Chapter 15 Additional Aspects of Aqueous Equilibria 2009

Chapter 15 Additional Aspects of Aqueous Equilibria © 2009, Prentice-Hall, Inc.

The Common-Ion Effect Consider CH 3 COOH(aq) + H 2 O(l) a solution of acetic acid: H 3 O+(aq) + CH 3 COO−(aq) If acetate ion is added to the solution, Le Châtelier says … the equilibrium will shift to the left. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

The Common-Ion Effect If a strong and weak electrolyte both have the same ion the weak electrolyte ionizes less, compared to if it was in solution alone. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

The Common-Ion Effect Calculate the fluoride ion concentration and p. H of a solution that is 0. 20 M in HF and 0. 10 M in HCl. Ka for HF is 6. 8 10− 4. [H 3 O+] [F−] = 6. 8 10 -4 Ka = [HF] Aqueous Equilibria © 2009, Prentice-Hall, Inc.

The Common-Ion Effect HF(aq) + H 2 O(l) H 3 O+(aq) + F−(aq) Because HCl, a strong acid, is also present, the initial [H 3 O+] is not 0, but rather 0. 10 M. [HF], M [H 3 O+], M [F−], M Initially 0. 20 0. 10 0 Change −x +x +x 0. 20 − x 0. 20 0. 10 + x 0. 10 x At Equilibrium Aqueous Equilibria © 2009, Prentice-Hall, Inc.

The Common-Ion Effect 6. 8 10− 4 (0. 10) (x) = (0. 20) (6. 8 10− 4) =x (0. 10) 1. 4 10− 3 = x Aqueous Equilibria © 2009, Prentice-Hall, Inc.
![The Common-Ion Effect Therefore, [F−] = x = 1. 4 10− 3 [H 3 The Common-Ion Effect Therefore, [F−] = x = 1. 4 10− 3 [H 3](http://slidetodoc.com/presentation_image_h/1e1b7978993f8bb4447a0d16d1f5251a/image-7.jpg)
The Common-Ion Effect Therefore, [F−] = x = 1. 4 10− 3 [H 3 O+] = 0. 10 + x = 0. 10 + 1. 4 10− 3 = 0. 10 M So, p. H = −log (0. 10) p. H = 1. 00 Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Buffers are solutions of a weak conjugate acid-base pair. They are particularly resistant to p. H changes, even when strong acid or base is added. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Buffers If a small amount of hydroxide is added to an equimolar solution of HF in Na. F, for example, the HF Aqueous reacts with the OH− to make F− and water. Equilibria © 2009, Prentice-Hall, Inc.
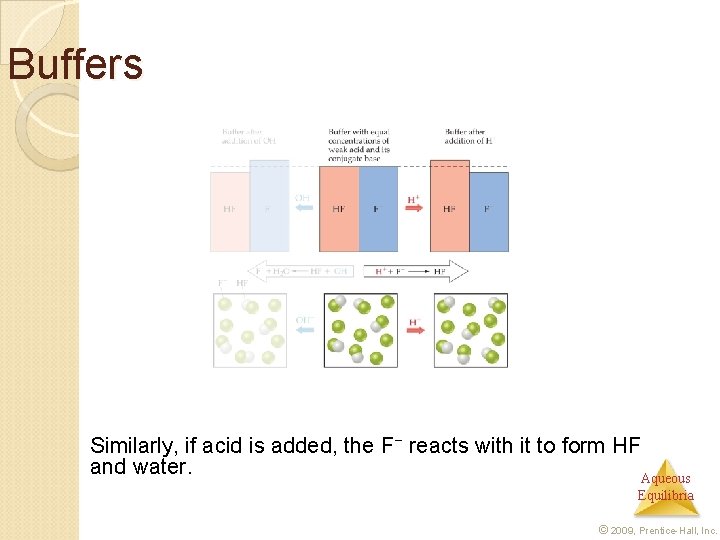
Buffers Similarly, if acid is added, the F− reacts with it to form HF and water. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Buffer Calculations Consider the equilibrium constant expression for the dissociation of a generic acid, HA: HA + H 2 O H 3 O + + A − [H 3 O+] [A−] Ka = [HA] Aqueous Equilibria © 2009, Prentice-Hall, Inc.
![Buffer Calculations Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA] Buffer Calculations Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA]](http://slidetodoc.com/presentation_image_h/1e1b7978993f8bb4447a0d16d1f5251a/image-12.jpg)
Buffer Calculations Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA] Taking the negative log of both side, we get base −] [A −log Ka = −log [H 3 O+] + −log [HA] p. Ka p. H acid © 2009, Prentice-Hall, Inc. Aqueous Equilibria
![Buffer Calculations So [base] p. Ka = p. H − log [acid] • Rearranging, Buffer Calculations So [base] p. Ka = p. H − log [acid] • Rearranging,](http://slidetodoc.com/presentation_image_h/1e1b7978993f8bb4447a0d16d1f5251a/image-13.jpg)
Buffer Calculations So [base] p. Ka = p. H − log [acid] • Rearranging, this becomes [base] p. H = p. Ka + log [acid] • This is the Henderson–Hasselbalch equation. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Henderson–Hasselbalch Equation What is the p. H of a buffer that is 0. 12 M in lactic acid, CH 3 CH(OH)COOH, and 0. 10 M in sodium lactate? Ka for lactic acid is 1. 4 10− 4. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
![Henderson–Hasselbalch Equation [base] p. H = p. Ka + log [acid] p. H = Henderson–Hasselbalch Equation [base] p. H = p. Ka + log [acid] p. H =](http://slidetodoc.com/presentation_image_h/1e1b7978993f8bb4447a0d16d1f5251a/image-15.jpg)
Henderson–Hasselbalch Equation [base] p. H = p. Ka + log [acid] p. H = −log (1. 4 10− 4) (0. 10) + log (0. 12) p. H = 3. 85 + (− 0. 08) p. H = 3. 77 Aqueous Equilibria © 2009, Prentice-Hall, Inc.

