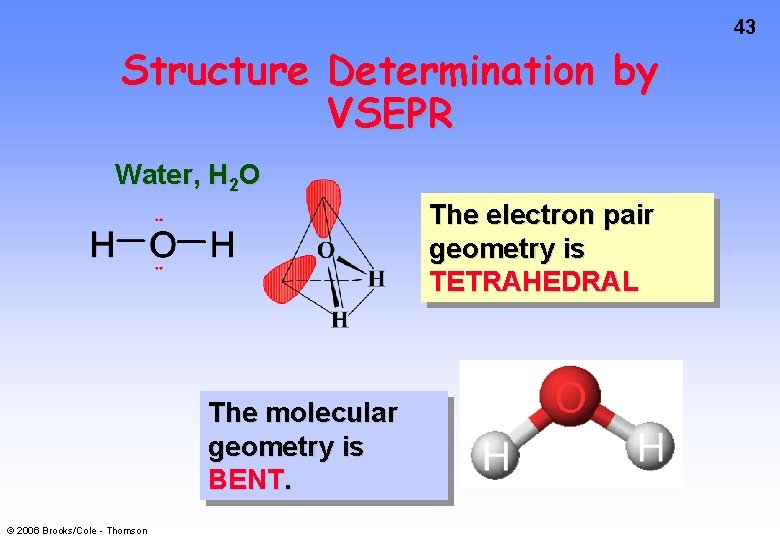
43 Structure Determination by VSEPR Water, H 2 O The electron pair geometry is TETRAHEDRAL The molecular geometry is BENT. © 2006 Brooks/Cole - Thomson

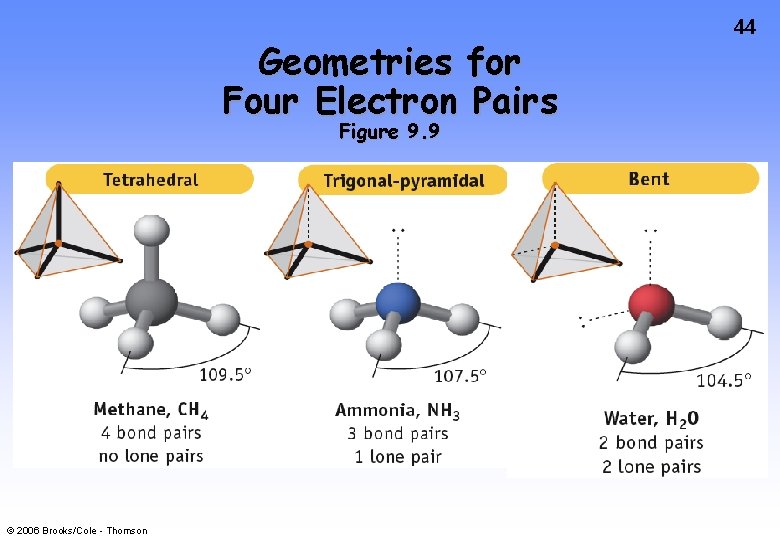
Geometries for Four Electron Pairs Figure 9. 9 © 2006 Brooks/Cole - Thomson 44

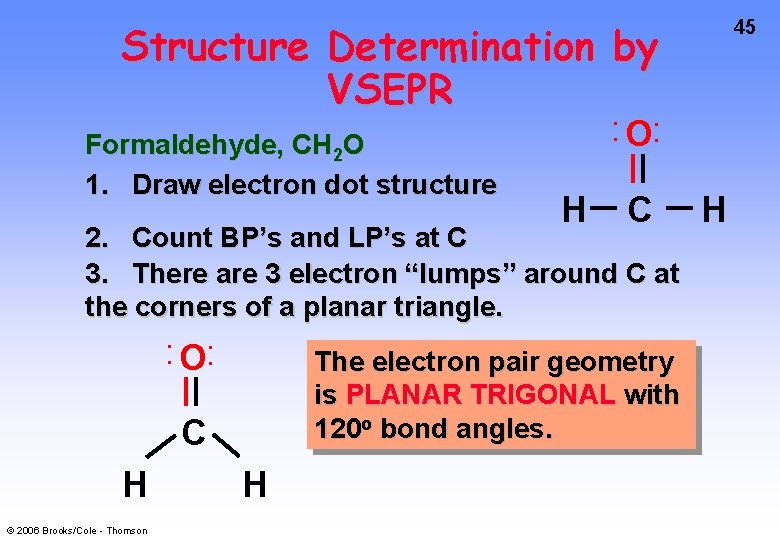
Structure Determination by VSEPR Formaldehyde, CH 2 O 1. Draw electron dot structure • • H O • • C 2. Count BP’s and LP’s at C 3. There are 3 electron “lumps” around C at the corners of a planar triangle. • • O • • The electron pair geometry is PLANAR TRIGONAL with 120 o bond angles. C H © 2006 Brooks/Cole - Thomson H 45 H

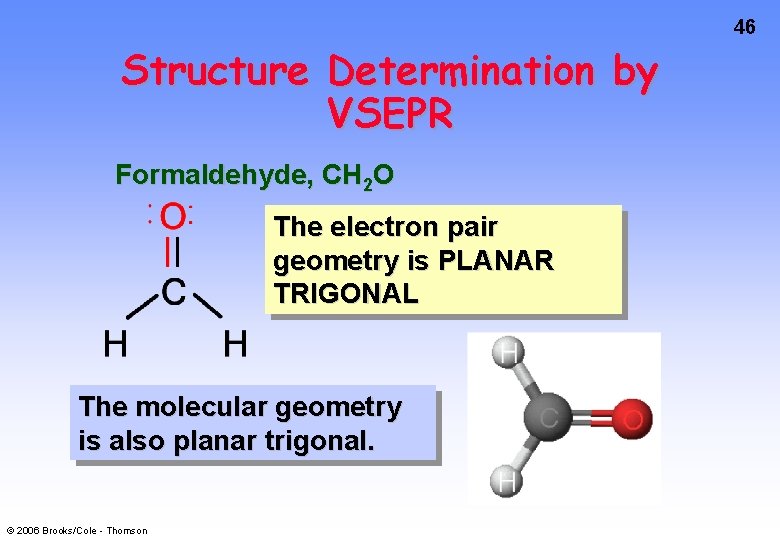
46 Structure Determination by VSEPR Formaldehyde, CH 2 O The electron pair geometry is PLANAR TRIGONAL The molecular geometry is also planar trigonal. © 2006 Brooks/Cole - Thomson

