AP CHEMISTRY MIDTERM REVIEW TOPIC GASES Savannah Snyder


































- Slides: 37

AP CHEMISTRY MIDTERM REVIEW TOPIC: GASES Savannah Snyder Michael Kleber 4 th Period

Converting Pressures with Different Units of Measurement 1 atm = 760 mm. Hg = 760 torr (The unit in which you are converting FROM must cancel out) Example: Convert 30 atm to torr. 30 atm X 760 torr = 22800 torr 1 atm

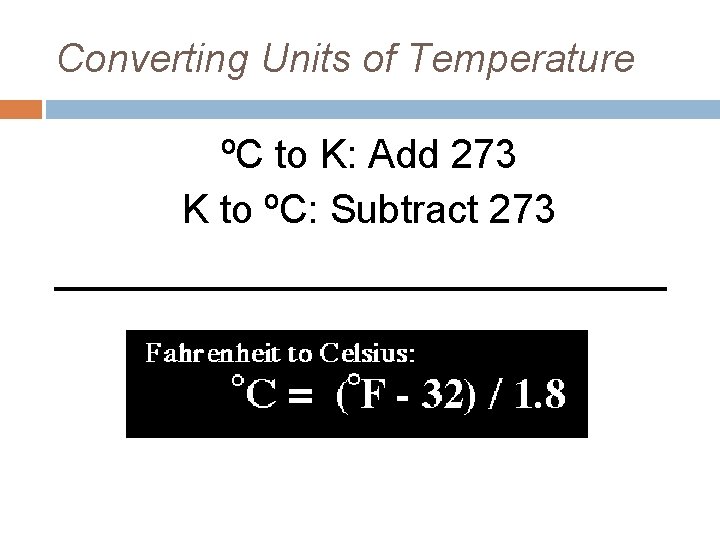
Converting Units of Temperature ºC to K: Add 273 K to ºC: Subtract 273 _____________

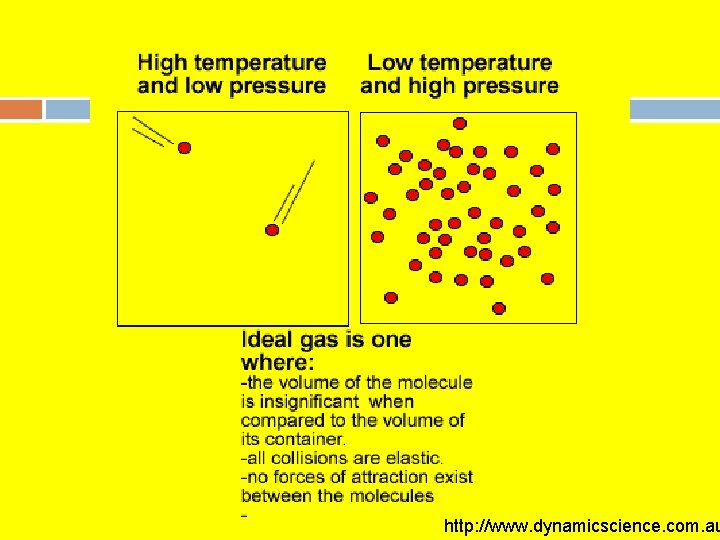
Differentiating between Ideal Gases and Non-Ideal Gases Basic points of the kinetic theory of gases (Ideal Gas) 1. The volume of gas molecules is negligible (not important) compared to the volume of space in which they move. 2. Gas molecules move in a straight line between frequent collisions with the walls of the container and themselves. 3. All collisions are elastic (no energy is lost). 4. No intermolecular forces are present. 5. The average kinetic energy of the gas molecules is directly proportional to the temperature.

http: //www. dynamicscience. com. au

Examples of Questions Pertaining to Ideal Gas. -When does a gas behave like a real gas? Low Temperature and High Pressure. -When would a gas behave like an ideal gas? High Temperature and Low Pressure

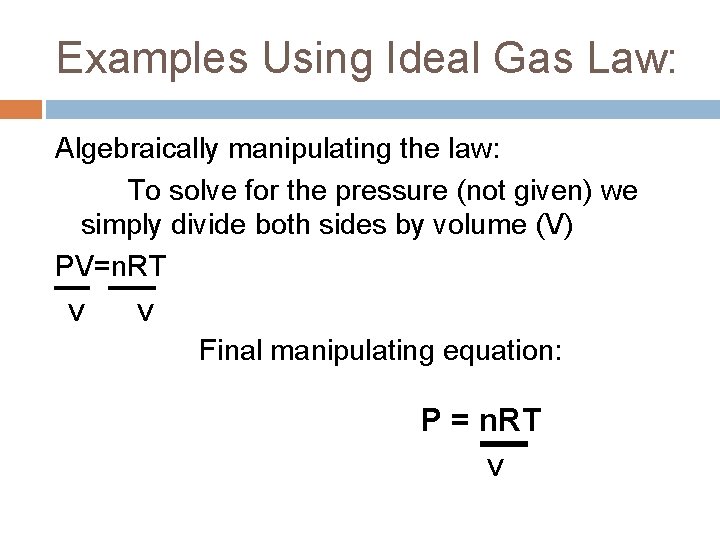
The Ideal Gas Law As we have learned from our previous year in Chemistry as well as reviewed this year, PV=n. RT R=. 0821 (gas constant) When solving for one specific component of the ideal gas law the equation can be algebraically manipulated to accommodate the equation’s need.