p. H Range The p. H range is the range of p. H values over which a buffer system works effectively. It is best to choose an acid with a p. Ka close to the desired p. H. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
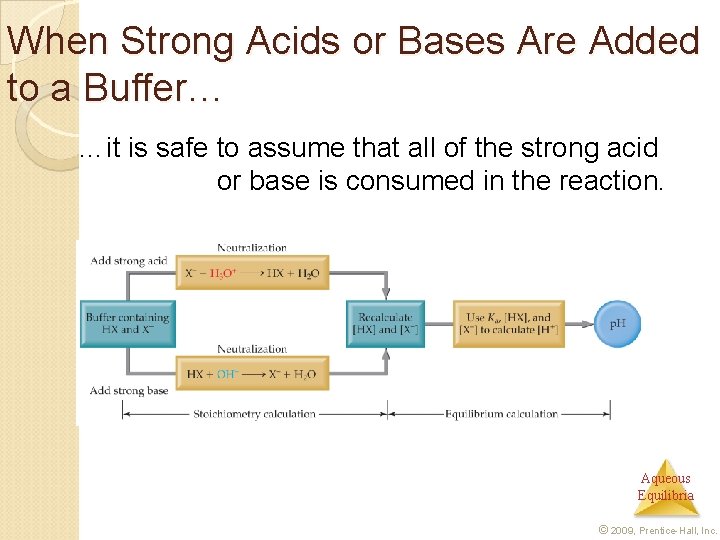
When Strong Acids or Bases Are Added to a Buffer… …it is safe to assume that all of the strong acid or base is consumed in the reaction. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Addition of Strong Acid or Base to a Buffer Determine how the neutralization reaction affects the amounts of the weak acid and its conjugate base in solution. 2. Use the Henderson–Hasselbalch equation to determine the new p. H of the solution. 1. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Calculating p. H Changes in Buffers A buffer is made by adding 0. 300 mol HC 2 H 3 O 2 and 0. 300 mol Na. C 2 H 3 O 2 to enough water to make 1. 00 L of solution. The p. H of the buffer is 4. 74. Calculate the p. H of this solution after 0. 020 mol of Na. OH is added. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Calculating p. H Changes in Buffers Before the reaction, since mol HC 2 H 3 O 2 = mol C 2 H 3 O 2− p. H = p. Ka = −log (1. 8 10− 5) = 4. 74 Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Calculating p. H Changes in Buffers The 0. 020 mol Na. OH will react with 0. 020 mol of the acetic acid: HC 2 H 3 O 2(aq) + OH−(aq) C 2 H 3 O 2−(aq) + H 2 O(l) HC 2 H 3 O 2 C 2 H 3 O 2 − OH− Before reaction 0. 300 mol 0. 020 mol After reaction 0. 280 mol 0. 320 mol 0. 000 mol Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Calculating p. H Changes in Buffers Now use the Henderson–Hasselbalch equation to calculate the new p. H: (0. 320) p. H = 4. 74 + log (0. 200) p. H = 4. 74 + 0. 06 p. H = 4. 80 Aqueous Equilibria © 2009, Prentice-Hall, Inc.
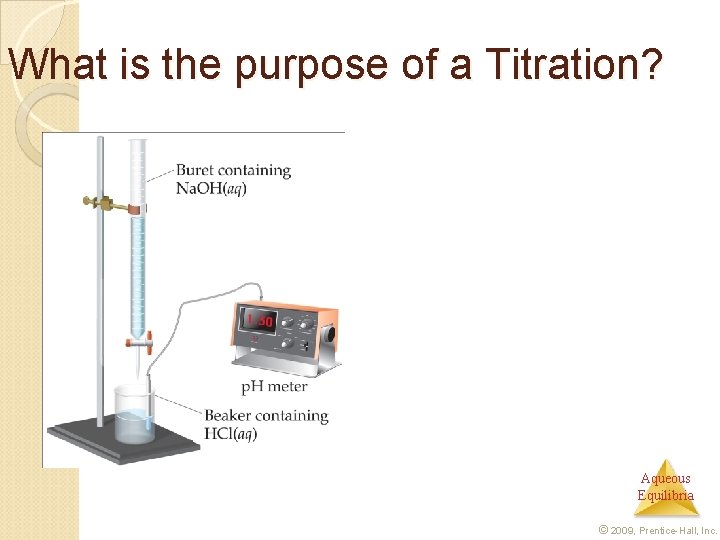
What is the purpose of a Titration? Aqueous Equilibria © 2009, Prentice-Hall, Inc.

