Solution Stoichiometry Reactions in Aqueous Solution Double Replacement

Solution Stoichiometry

Reactions in Aqueous Solution Double Replacement: (precipitate) 2 KI(aq) + Pb(NO 3)2(aq) 2 KNO 3(aq) + Pb. I 2(s) Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq) Single Replacement:
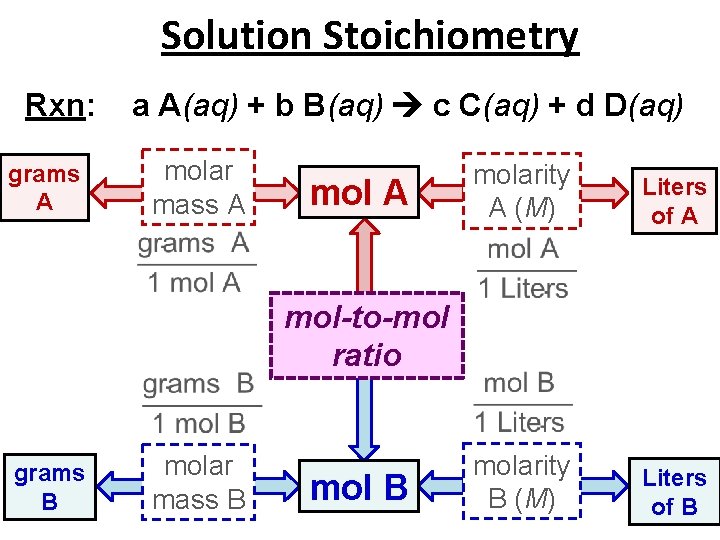
Solution Stoichiometry Rxn: grams A a A(aq) + b B(aq) c C(aq) + d D(aq) molar mass A molarity A (M) Liters of A molarity B (M) Liters of B mol-to-mol ratio grams B molar mass B mol B

Example #1: Liters of A Liters of B Ammonia and phosphoric acid solutions are used to produce ammonium hydrogen phosphate fertilizer. What volume of 14. 8 M NH 3 is needed to react with 120. L of 12. 9 M of H 3 PO 4 ? 2 NH 3(aq) + H 3 PO 4(aq) (NH 4)2 HPO 4(aq) V=? L V = 120. L M = 14. 8 mol/L M = 12. 9 mol/L molarity B 1 L NH 3 12. 9 mol H 3 PO 4 2 mol NH 3 x x 120. L H 3 PO 4 x 1 L H 3 PO 4 1 mol H 3 PO 4 14. 8 mol NH 3 Liters molarity A 209 LNH NH = ____L of A 3 3

Example #2: Liters of A Molarity of B A 159 m. L sample of KOH solution reacts completely with 100. m. L of 1. 50 M H 2 SO 4. Calculate the molarity of the KOH solution. H 2 SO 4(aq) + 2 KOH(aq) 2 H 2 O(l) + K 2 SO 4(aq) V = 0. 100 L V = 0. 159 L M = 1. 50 mol/L M = ? mol/L 0. 100 L H 2 SO 4 x Liters of A 1. 50 mol H 2 SO 4 1 L H 2 SO 4 molarity A 0. 300 x = 1 mol H 2 SO 4 mol KOH 2 mol KOH molarity B 0. 300 mol KOH 1. 89 M = _____ M KOH 0. 159 L KOH

Example #3: grams of A Liters of B Solid aluminum is added to an aqueous solution of zinc chloride. What volume of 7. 50 M Zn. Cl 2(aq) solution is needed to react completely with 54. 0 grams of Al(s) ? 2 Al(s) + 3 Zn. Cl 2(aq) 2 Al. Cl 3(aq) + 3 Zn(s) m = 54. 0 g V=? L M = 7. 50 mol/L molarity B 1 mol Al x 3 mol Zn. Cl 2 x 1 L Zn. Cl 2 54. 0 g Al x 26. 98 g Al 2 mol Al 7. 50 mol Zn. Cl 2 grams molar mass A 0. 400 L Zn. Cl = ____ of A 2 2

• Example #3 involves grams. To do a problem with moles you would replace the first two steps with the # of moles.

Example #4: Liters of A grams of B Adding 102 m. L of 0. 508 M Cu. Cl 2(aq) solution to an excess of Ag. NO 3(aq) forms solid Ag. Cl(s) precipitate. What is the maximum mass of Ag. Cl(s) precipitate? Cu. Cl 2(aq) + 2 Ag. NO 3(aq) Cu(NO 3)2(aq) + 2 Ag. Cl(s) V = 0. 102 L M = 0. 508 mol/L m=? g molar mass B 0. 508 mol Cu. Cl 2 x 2 mol Ag. Cl 143. 32 g Ag. Cl 0. 102 L Cu. Cl 2 x x 1 mol Ag. Cl 1 L Cu. Cl 2 1 mol Cu. Cl 2 Liters molarity A of A 14. 9 gg. Ag. Cl = ____

• Example #4 involves grams. To do a problem with moles you would omit the last step.
- Slides: 9