Examples Using Ideal Gas Law: Algebraically manipulating the law: To solve for the pressure (not given) we simply divide both sides by volume (V) PV=n. RT V V Final manipulating equation: P = n. RT V

REVIEW QUESTION CHECKPOINT 1 An 18 liter container holds 16. 00 grams of oxygen gas (O 2) at 45 °C. What is the pressure in the container? A. 11. 0 atm B. 23. 2 atm C. 1. 45 atm D. 0. 725 atm


Boyle’s Law This law describes the relationship between the volume and the pressure of a gas when the temperature and amount of moles are constant This equation is helpful when determining what a new pressure would be if the volume is decreased/increased (same for pressure)

REVIEW QUESTION CHECKPOINT 2 A balloon with a volume of 2. 0 L is filled with a gas at 3 atmospheres. If the pressure is reduced to 0. 5 atmospheres without a change in temperature, what would be the volume of the balloon?

The volume of the balloon will expand to 12 L.

Charles’s Law This law describes the volume and temperature relationship of a gas when the pressure and amount are constant.

REVIEW QUESTION CHECKPOINT 3 A 600 m. L sample of nitrogen is heated from 27 °C to 77 °C at constant pressure. What is the final volume?

The final volume after heating will be 700 m. L.

Gay-Lussac’s Law This law describes the relationship between the pressure of a gas and it’s temperature IF the volume and amount of moles are held constant. -As a gas is heated, particles move with greater kinetic energy causing the pressure to increase.

REVIEW QUESTION CHECKPOINT 4 A 20 L cylinder containing 6 atm of gas at 27 °C. What would the pressure of the gas be if the gas was heated to 77 °C?

The pressure will increase to 7 atm after heating the gas from 27 °C to 77 °C.

Combined Gas Law If the number of moles of gas are constant (No gas can get in or out) then we can combine the previous three gas laws.

REVIEW QUESTION CHECKPOINT 5 A cylinder contain a gas of volume 30 L, at a pressure of 340 mm. Hg and a temperature of 420 K. Find the temperature of the gas which has a volume 40. L at a pressure of 410 mm. Hg.

The temperature of the gas will be 670 K

Video of Gas Laws Experiments (6: 39) Ideal Gas Law Lecture (8: 45)

Graham’s Law of Effusion Defines the relationship of the speed of a gas diffusion and the gases’ molecular mass. The lighter the gas, the faster is its rate of effusion.

REVIEW QUESTION CHECKPOINT 6 How much faster does hydrogen escape through a porous container than sulfur dioxide? (Lighter gases will travel faster)

Hydrogen will escape 5. 66 times faster.

Root Mean Square Speed vrms is the root mean square of the speed Mm is the molar mass of the gas in grams per mole R is the molar gas constant T is the temperature in kelvin

REVIEW QUESTION CHECKPOINT 7 What is the root mean square speed of the molecules in a sample of oxygen gas weighing 30 k. G at 0 °C?

The speed of the oxygen gas will be 1. 497

Calculating Kinetic Energy: K. E. The equation K. E. is used when expressing the kinetic energy per mol of gas. The equation RT is used represent the average kinetic energy per molecule. -Questions on this topic are used heavily to trick students who over think. I. E. Will two gasses kinetic energies be equal or not equal if the mass of one is double the other.

REVIEW QUESTION CHECKPOINT 8 Two samples of gases are placed in identical containers. Both gases are at STP. The first gas weighs 32 g/mol and the second weighs 40 g/mol. Which gas will have a higher kinetic energy? The kinetic energies are equal.

Dalton’s Law of Partial Pressures Dalton’s Law states that in a mixture of gases the total pressure is simply the sum of the partial pressures. Where…. Calculating the mole fraction: XA= Moles A Total Moles To calculate the actual partial pressure (In atm): P 1= Ptotal X (XA)

REVIEW QUESTION CHECKPOINT 9 For a deep descent, a scuba diver uses a mixture of helium and oxygen with a total pressure of 8. 00 atm. If the oxygen has a partial pressure of 1280 mm. Hg, what is the partial pressure of the helium?

The partial pressure of helium is 4800 mm. Hg

Questions ?

Sources www. apcentral. collegeboard. com www. sciencegeek. net/APChemistry www. chemmybear. com 5 Steps to a 5 AP Chemistry, 2012 -2013 Edition (5 Steps to a 5 on the Advanced Placement Examinations Series) Richard H. Langley (Author), John Moore (Author)

Self Study: Free Response Question One