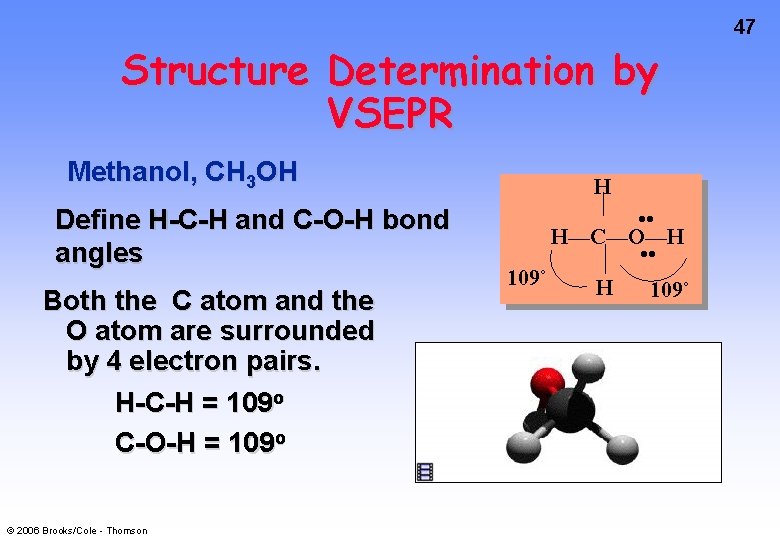
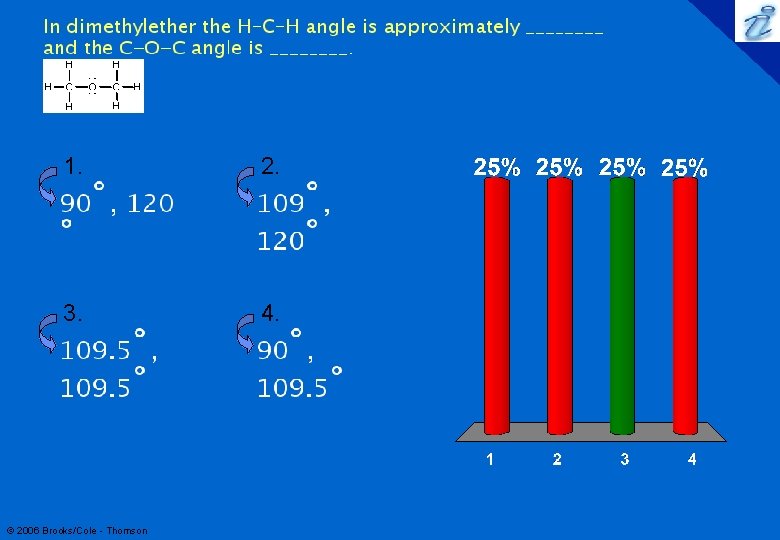
47 Structure Determination by VSEPR Methanol, CH 3 OH Define H-C-H and C-O-H bond angles Both the C atom and the O atom are surrounded by 4 electron pairs. H-C-H = 109 o C-O-H = 109 o © 2006 Brooks/Cole - Thomson H 109˚ • • H—C—O—H • • H 109˚

48 Structure Determination by VSEPR H Acetonitrile, CH 3 CN H—C—C 109˚ H 180˚ One C is surrounded by 4 electron “lumps” and the other by 2 “lumps” © 2006 Brooks/Cole - Thomson N • • Define unique bond angles H-C-H = 109 o C-C-N = 180 o

49 Fast Method to determine molecular structure!! 1. Count total valence electrons. SO 2 =18 2. Multiply the number of bonded atoms times 8. (H is multiplied by 2. ) 2 x 8 = 16 3. Subtract these two numbers to get the number of lone pairs. 18 -16 = 2 = 1 lp 4. Add #bonded atoms + # lp to get electronic geometry. 2 bonded + 1 lp =3= trigonal planal; 120º angles © 2006 Brooks/Cole - Thomson

50 Find electronic geometry for: (IF 4)Total valence electrons = 5(7)+1 =36 # lp = 36 - 4(8)= 4 = 2 lp Electonic geometry and bond angles = 4+2=6; octahedral, 90º © 2006 Brooks/Cole - Thomson

51 Find electronic geometry for: (Br. O 4)Total valence electrons = 7+4(6)+1 =32 # lp = 32 - 4(8)= 0 lp Electonic geometry and bond angles = 4+0=4; tetrahedral, 109º © 2006 Brooks/Cole - Thomson

52 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson

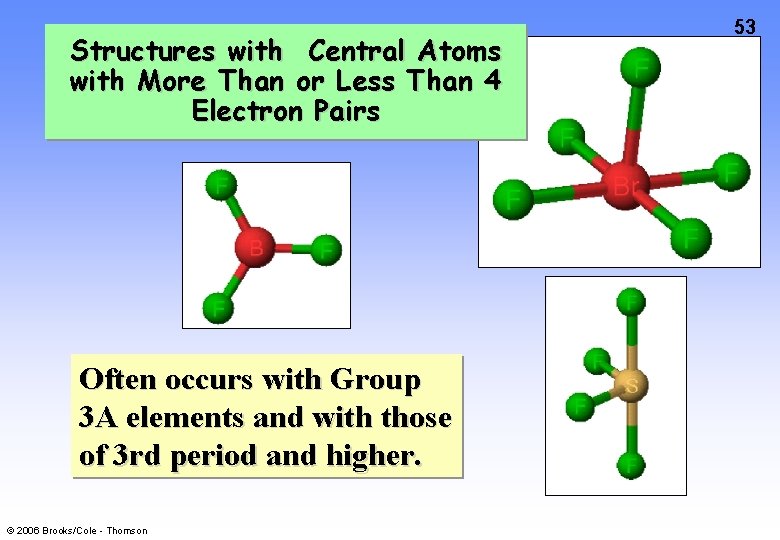
Structures with Central Atoms with More Than or Less Than 4 Electron Pairs Often occurs with Group 3 A elements and with those of 3 rd period and higher. © 2006 Brooks/Cole - Thomson 53

