Chapter 4 Reactions in Aqueous Solution A solution






































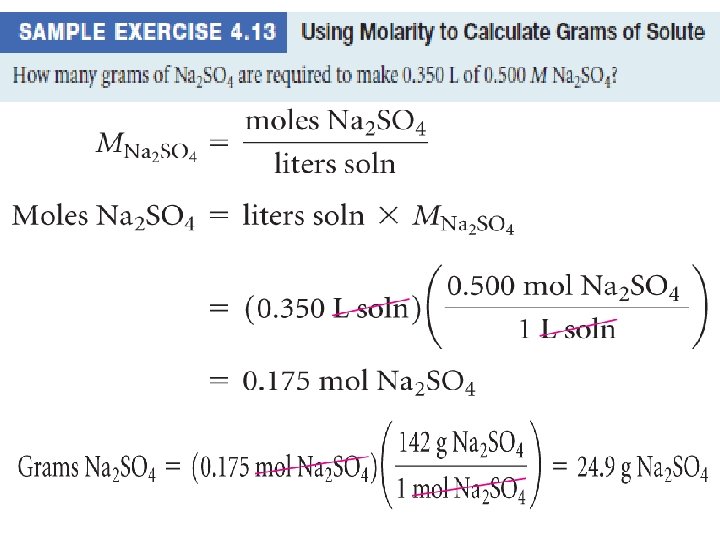





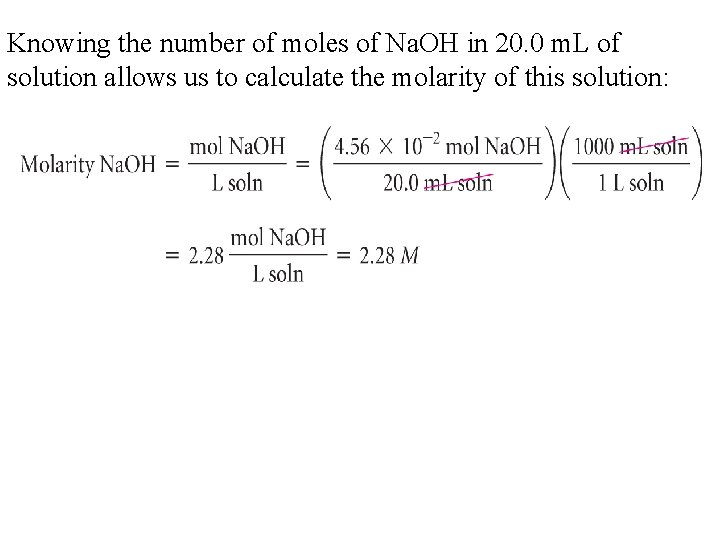

- Slides: 63

Chapter 4 Reactions in Aqueous Solution

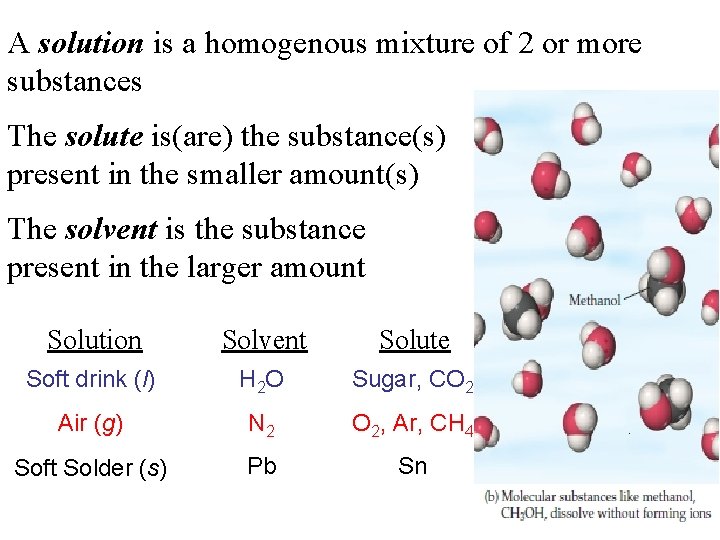
A solution is a homogenous mixture of 2 or more substances The solute is(are) the substance(s) present in the smaller amount(s) The solvent is the substance present in the larger amount Solution Solvent Solute Soft drink (l) H 2 O Sugar, CO 2 Air (g) N 2 O 2, Ar, CH 4 Soft Solder (s) Pb Sn