What is the purpose of a Titration? In this technique a known concentration of base (or acid) is slowly added to a solution of acid (or base). Aqueous Equilibria © 2009, Prentice-Hall, Inc.

What is an equivalence point? A p. H meter or indicators are used to determine when the solution has reached the equivalence point, at which the stoichiometric amount of acid equals that of base. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

What happens to the p. H during a titration? Scenario: The molarity of sodium hydroxide if known, but hydrochloric acid is not known. Also note the strength of the acid and base. Initial p. H: Between the p. H and the equivalence point: The equivalence point: After the equivalence point: Aqueous Equilibria © 2009, Prentice-Hall, Inc.
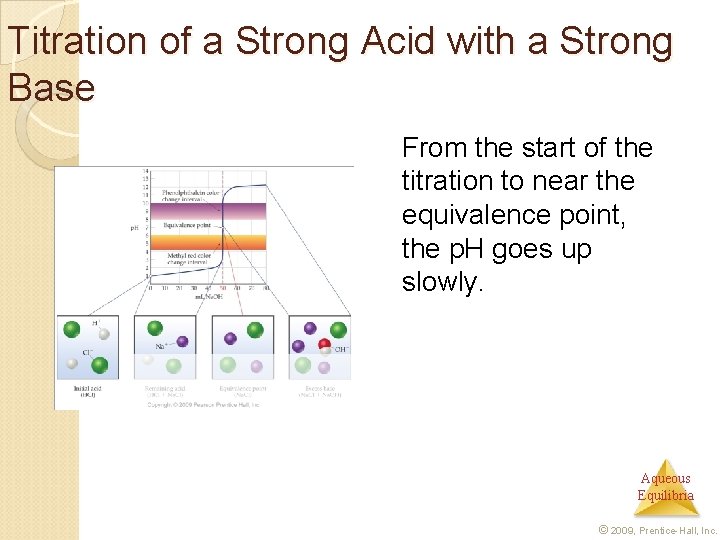
Titration of a Strong Acid with a Strong Base From the start of the titration to near the equivalence point, the p. H goes up slowly. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Titration of a Strong Acid with a Strong Base Just before (and after) the equivalence point, the p. H increases rapidly. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
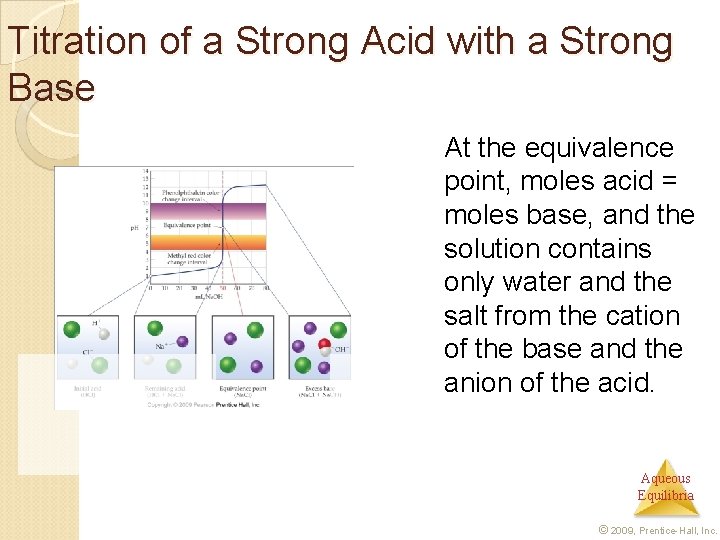
Titration of a Strong Acid with a Strong Base At the equivalence point, moles acid = moles base, and the solution contains only water and the salt from the cation of the base and the anion of the acid. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Titration of a Strong Acid with a Strong Base As more base is added, the increase in p. H again levels off. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