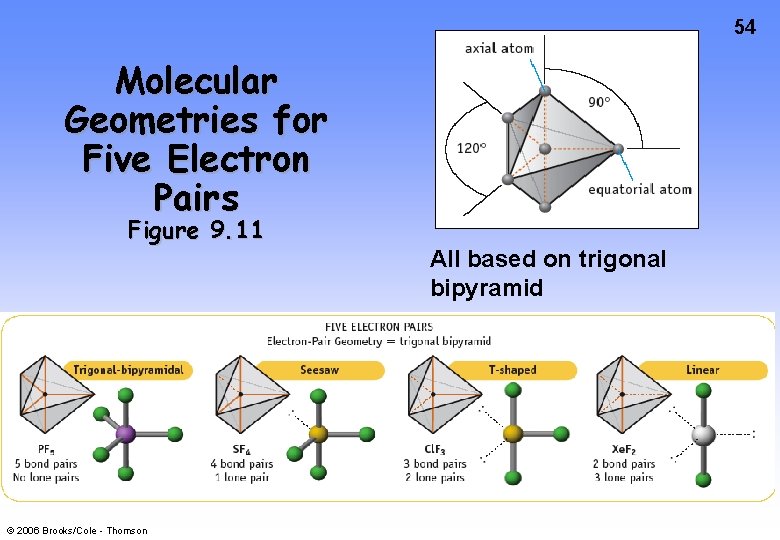
54 Molecular Geometries for Five Electron Pairs Figure 9. 11 © 2006 Brooks/Cole - Thomson All based on trigonal bipyramid

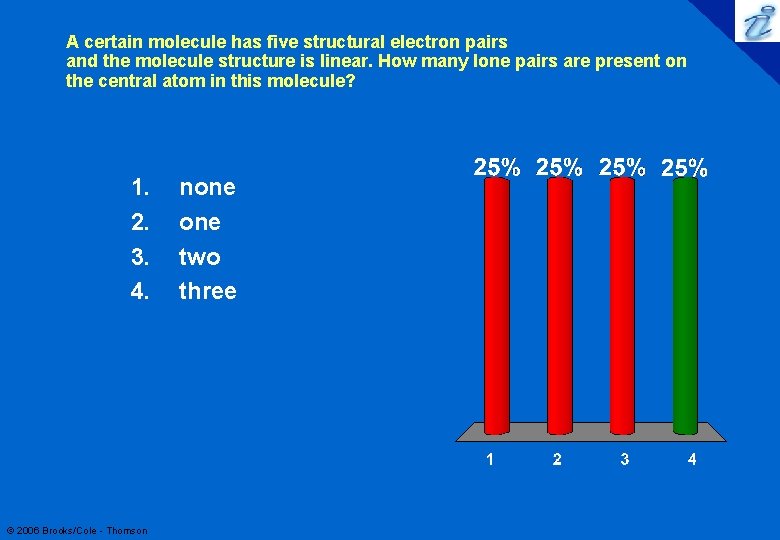
A certain molecule has five structural electron pairs and the molecule structure is linear. How many lone pairs are present on the central atom in this molecule? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson none two three 55

56 A certain molecule has six structural electron pairs and the molecule structure is a square pyramid. How many lone pairs are present on the central atom in this molecule? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson none two one three

59 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson

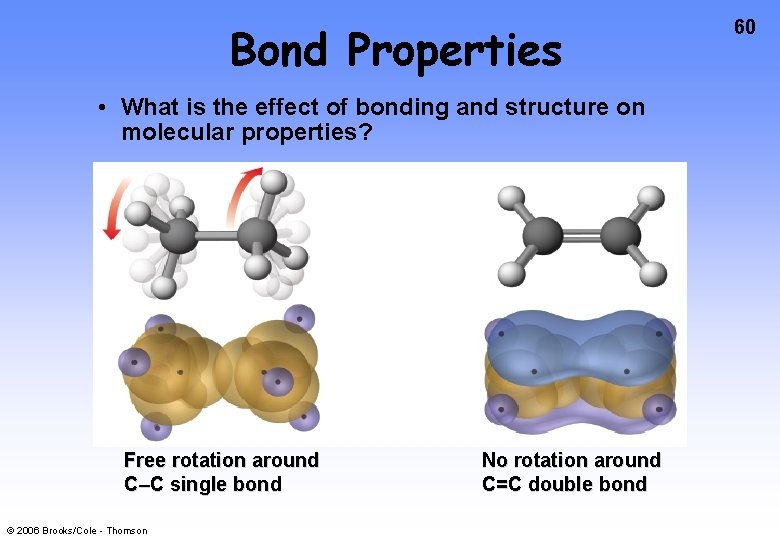
Bond Properties • What is the effect of bonding and structure on molecular properties? Free rotation around C–C single bond © 2006 Brooks/Cole - Thomson No rotation around C=C double bond 60

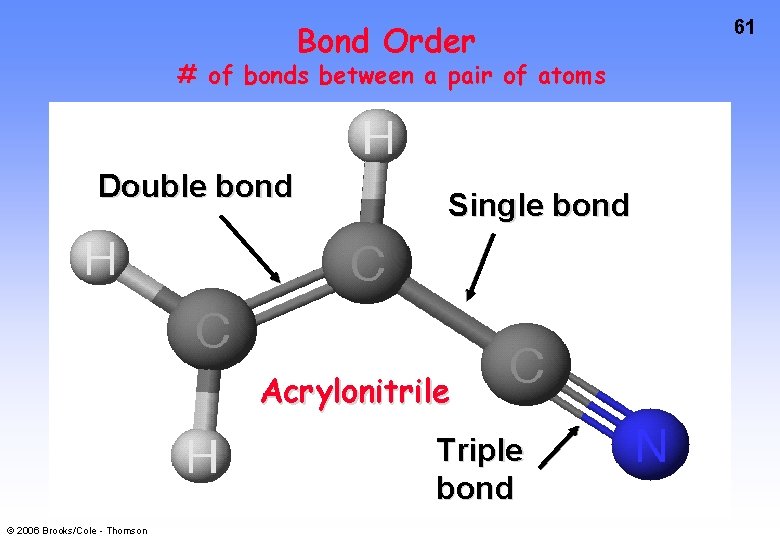
Bond Order # of bonds between a pair of atoms Double bond Single bond Acrylonitrile Triple bond © 2006 Brooks/Cole - Thomson 61

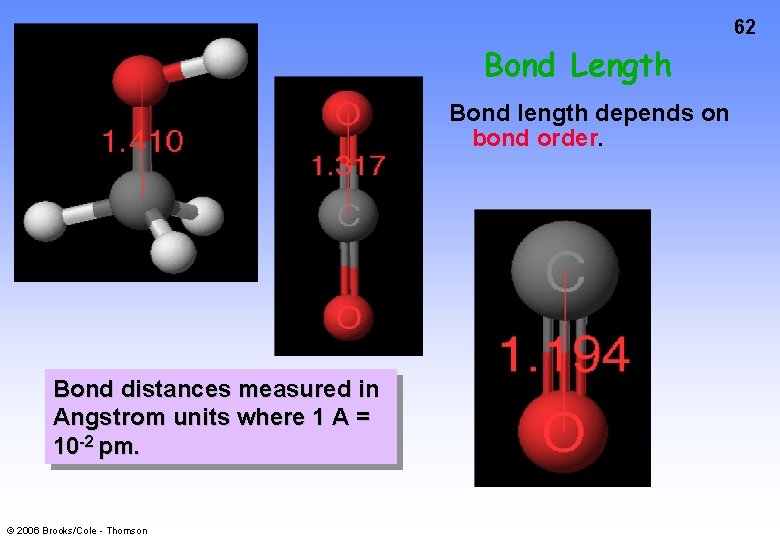
62 Bond Length Bond length depends on bond order. Bond distances measured in Angstrom units where 1 A = 10 -2 pm. © 2006 Brooks/Cole - Thomson