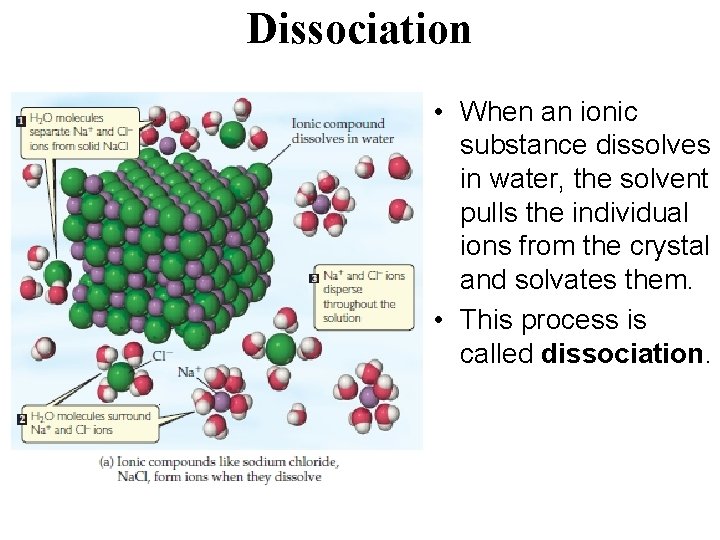
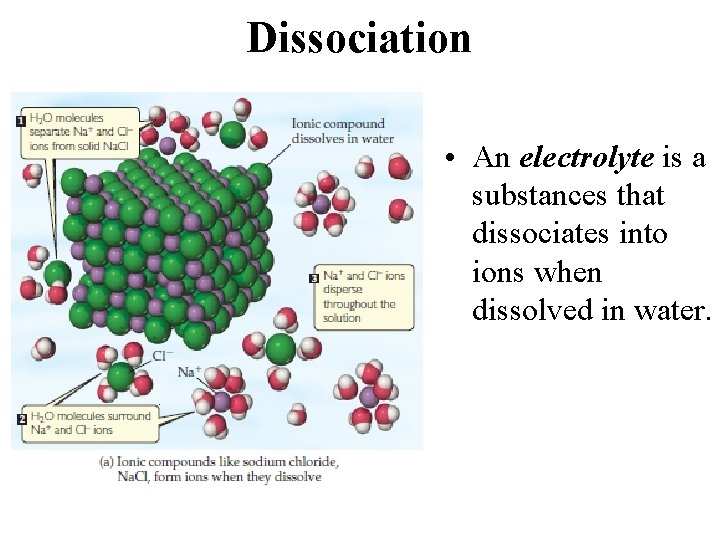
Dissociation • When an ionic substance dissolves in water, the solvent pulls the individual ions from the crystal and solvates them. • This process is called dissociation.

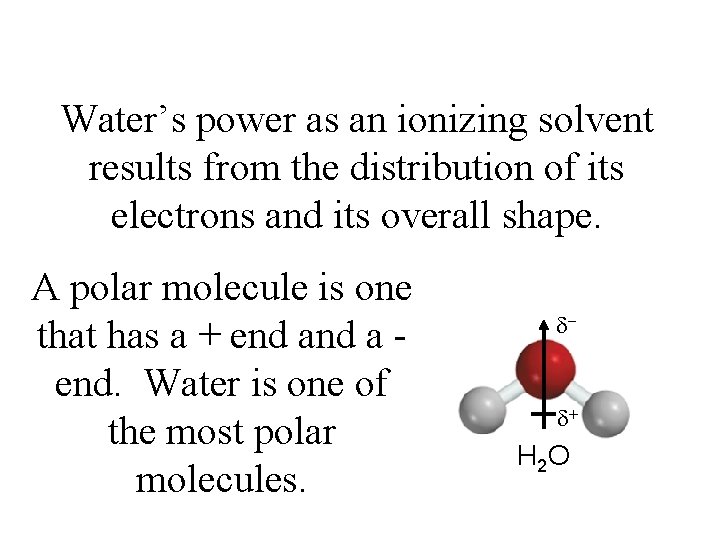
Water’s power as an ionizing solvent results from the distribution of its electrons and its overall shape. A polar molecule is one that has a + end a end. Water is one of the most polar molecules. d- d+ H 2 O