How do you think the titration of a weak acid and strong base differ? Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Titration of a Weak Acid with a Strong Base Unlike in the previous case, the conjugate base of the acid affects the p. H when it is formed. At the equivalence point the p. H is >7. Phenolphthalein is commonly used as an indicator in these titrations. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Titration of a Weak Acid with a Strong Base At each point below the equivalence point, the p. H of the solution during titration is determined from the amounts of the acid and its conjugate base present at that particular time. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
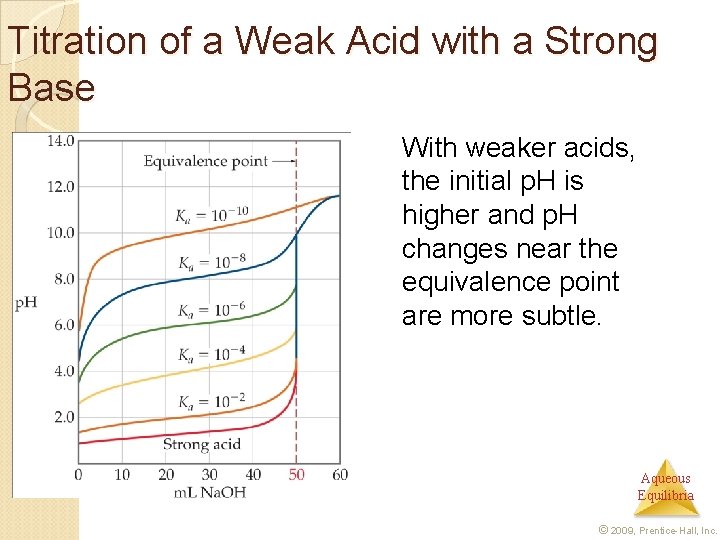
Titration of a Weak Acid with a Strong Base With weaker acids, the initial p. H is higher and p. H changes near the equivalence point are more subtle. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