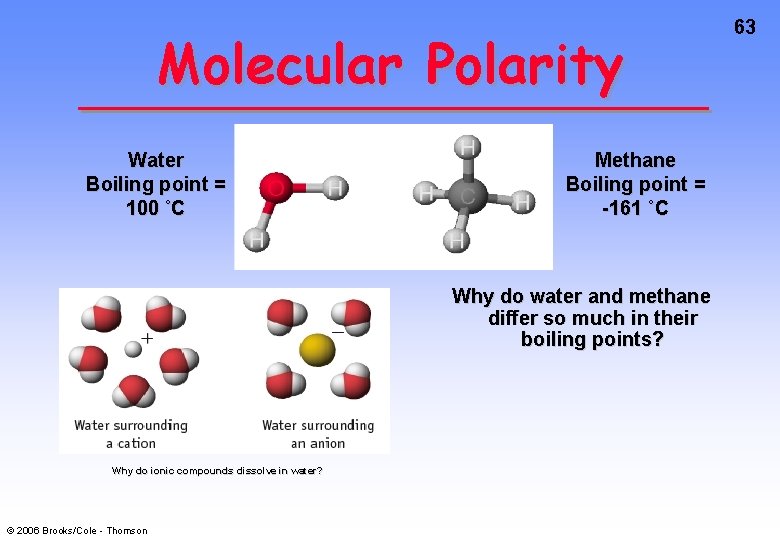
Molecular Polarity Water Boiling point = 100 ˚C Methane Boiling point = -161 ˚C Why do water and methane differ so much in their boiling points? Why do ionic compounds dissolve in water? © 2006 Brooks/Cole - Thomson 63

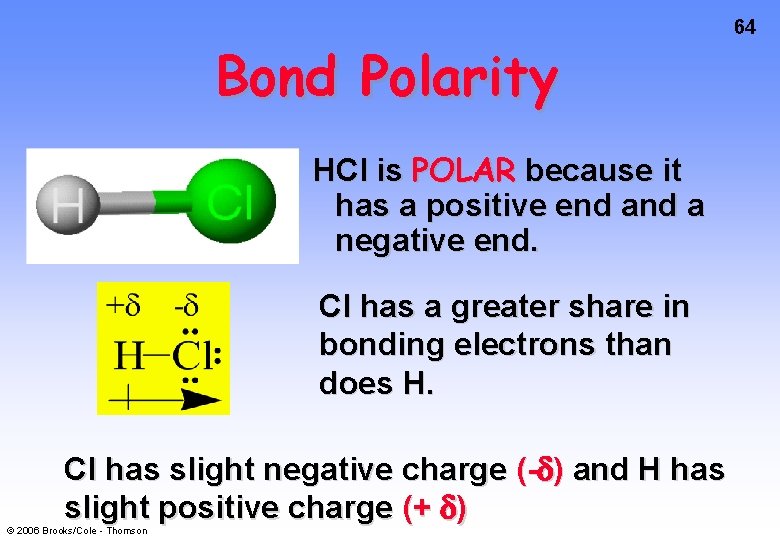
Bond Polarity HCl is POLAR because it has a positive end a negative end. Cl has a greater share in bonding electrons than does H. Cl has slight negative charge (- ) and H has slight positive charge (+ ) © 2006 Brooks/Cole - Thomson 64

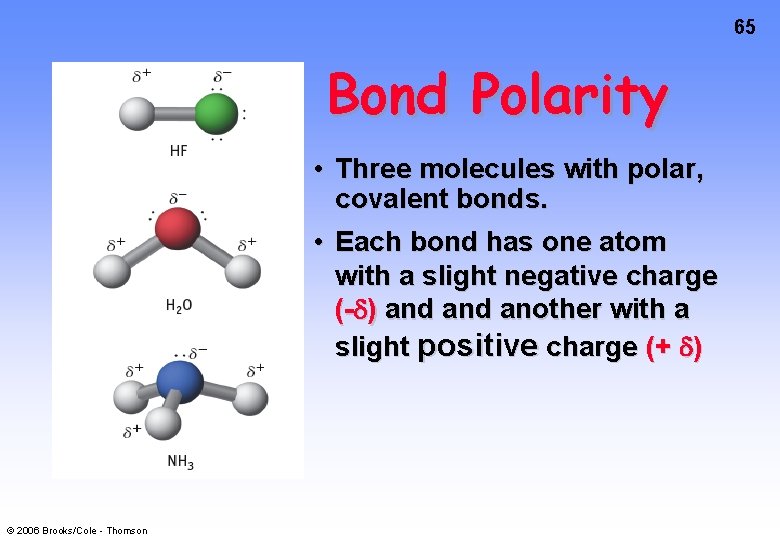
65 Bond Polarity • Three molecules with polar, covalent bonds. • Each bond has one atom with a slight negative charge (- ) and another with a slight positive charge (+ ) © 2006 Brooks/Cole - Thomson

Bond Polarity This model, calc’d using CAChe software for molecular calculations, shows that H is + (red) and Cl is - (yellow). Calc’d charge is + or - 0. 20. © 2006 Brooks/Cole - Thomson 66

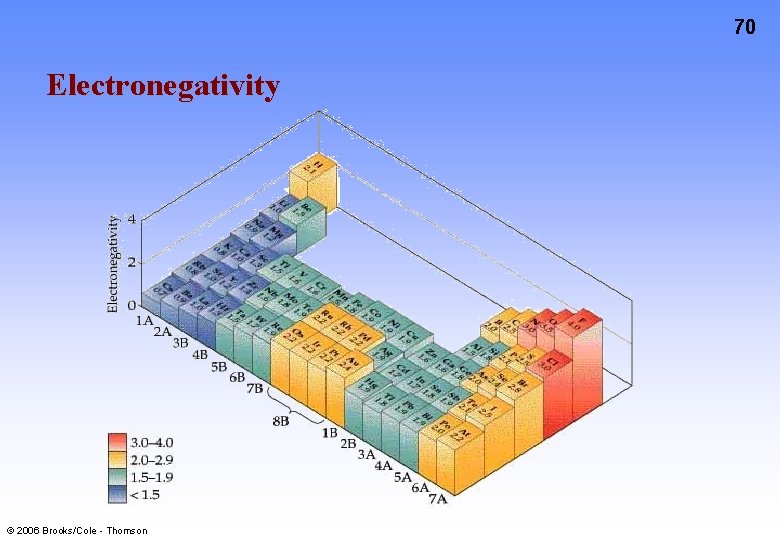
68 Electronegativity, is a measure of the ability of an atom in a molecule to attract electrons to itself. Concept proposed by Linus Pauling 1901 -1994 © 2006 Brooks/Cole - Thomson

Linus Pauling, 1901 -1994 The only person to receive two unshared Nobel prizes (for Peace and Chemistry). Chemistry areas: bonding, electronegativity, protein structure © 2006 Brooks/Cole - Thomson 69

70 Electronegativity © 2006 Brooks/Cole - Thomson

Electronegativity Figure 9. 14 © 2006 Brooks/Cole - Thomson 71

72 Electronegativity and Bond Polarity Difference in electronegativity is a gauge of bond polarity. If differences are: • around 0…then the covalent bond is non-polar • around 2…then the covalent bond is polar • around 3…then the bond is ionic There is no sharp distinction between bonding types. The positive end (or pole) in a polar bond is represented + and the negative pole -. © 2006 Brooks/Cole - Thomson

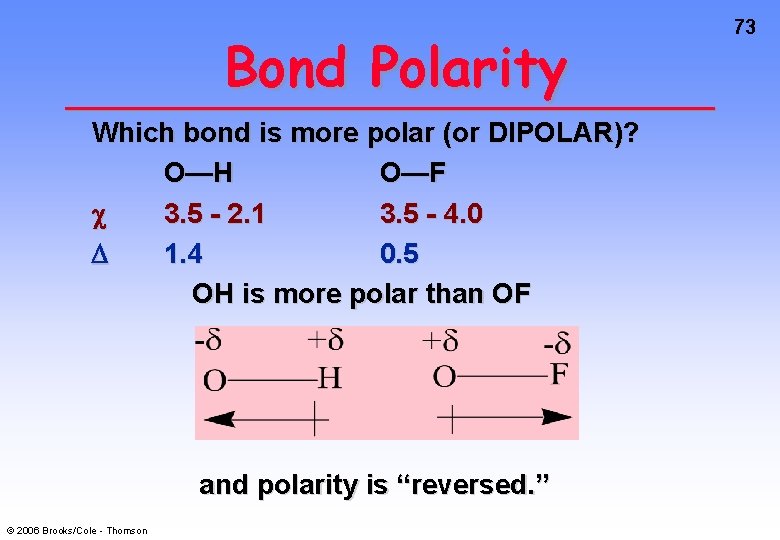
Bond Polarity Which bond is more polar (or DIPOLAR)? O—H O—F 3. 5 - 2. 1 3. 5 - 4. 0 1. 4 0. 5 OH is more polar than OF and polarity is “reversed. ” © 2006 Brooks/Cole - Thomson 73

74 Which of the following groups of elements is arranged correctly in order of increasing electronegativity? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson B < O < Al < F B < O < F < Al Al < B < O < F F < O < B < Al

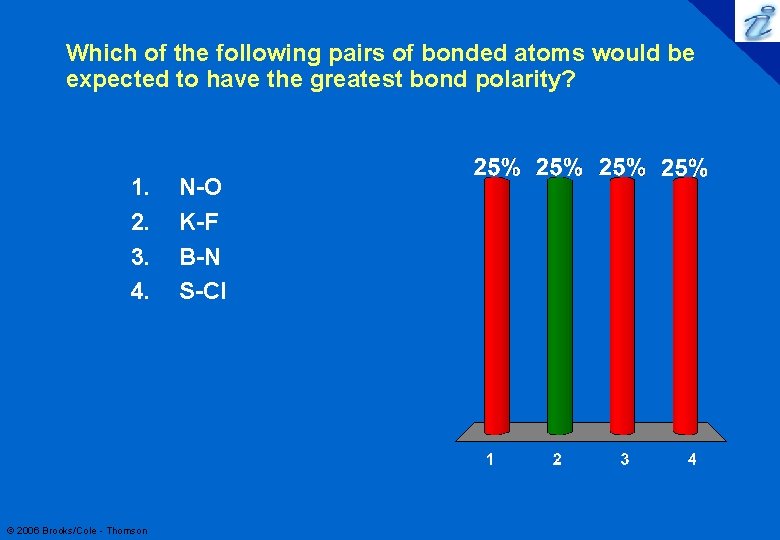
75 Which of the following pairs of bonded atoms would be expected to have the greatest bond polarity? 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson N-O K-F B-N S-Cl

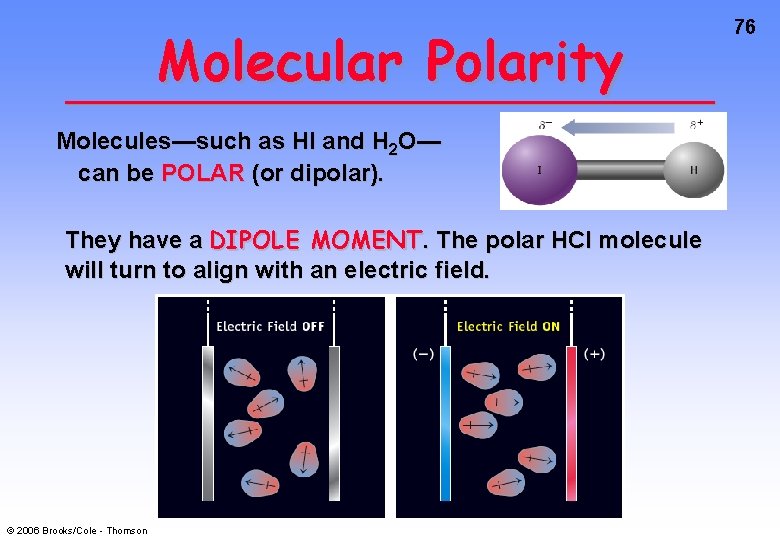
Molecular Polarity Molecules—such as HI and H 2 O— can be POLAR (or dipolar). They have a DIPOLE MOMENT. The polar HCl molecule will turn to align with an electric field. © 2006 Brooks/Cole - Thomson 76

Molecular Polarity The magnitude of the dipole is given in Debye units. Named for Peter Debye (1884 - 1966). Rec’d 1936 Nobel prize for work on xray diffraction and dipole moments. © 2006 Brooks/Cole - Thomson 77

Dipole Moments Why are some molecules polar but others are not? © 2006 Brooks/Cole - Thomson 78

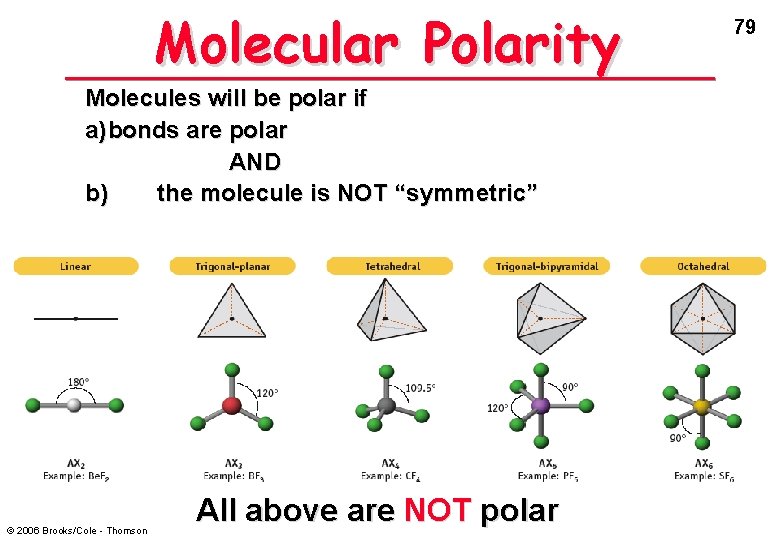
Molecular Polarity Molecules will be polar if a) bonds are polar AND b) the molecule is NOT “symmetric” © 2006 Brooks/Cole - Thomson All above are NOT polar 79

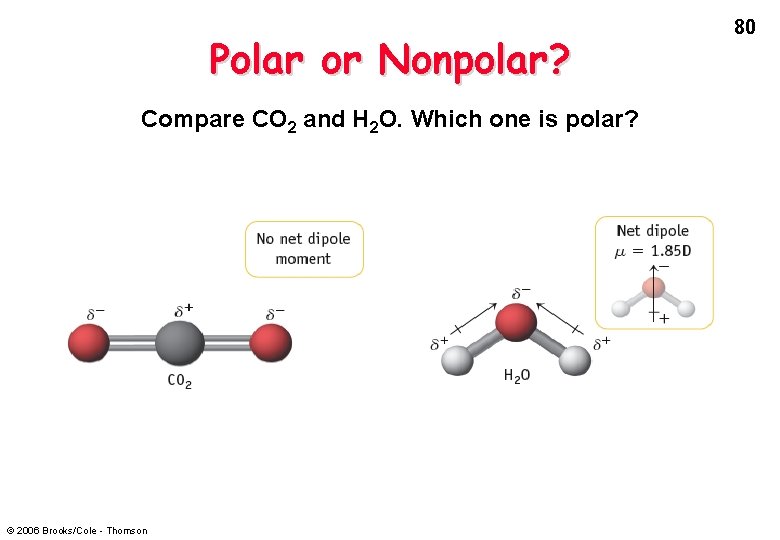
Polar or Nonpolar? Compare CO 2 and H 2 O. Which one is polar? © 2006 Brooks/Cole - Thomson 80

81 To determine whether or not a molecule is polar, 1. Mark all bonds that are polar. These are vectors; they have direction and charge. 2. Equal and opposite vectors cancel out. 3. Determine the direction of any uncancelled vectors. 4. If molecule is symmetric, and all bonded atoms are the same, the molecule is nonpolar. © 2006 Brooks/Cole - Thomson

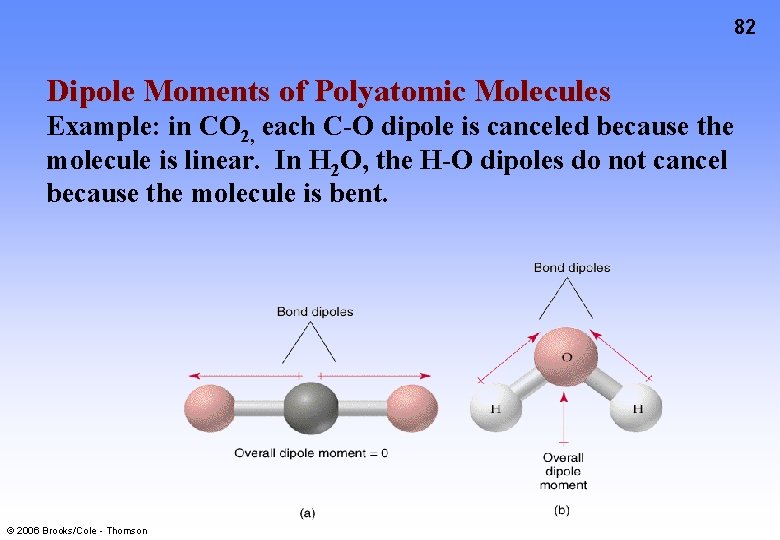
82 Dipole Moments of Polyatomic Molecules Example: in CO 2, each C-O dipole is canceled because the molecule is linear. In H 2 O, the H-O dipoles do not cancel because the molecule is bent. © 2006 Brooks/Cole - Thomson

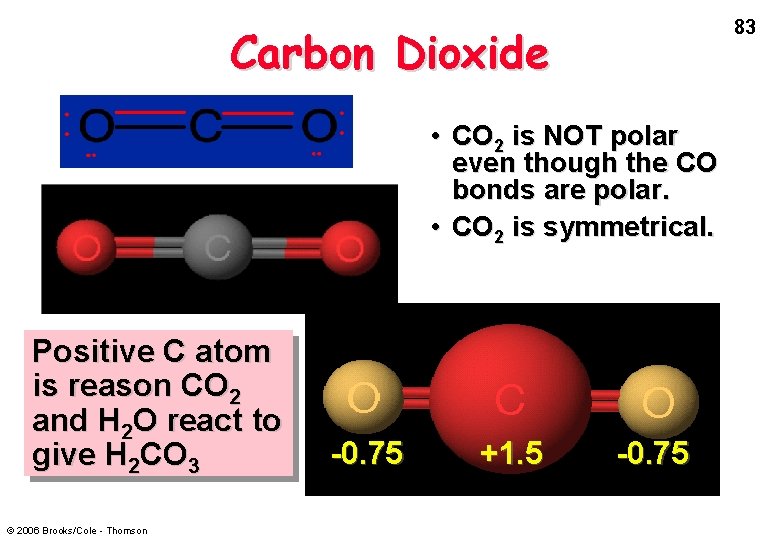
83 Carbon Dioxide • CO 2 is NOT polar even though the CO bonds are polar. • CO 2 is symmetrical. Positive C atom is reason CO 2 and H 2 O react to give H 2 CO 3 © 2006 Brooks/Cole - Thomson -0. 75 +1. 5 -0. 75