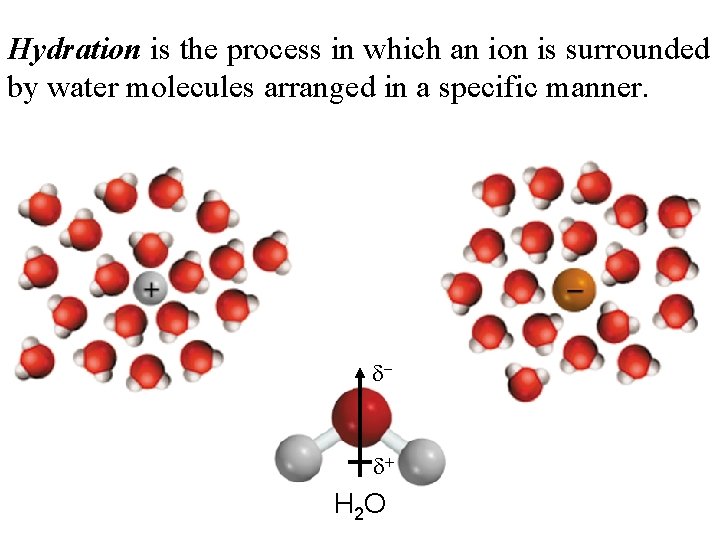
Hydration is the process in which an ion is surrounded by water molecules arranged in a specific manner. d- d+ H 2 O

Dissociation • An electrolyte is a substances that dissociates into ions when dissolved in water.

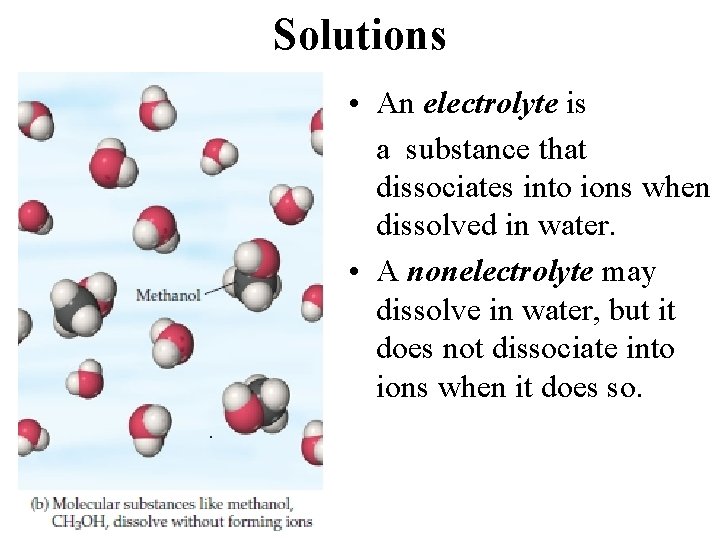
Solutions • An electrolyte is a substance that dissociates into ions when dissolved in water. • A nonelectrolyte may dissolve in water, but it does not dissociate into ions when it does so.

An electrolyte is a substance that, when dissolved in water, results in a solution that can conduct electricity. A nonelectrolyte is a substance that, when dissolved, results in a solution that does not conduct electricity. nonelectrolyte weak electrolyte strong electrolyte

Electrolytes and Nonelectrolytes Soluble ionic compounds tend to be electrolytes.

Electrolytes and Nonelectrolytes Molecular compounds tend to be nonelectrolytes, except for acids and bases.

Electrolytes • A strong electrolyte dissociates completely when dissolved in water. Strong Electrolyte – 100% dissociation Na. Cl (s) H 2 O Na+ (aq) + Cl- (aq) • A weak electrolyte only dissociates partially when dissolved in water. CH 3 COOH CH 3 COO- (aq) + H+ (aq) Reversible reaction - Chemical equilibrium

Strong Electrolytes Are… • Strong acids: HCl, HBr, HI, HNO 3, HCl. O 4, HCl. O 3, H 2 SO 4. • Strong bases: Group 1 A metal hydroxide [Li. OH, KOH, Na. OH, Rb. OH, Cs. OH] Heavy group 2 A metal hydroxides [Ca(OH)2, Sr(OH)2, Ba(OH)2]

Strong Electrolytes Are… • Strong acids • Strong bases • Soluble ionic salts

Precipitation Reactions When one mixes ions that form compounds that are insoluble (as could be predicted by the solubility guidelines), a precipitate is formed.

Metathesis (Exchange) Reactions • Metathesis comes from a Greek word that means “to transpose. ” Ag. NO 3(aq) + KCl(aq) Ag. Cl(s) + KNO 3(aq)

Metathesis (Exchange) Reactions • Metathesis comes from a Greek word that means “to transpose. ” • It appears as though the ions in the reactant compounds exchange, or transpose, ions: Ag. NO 3(aq) + KCl(aq) Ag. Cl(s) + KNO 3(aq)

Solution Chemistry • It is helpful to pay attention to exactly what species are present in a reaction mixture (i. e. , solid, liquid, gas, aqueous solution). • If we are to understand reactivity, we must be aware of just what is changing during the course of a reaction.