How do you think the titration of a weak base and strong acid differ? Aqueous Equilibria © 2009, Prentice-Hall, Inc.
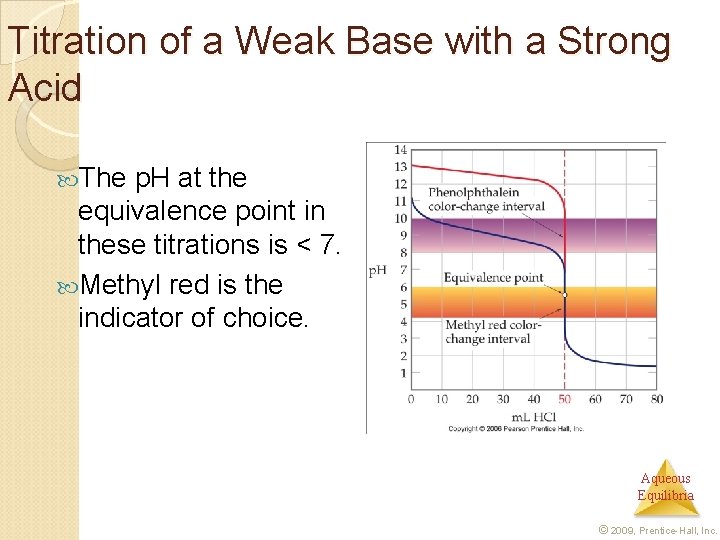
Titration of a Weak Base with a Strong Acid The p. H at the equivalence point in these titrations is < 7. Methyl red is the indicator of choice. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
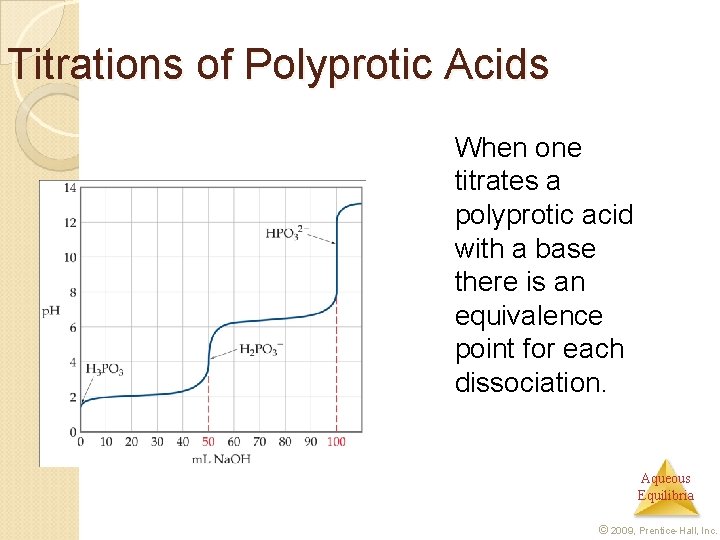
Titrations of Polyprotic Acids When one titrates a polyprotic acid with a base there is an equivalence point for each dissociation. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Solubility Products Consider the equilibrium that exists in a saturated solution of Ba. SO 4 in water: Ba. SO 4(s) Ba 2+(aq) + SO 42−(aq) Aqueous Equilibria © 2009, Prentice-Hall, Inc.
![Solubility Products The equilibrium constant expression for this equilibrium is Ksp = [Ba 2+] Solubility Products The equilibrium constant expression for this equilibrium is Ksp = [Ba 2+]](http://slidetodoc.com/presentation_image_h/1e1b7978993f8bb4447a0d16d1f5251a/image-39.jpg)
Solubility Products The equilibrium constant expression for this equilibrium is Ksp = [Ba 2+] [SO 42−] where the equilibrium constant, Ksp, is called the solubility product. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
Solubility Products Ksp is not the same as solubility. Solubility is generally expressed as the mass of solute dissolved in 1 L (g/L) or 100 m. L (g/m. L) of solution, or in mol/L (M). Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Factors Affecting Solubility The Common-Ion Effect ◦ If one of the ions in a solution equilibrium is already dissolved in the solution, the equilibrium will shift to the left and the solubility of the salt will decrease. Ba. SO 4(s) Ba 2+(aq) + SO 42−(aq) Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Factors Affecting Solubility p. H ◦ If a substance has a basic anion, it will be more soluble in an acidic solution. ◦ Substances with acidic cations are more soluble in basic solutions. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Factors Affecting Solubility Complex Ions ◦ Metal ions can act as Lewis acids and form complex ions with Lewis bases in the solvent. ◦ Discuss and review the complex ion reaction worksheet. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Factors Affecting Solubility Complex Ions ◦ The formation of these complex ions increases the solubility of these salts. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
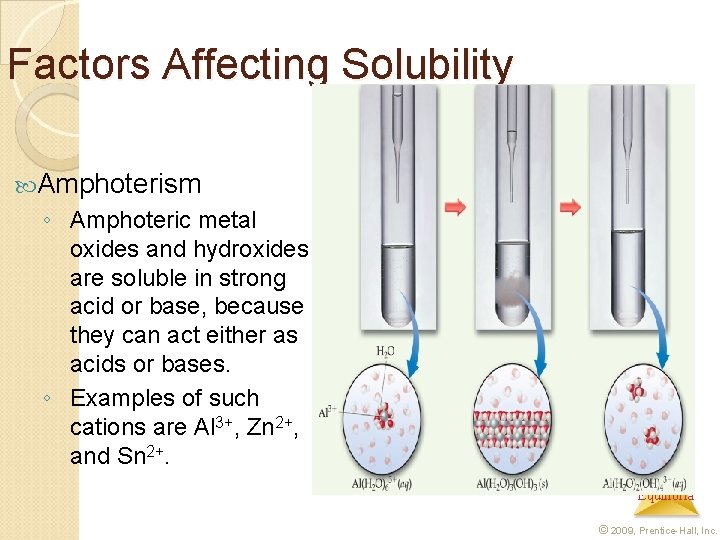
Factors Affecting Solubility Amphoterism ◦ Amphoteric metal oxides and hydroxides are soluble in strong acid or base, because they can act either as acids or bases. ◦ Examples of such cations are Al 3+, Zn 2+, and Sn 2+. Aqueous Equilibria © 2009, Prentice-Hall, Inc.

Will a Precipitate Form? In a solution, ◦ If Q = Ksp, the system is at equilibrium and the solution is saturated. ◦ If Q < Ksp, more solid can dissolve until Q = Ksp. ◦ If Q > Ksp, the salt will precipitate until Q = Ksp. Aqueous Equilibria © 2009, Prentice-Hall, Inc.
- Slides: 46