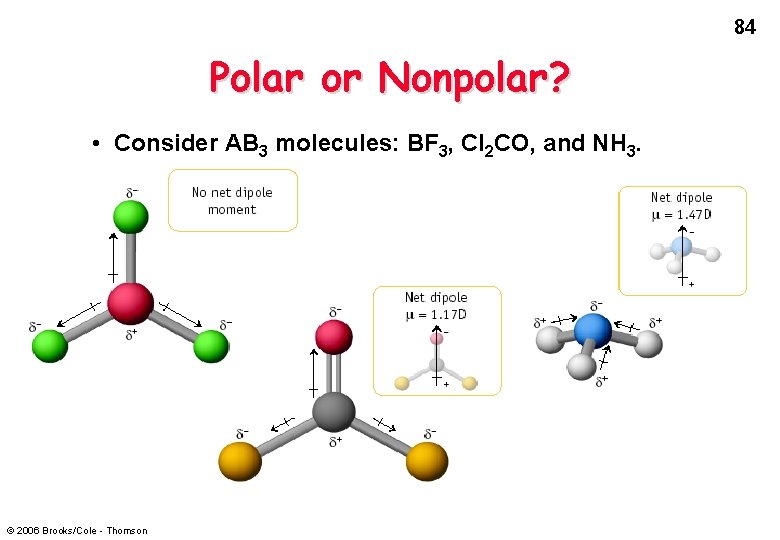
84 Polar or Nonpolar? • Consider AB 3 molecules: BF 3, Cl 2 CO, and NH 3. © 2006 Brooks/Cole - Thomson

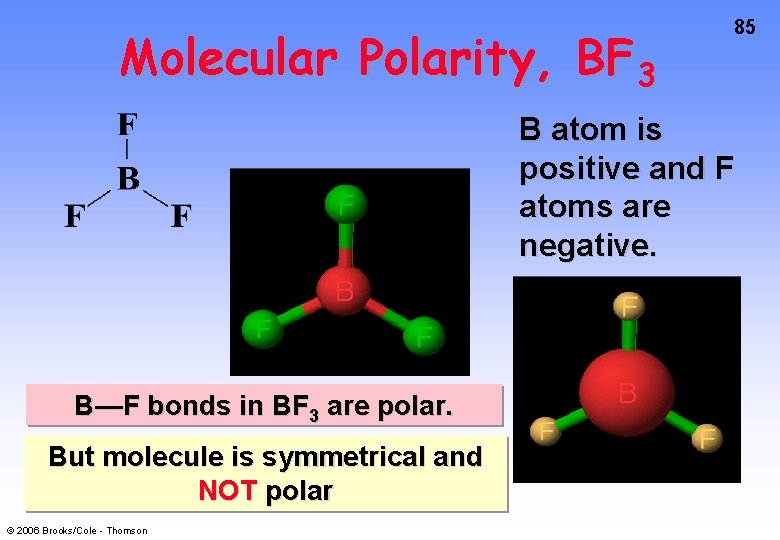
Molecular Polarity, BF 3 85 B atom is positive and F atoms are negative. B—F bonds in BF 3 are polar. But molecule is symmetrical and NOT polar © 2006 Brooks/Cole - Thomson

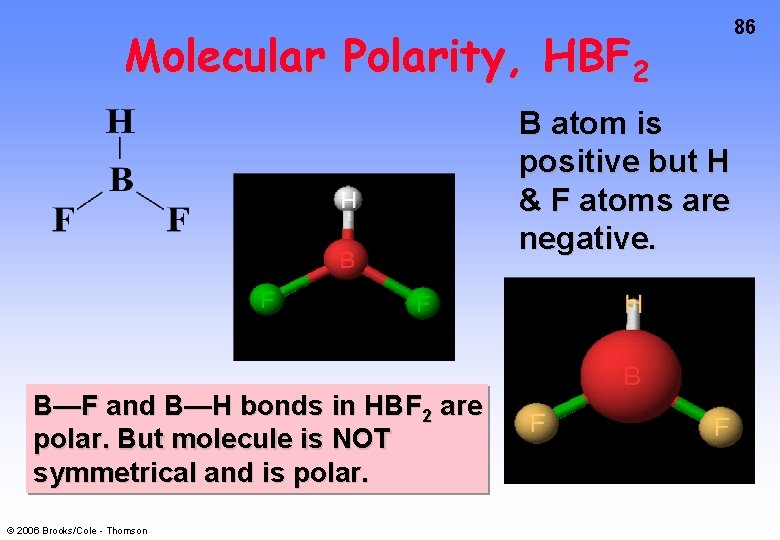
Molecular Polarity, HBF 2 B atom is positive but H & F atoms are negative. B—F and B—H bonds in HBF 2 are polar. But molecule is NOT symmetrical and is polar. © 2006 Brooks/Cole - Thomson 86

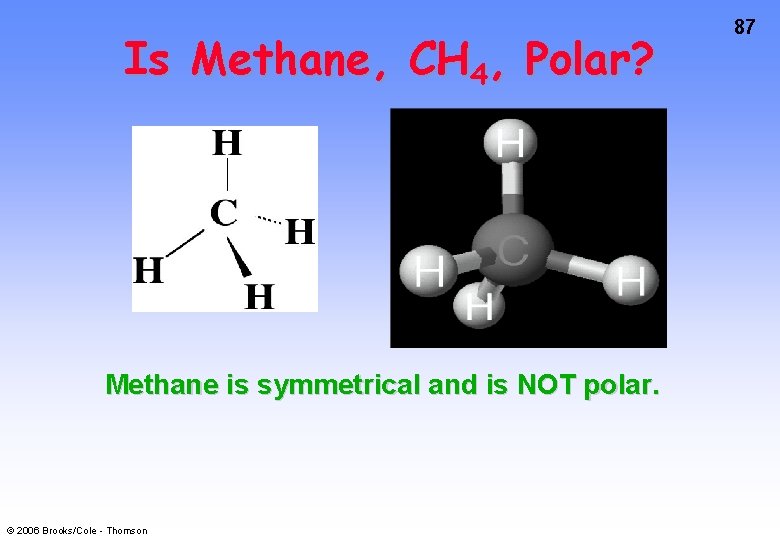
Is Methane, CH 4, Polar? Methane is symmetrical and is NOT polar. © 2006 Brooks/Cole - Thomson 87

Is CH 3 F Polar? C—F bond is very polar. Molecule is not symmetrical and so is polar. © 2006 Brooks/Cole - Thomson 88

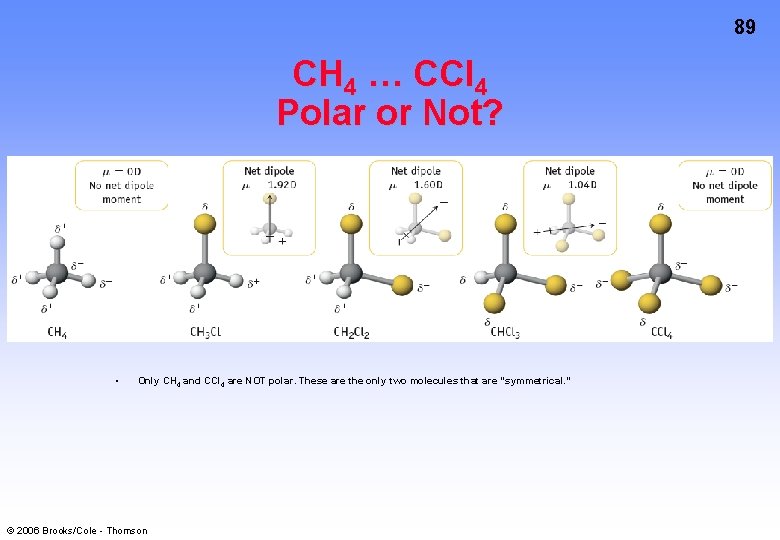
89 CH 4 … CCl 4 Polar or Not? • Only CH 4 and CCl 4 are NOT polar. These are the only two molecules that are “symmetrical. ” © 2006 Brooks/Cole - Thomson

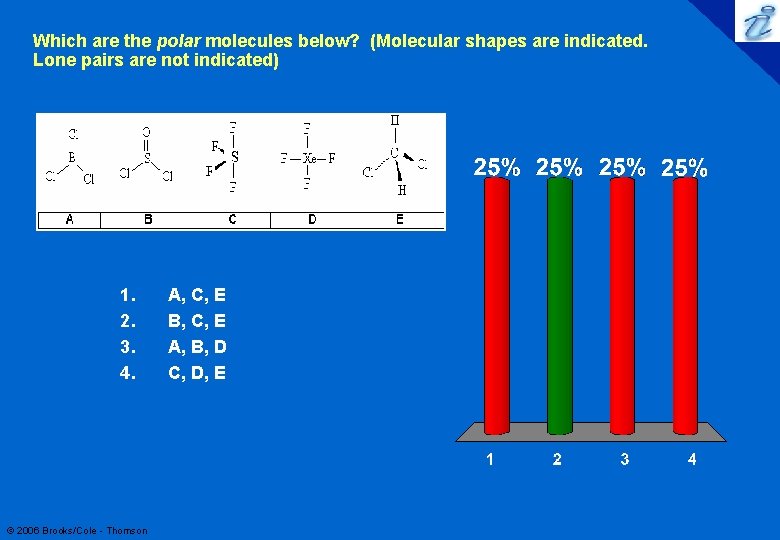
Which are the polar molecules below? (Molecular shapes are indicated. Lone pairs are not indicated) 1. 2. 3. 4. © 2006 Brooks/Cole - Thomson A, C, E B, C, E A, B, D C, D, E 90

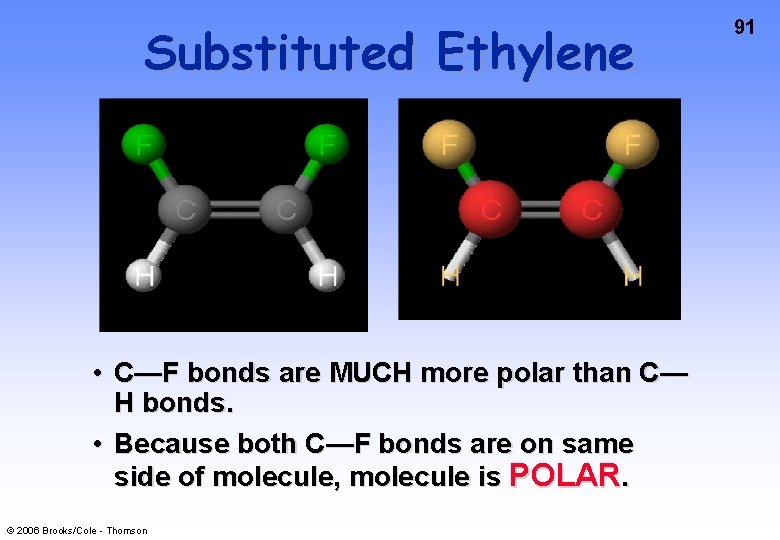
Substituted Ethylene • C—F bonds are MUCH more polar than C— H bonds. • Because both C—F bonds are on same side of molecule, molecule is POLAR. © 2006 Brooks/Cole - Thomson 91

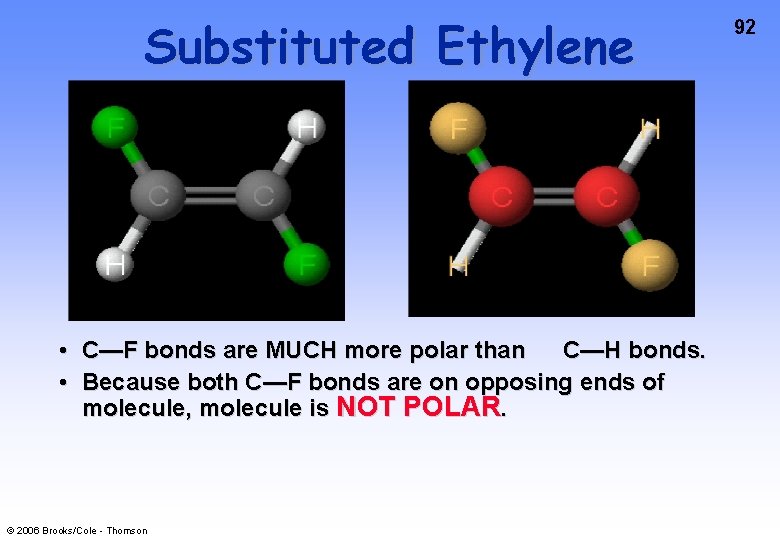
Substituted Ethylene • C—F bonds are MUCH more polar than C—H bonds. • Because both C—F bonds are on opposing ends of molecule, molecule is NOT POLAR. © 2006 Brooks/Cole - Thomson 92