Precipitation Reactions Precipitate – insoluble solid that separates from solution precipitate Pb(NO 3)2 (aq) + 2 Na. I (aq) Pb. I 2 (s) + 2 Na. NO 3 (aq molecular equation Pb 2+ + 2 NO 3 - + 2 Na+ + 2 I- Pb. I 2 (s) + 2 Na+ + 2 NO 3 - ionic equation Pb. I 2 Pb 2+ + 2 I- Pb. I 2 (s) net ionic equation Na+ and NO 3 - are spectator ions


Writing Net Ionic Equations 1. Write a balanced molecular equation. 2. Dissociate all strong electrolytes. 3. Cross out anything that remains unchanged from the left side to the right side of the equation. 4. Write the net ionic equation with the species that remain.

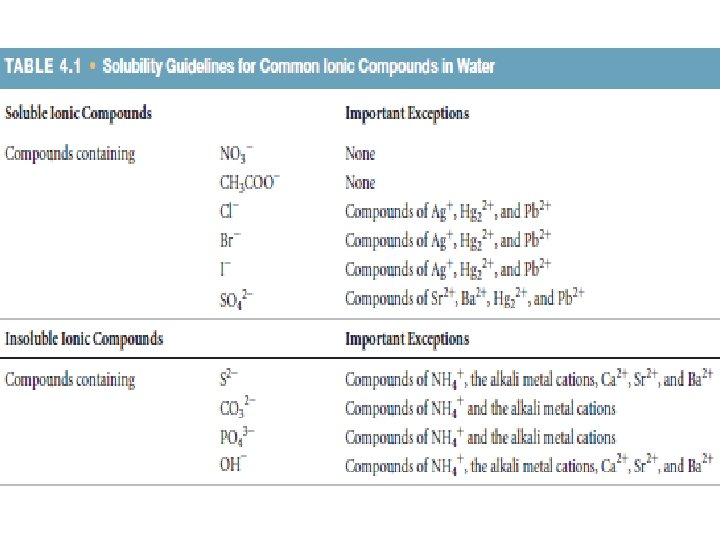
According to the solubility guidelines in Table 4. 1, Ca. CO 3 is insoluble and Na. Cl is soluble. The balanced molecular equation is Ca. Cl 2(aq) + Na 2 CO 3(aq) Ca. CO 3(s) + 2 Na. Cl(aq) Thus, the complete ionic equation is Ca 2+(aq) + 2 Cl–(aq) + 2 Na+(aq) + CO 32–(aq) Ca. CO 3(s) + 2 Na+(aq) + 2 Cl–(aq) Cl– and Na+ are spectator ions. Canceling them gives the following net ionic equation: Ca 2+(aq) + CO 32–(aq) Ca. CO 3(s)

Acids • The Swedish physicist and chemist S. A. Arrhenius defined acids as substances that increase the concentration of H+ when dissolved in water. • Both the Danish chemist J. N. Brønsted and the British chemist T. M. Lowry defined them as proton donors.

Acids There are only seven strong acids: • Hydrochloric (HCl) • Hydrobromic (HBr) • Hydroiodic (HI) • Nitric (HNO 3) • Sulfuric (H 2 SO 4) • Chloric (HCl. O 3) • Perchloric (HCl. O 4)

Acids that ionize to form one H+ ion are called monoprotic acids. Common monoprotic acids include HCl, HNO 3 and HC 2 H 3 O 2. Acids that ionize to form two H+ ions are called diprotic acids. A common diprotic acid is H 2 SO 4.

Bases • Arrhenius defined bases as substances that increase the concentration of OH− when dissolved in water. • Brønsted and Lowry defined them as proton acceptors.

Bases The strong bases are the soluble metal salts of hydroxide ion: • Alkali metals • Calcium • Strontium • Barium

Common bases are Na. OH, KOH, and Ca(OH)2. Compounds that do not contain OH– ions can also be bases. Proton transfer to NH 3 (a weak base) from water (a weak acid) is an example of an acid–base reaction. Since there is a mixture of NH 3, H 2 O, NH 4+, and OH– in solution, We write: NH 3(aq) + H 2 O(l) NH 4+(aq) + OH–(aq)

Strong and Weak Acids and Bases • Strong acids and strong bases are strong electrolytes. • They are completely ionized in solution. • Strong bases include: Group 1 A metal hydroxides, Ca(OH)2, Ba(OH)2, and Sr(OH)2. • Strong acids include: HCl, HBr, HI, HCl. O 3, HCl. O 4, H 2 SO 4, and HNO 3. • We write the ionization of HCl as: HCl H+ + Cl– • Weak acids and weak bases are weak electrolytes. • They are partially ionized in aqueous solution. • HF(aq) is a weak acid; most acids are weak acids. • We write the ionization of HF as: HF H+ + F–

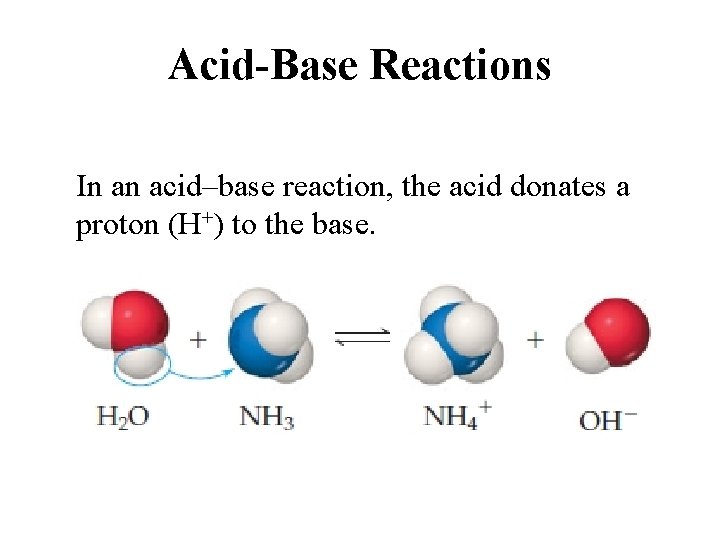
Acid-Base Reactions In an acid–base reaction, the acid donates a proton (H+) to the base.

Neutralization Reactions Generally, when solutions of an acid and a base are combined, the products are a salt and water: CH 3 COOH(aq) + Na. OH(aq) CH 3 COONa(aq) + H 2 O(l)

Neutralization Reactions When a strong acid reacts with a strong base, the net ionic equation is HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l)

Neutralization Reactions When a strong acid reacts with a strong base, the net ionic equation is HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l) H+(aq) + Cl−(aq) + Na+(aq) + OH−(aq) Na+(aq) + Cl−(aq) + H 2 O(l)

Neutralization Reactions When a strong acid reacts with a strong base, the net ionic equation is HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(l) H+(aq) + Cl−(aq) + Na+(aq) + OH−(aq) Na+(aq) + Cl−(aq) + H 2 O(l) H+(aq) + OH−(aq) H 2 O(l)

The balanced molecular equation is: The complete ionic equation is: The net ionic equation is:

Gas-Forming Reactions • Some metathesis reactions do not give the product expected. • In this reaction, the expected product (H 2 CO 3) decomposes to give a gaseous product (CO 2): Ca. CO 3(s) + 2 HCl(aq) Ca. Cl 2(aq) + CO 2(g) + H 2 O(l)

Gas-Forming Reactions When a carbonate or bicarbonate reacts with an acid, the products are a salt, carbon dioxide, and water: Ca. CO 3(s) + 2 HCl(aq) Ca. Cl 2(aq) + CO 2(g) + H 2 O(l) Na. HCO 3(aq) + HBr(aq) Na. Br(aq) + CO 2(g) + H 2 O(l)

Gas-Forming Reactions Similarly, when a sulfite reacts with an acid, the products are a salt, sulfur dioxide, and water: Sr. SO 3(s) + 2 HI(aq) Sr. I 2(aq) + SO 2(g) + H 2 O(l)

Gas-Forming Reactions • This reaction gives the predicted product, but you had better carry it out in the hood, or you will be very unpopular! • But just as in the previous examples, a gas is formed as a product of this reaction: Na 2 S(aq) + H 2 SO 4(aq) Na 2 SO 4(aq) + H 2 S(g)

Oxidation-Reduction Reactions • An oxidation occurs when an atom or ion loses electrons. • A reduction occurs when an atom or ion gains electrons. • One cannot occur without the other.

Oxidation Numbers To determine if an oxidation–reduction reaction has occurred, we assign an oxidation number to each element in a neutral compound or charged entity.

Oxidation Numbers § Elements in their elemental form have an oxidation number of 0. § The oxidation number of a monatomic ion is the same as its charge. § Nonmetals tend to have negative oxidation numbers, although some are positive in certain compounds or ions

Oxidation Numbers § Fluorine always has an oxidation number of − 1. § The other halogens have an oxidation number of − 1 when they are negative; they can have positive oxidation numbers, however, most notably in oxyanions. § Hydrogen is − 1 when bonded to a metal, +1 when bonded to a nonmetal. § Oxygen has an oxidation number of − 2, except in the peroxide ion, in which it has an oxidation number of − 1.

Oxidation Numbers § The sum of the oxidation numbers in a neutral compound is 0. § The sum of the oxidation numbers in a polyatomic ion is the charge on the ion.

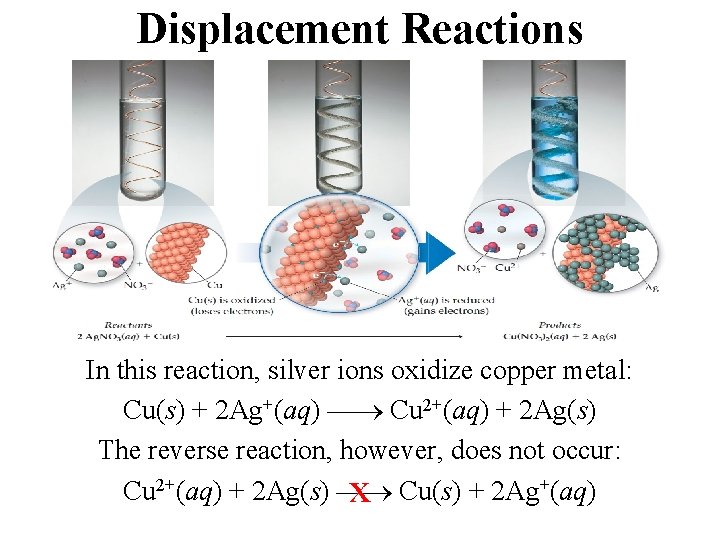
Displacement Reactions • In displacement reactions, ions oxidize an element. • The ions, then, are reduced.

Displacement Reactions In this reaction, silver ions oxidize copper metal: Cu(s) + 2 Ag+(aq) Cu 2+(aq) + 2 Ag(s) The reverse reaction, however, does not occur: Cu 2+(aq) + 2 Ag(s) x Cu(s) + 2 Ag+(aq)

The molecular equation: 2 Al(s) + 6 HBr(aq) 2 Al. Br 3(aq) + 3 H 2(g) Both HBr and Al. Br 3 are soluble strong electrolytes. Thus, the complete ionic equation is: 2 Al(s) + 6 H+(aq) + 6 Br–(aq) + 3 H 2(g) 2 Al 3+(aq) + Because Br– is a spectator ion, the net ionic equation is: 2 Al(s) + 6 H+(aq) 2 Al 3+(aq) + 3 H 2(g)

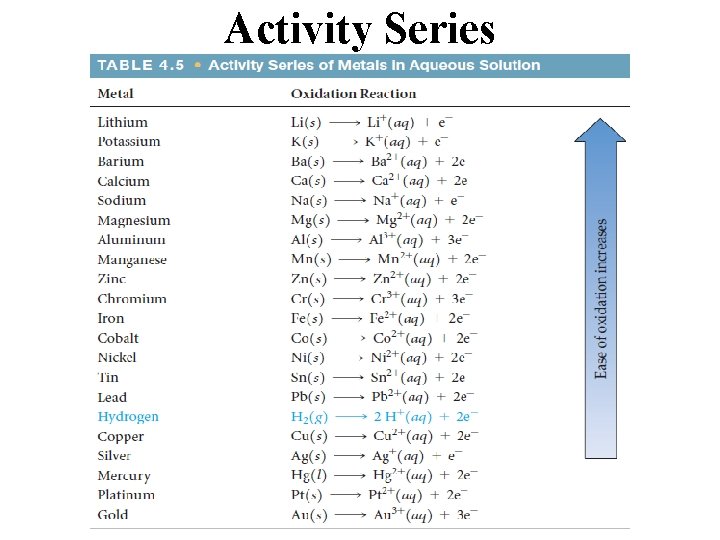
Activity Series

The magnesium salt formed in the reaction is Mg. Cl 2, meaning The balanced molecular equation is: Mg(s) + Fe. Cl 2(aq) Mg. Cl 2(aq) + Fe(s) Both Fe. Cl 2 and Mg. Cl 2 are soluble strong electrolytes and can be written in ionic form, which shows us that Cl–is a spectator ion in the reaction. The net ionic equation is: Mg(s) + Fe 2+(aq) Mg 2+(aq) + Fe(s) The net ionic equation shows that Mg is oxidized and Fe 2+ is reduced in this reaction.

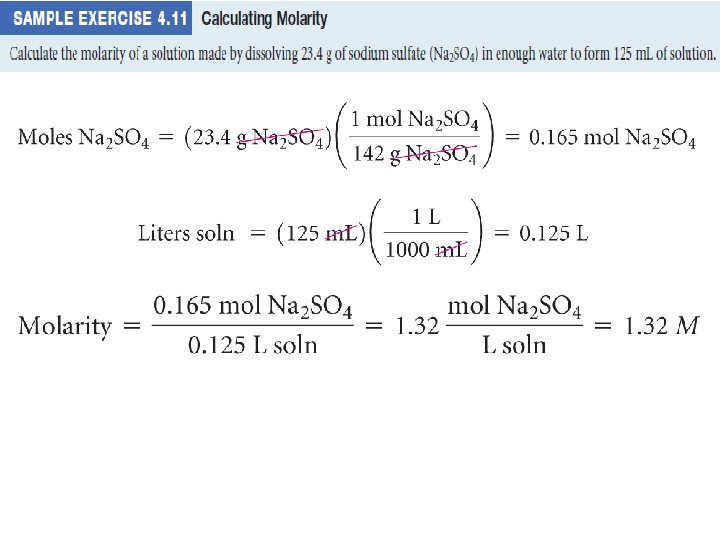
Molarity • Two solutions can contain the same compounds but be quite different because the proportions of those compounds are different. • Molarity is one way to measure the concentration of a solution: moles of solute Molarity (M) = volume of solution in liters

Mixing a Solution • To create a solution of a known molarity, one weighs out a known mass (and, therefore, number of moles) of the solute. • The solute is added to a volumetric flask, and solvent is added to the line on the neck of the flask.


Calcium nitrate is composed of calcium ions (Ca 2+) and nitrate ions (NO 3–), so its chemical formula is Ca(NO 3)2. Because there are two NO 3– ions for each Ca 2+ ion, each mole of Ca(NO 3)2 that dissolves dissociates into 1 mol of Ca 2+ and 2 mol of NO 3–. Thus, a solution that is 0. 025 M in Ca(NO 3)2 is 0. 025 M in Ca 2+ and 2 0. 025 M = 0. 050 M in NO 3–:
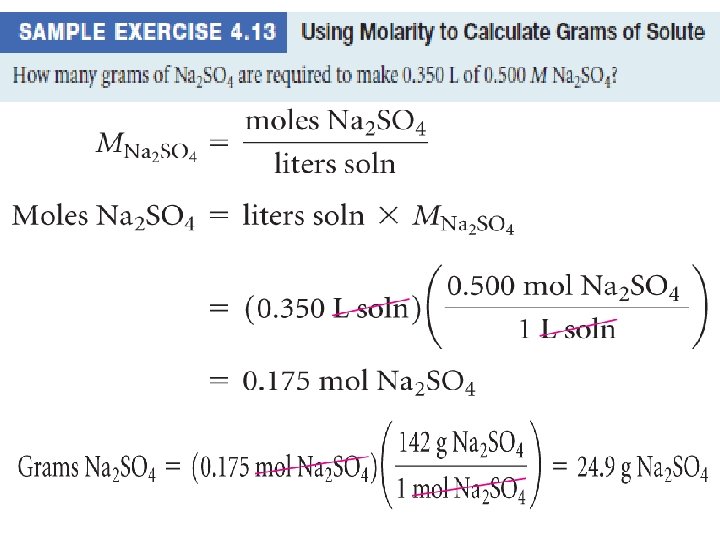

Dilution • One can also dilute a more concentrated solution by – Using a pipet to deliver a volume of the solution to a new volumetric flask, and – Adding solvent to the line on the neck of the new flask.

Dilution The molarity of the new solution can be determined from the equation Mc V c = Md V d where Mc and Md are the molarity of the concentrated and dilute soln. , respectively, and Vc and Vd are the volumes of the two solutions.


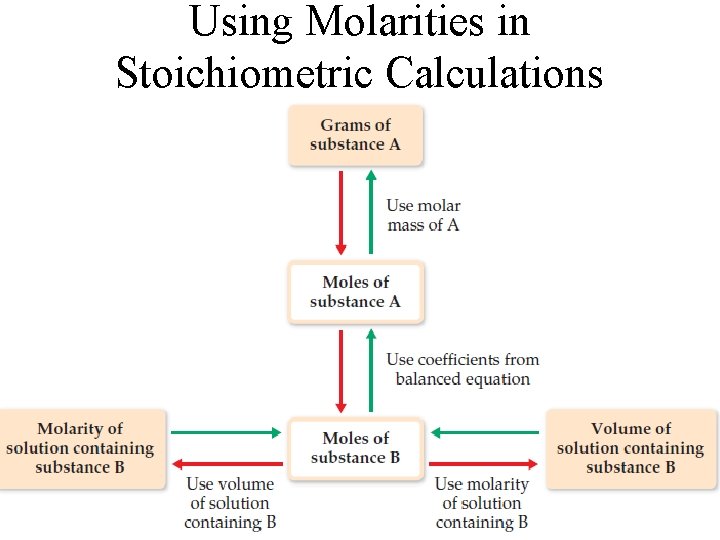
Using Molarities in Stoichiometric Calculations
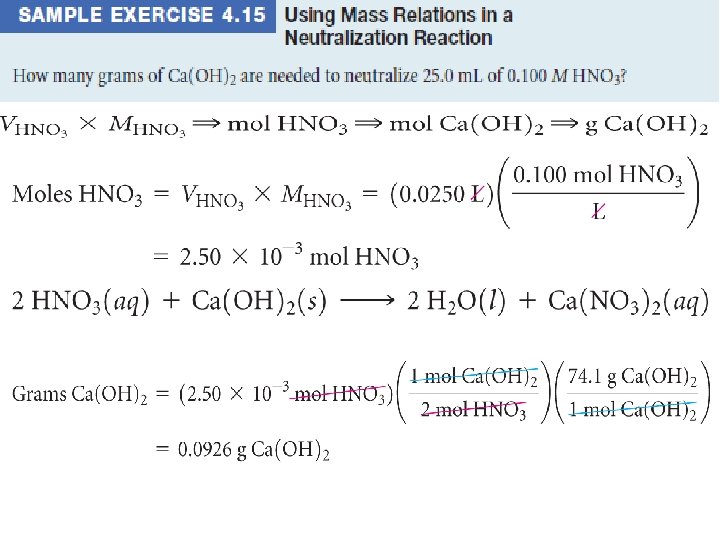

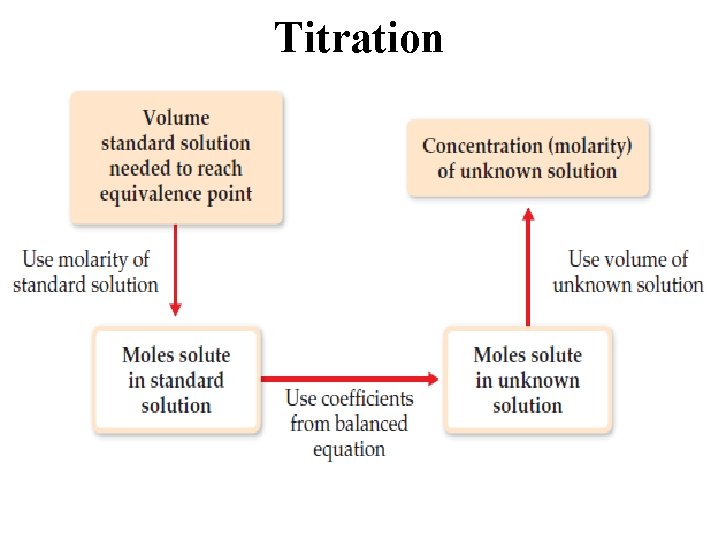
Titration is an analytical technique in which one can calculate the concentration of a solute in a solution.

Titration

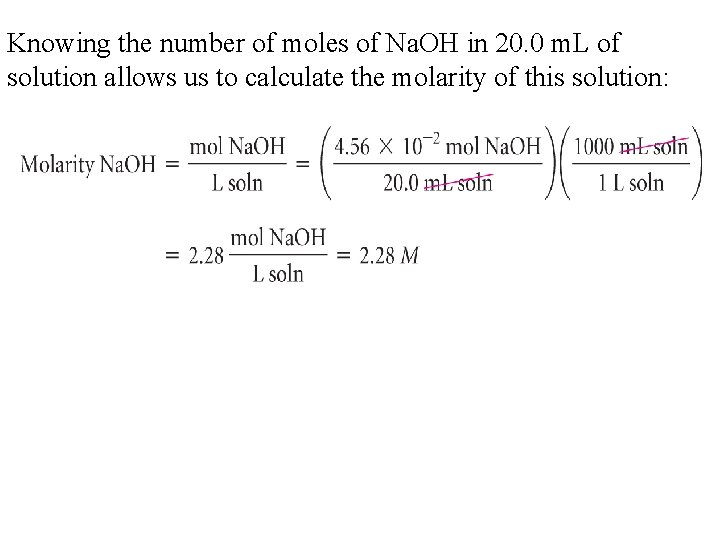
Knowing the number of moles of Na. OH in 20. 0 m. L of solution allows us to calculate the molarity of this solution:
