Dissolved Gases Important Gases 6 important gases are










































- Slides: 53

Dissolved Gases

Important Gases Ø Ø Ø Ø 6 important gases are dissolved in lakes, streams, seas Nitrogen Oxygen Carbon dioxide Methane Hydrogen sulfide Ammonia All have important functions, but differ in behavior, origin

Air Provides Some Gases Ø Atmosphere has enough nitrogen (78%), oxygen (21%), and carbon dioxide (0. 03%) to serve as primary source Ø Others present only in trace amounts in atmosphere

Other Gas Sources Methane - anaerobic breakdown of plants/animals Ø Hydrogen sulfide chemical/bacterial transformations Ø Ammonia breakdown of nitrogenous materials by bacteria, some animals Ø

How much gas is dissolved in water at any given time? Ø Dependent on several factors: Ø Solubility factor Ø Pressure Ø Temperature Ø Salinity

Solubility Factor Ø Not all gases dissolve in water to same extent Ø Some gases dissolve very easily in water, some dissolve very little

Pressure (atmosphere) Ø Amount of gas absorbed by water is proportional to its partial pressure in the atmosphere (conc. = solubility factor X partial pressure) Ø Altitude decreases saturation level by ~1. 4% per 100 m

Temperature Solubility of gas in water decreases as temperature rises Ø Generalization - cold water can hold more gas in solution than warm water Ø Nearly linear relationship within normal range of natural water temperatures Ø

Salinity Ø Presence of various minerals in solution lowers the solubility of gases Ø Generally disregarded in limnology because freshwaters have salinity near zero

Salinity Oceans (salinity of 3. 5%) have reduced gas saturation values of ~18 -20% Ø Saline pools/lakes can have much higher salinities (5 -6 X ocean values) Ø Important consideration here for gas solubilities Ø

Relative Saturation Relation between existing solubility (amount of gas present) and the equilibrium content expected at same temperature and partial pressure Ø Can be less, or more (supersaturation) Ø

Oxygen Abundant and dissolves readily in water Ø Needed for respiration by organisms and for complete breakdown of organic matter Ø Relatively easy to measure Ø

Oxygen 1/4 as abundant as nitrogen in atmosphere, but twice as soluble Ø Solubility of oxygen increases as temp. decreases, salinity decreases, and pressure increases Ø

Oxygen Ø Two sources for oxygen in lakes Ø Atmosphere Ø Photosynthesis

Atmosphere Ø Ø Diffusion across air-water interface and down into water column Years to reach depth of 5 m Wind-driven waves and currents distribute oxygen to lower levels Too much agitation can prevent water from becoming supersaturated

Photosynthesis Most oxygen in standing waters is byproduct of photosynthesis Ø Phytoplankton contribute most Ø Rooted macrophytes, attached algae, benthic algae mats are chief producers in shallow lakes, lake margins Ø

Loss of Oxygen Ø Physical - change in temperature, pressure Ø Biological - most important respiration by plants, animals, bacteria (decay processes) Ø Other - methane bubbles rising from sediments through water column

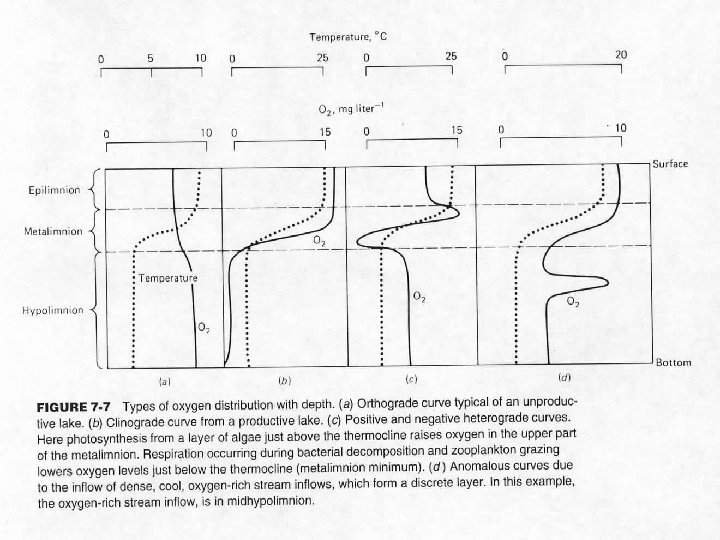
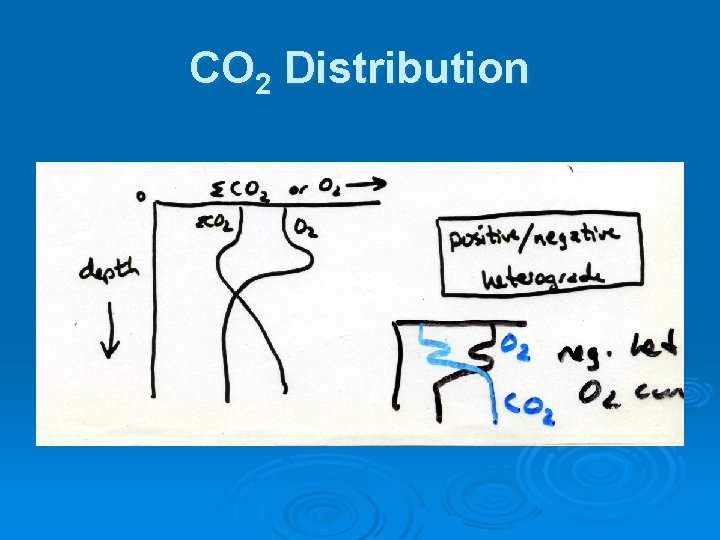
Oxygen Distribution Ø Distribution changes as lake goes through seasonal temperature cycle Ø Orthograde distribution during spring, fall turnovers in dimictic lake Ø Clinograde distribution during thermal stratification


Another distribution Ø Extreme clinograde - permanently meromictic lakes, anaerobic hypolimnion

Daily, seasonal variation in oxygen concentrations Ø The more plant material in a lake or pond, the more prone that system is to both daily and seasonal variations in dissolved oxygen content

Daily variation in oxygen concentrations Ø O 2 rises during day, declines at night Ø The greater the plant biomass, the greater the magnitude of the cycle

Daily variation in oxygen concentrations

Seasonal variation in oxygen concentrations Ø O 2 high during summer growing season, low in late-summer when plants die Ø May produce anoxia and die-offs of animals (summerkill)

Seasonal variation in oxygen concentrations Ø O 2 also may be low during winter in icecovered lakes Ø Reduced light transmission, respiration only - Winterkill of animals

Carbon Dioxide Ø CO 2 increasing in concentration in atmosphere Ø High solubility - 200 X > O 2 Ø Follows solubility laws (pressure, temp. ) Ø Many sources other than atmosphere: rainwater, runoff, groundwater, respiration, decomposition in sediments

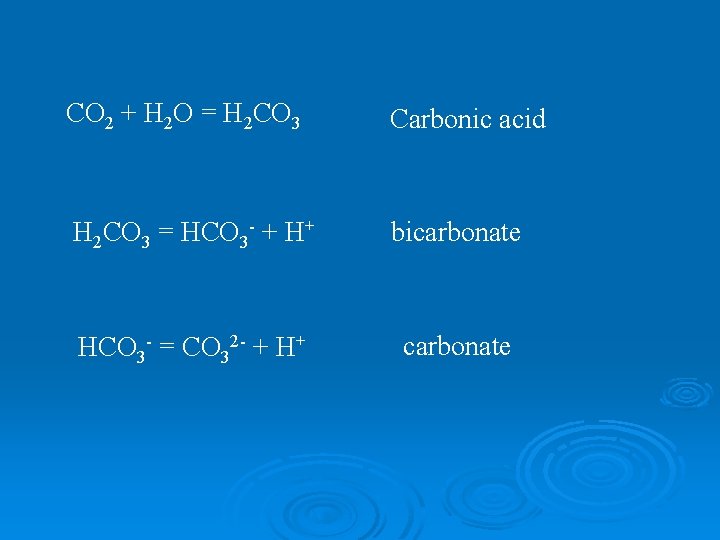
Carbon Dioxide Ø CO 2 behaves much differently than other gases once it dissolves in water Ø Exists in equilibrium with many additional forms of carbon

CO 2 + H 2 O = H 2 CO 3 Carbonic acid H 2 CO 3 = HCO 3 - + H+ bicarbonate HCO 3 - = CO 32 - + H+ carbonate

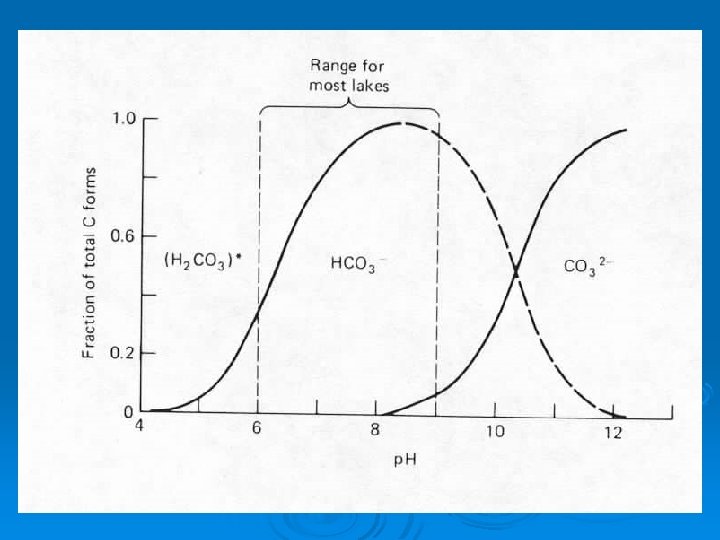
Putting it all together CO 2 + H 2 O = H 2 CO 3 = HCO 3 - + H+ = CO 32 - + 2 H+ Sensitive to changes in p. H Low p. H - left side dominates High p. H - right side dominates


Putting it all together CO 2 + H 2 O = H 2 CO 3 = HCO 3 - + H+ = CO 32 - + 2 H+ Addition of CO 2 via respiration pushes equilibrium to right and lowers p. H Removal of CO 2 via photosynthesis pulls equilibrium to left and raises p. H

In most natural lakes, CO 2 combines with alkali metals or alkaline earth metals to form carbonates, bicarbonates Ca. CO 3 + H 2 CO 3 = Ca (HCO 3)2 = Ca. CO 3 + H 2 O + CO 2 marl Free CO 2 Aggressive CO 2 dissolves Ca. CO 3 and drives equation to left Photosynthesis pulls equation to the right

Buffer System CO 2 + H 2 O = H 2 CO 3 = HCO 3 - + H+ = CO 32 - + 2 H+ Ca. CO 3 + H 2 CO 3 = Ca (HCO 3)2 = Ca. CO 3 + H 2 O + CO 2 Little change in p. H despite additions of lots of acids or base, as long as supply of carbonates & bicarbonates holds out

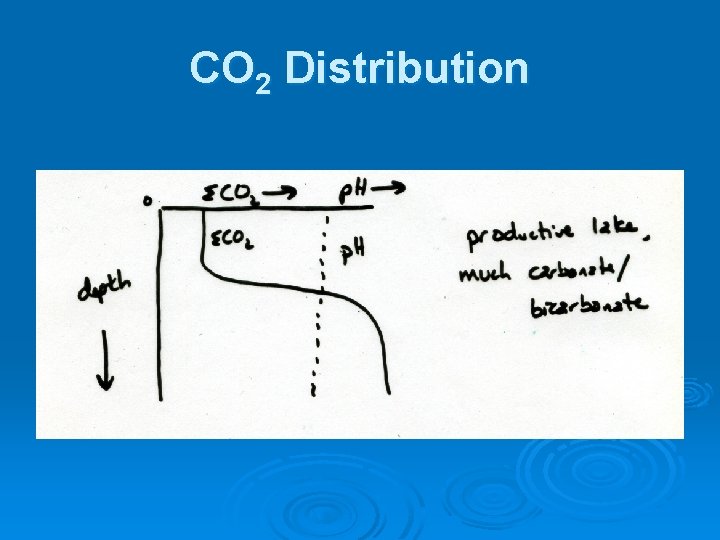
CO 2 Distribution

CO 2 Distribution

CO 2 Distribution

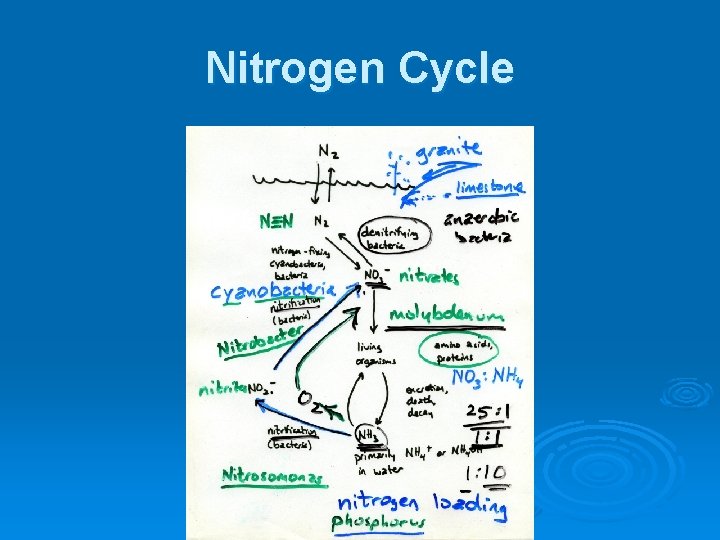
Nitrogen Ø Exists in many different forms in natural freshwater systems Ø A major nutrient that affects the productivity of aquatic systems

Nitrogen Dissolved gas - N 2 Ø Ammonia - NH 3 NH 4+ Ø Nitrite - NO 2Ø Nitrate - NO 3Ø Dissolved organics Ø l l l Amino acids Polypeptides Proteins Ø Sources: atmosphere, rain, runoff, groundwater ** Ø Losses: water outflow, adsorption to sediments, dinitrification by bacteria

Nitrogen Cycle

Ammonia Readily assimilated by plants Ø Nitrification by bacteria Ø Present in low concentrations in oxygenated waters Ø

Ammonia Accumulates in hypolimnion Ø No photosynthesis or nitrification Ø Release from sediments during anoxia Ø

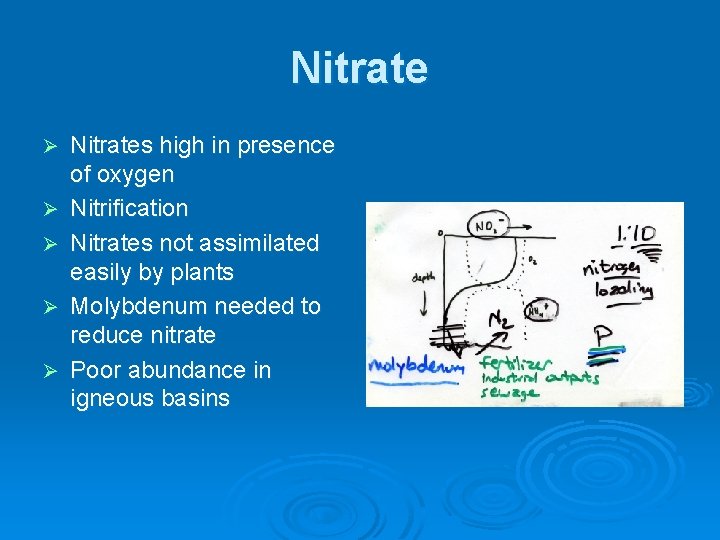
Nitrate Ø Ø Ø Nitrates high in presence of oxygen Nitrification Nitrates not assimilated easily by plants Molybdenum needed to reduce nitrate Poor abundance in igneous basins

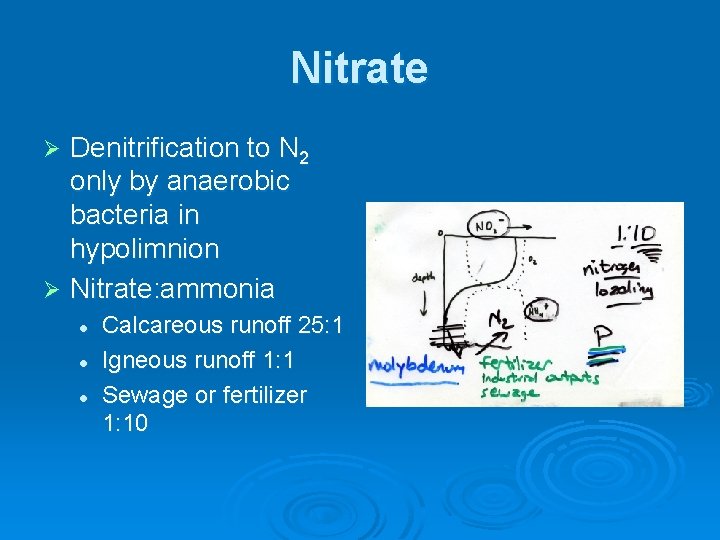
Nitrate Denitrification to N 2 only by anaerobic bacteria in hypolimnion Ø Nitrate: ammonia Ø l l l Calcareous runoff 25: 1 Igneous runoff 1: 1 Sewage or fertilizer 1: 10

Nitrate Ø Nitrogen loading by itself often does little to change lake productivity Ø Phosphorus more often limiting for plant growth than nitrogen

Phosphorus Ø Total concentrations in unpolluted waters 0. 01 -0. 05 mg/L Ø Sources: l l l Rainfall (unpolluted <0. 03 mg/L) (polluted >0. 1 mg/L) Groundwater ~0. 02 mg/L Surface runoff - variable - often major contributor to lakes (especially with pollutants)

Phosphorus Ø >90% of P in water is in form of organic phosphates or related materials in living things or their secretions Ø Great scarcity - limiting factor Ø Rapid turnover of organic P between living organisms l l Bacteria, phytoplankton, zooplankton, others 5 -100 minutes, more rapid under deficiency

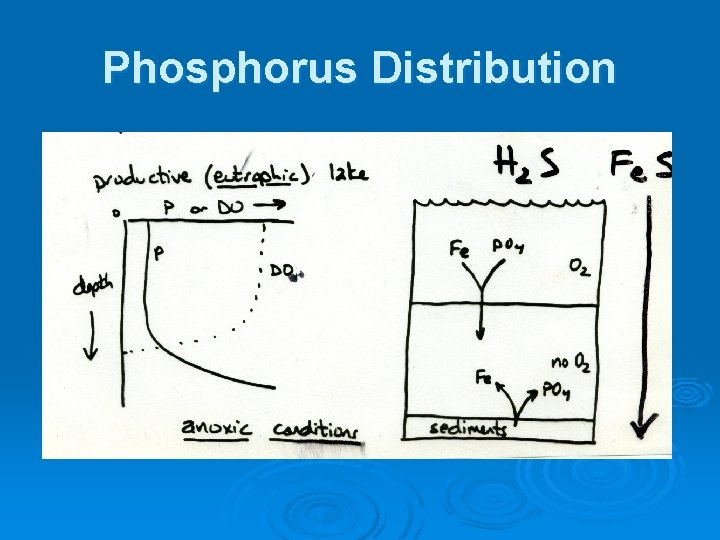
Phosphorus Ø In presence of O 2, various forms of phosphates form complexes, chelates, and insoluble salts with several metal ions l l E. g. , calcium and iron Induce precipitation of P in oxygenated waters

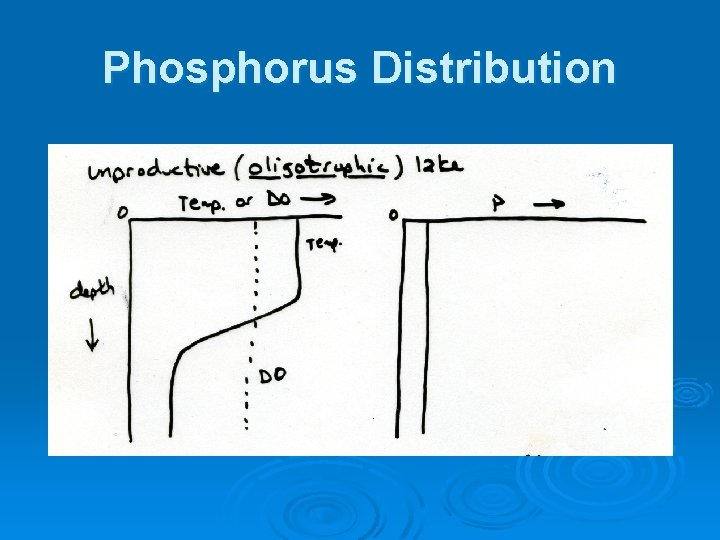
Phosphorus Distribution

Phosphorus Distribution

Confusing, interrelated terms Ø Alkalinity Ø Hardness Ø Salinity

Alkalinity Ø Measure of buffering capacity of water Ø Carbonates and bicarbonates of alkali metals

Hardness Ø Calcium and magnesium salt content Ø Temporary hardness - carbonates and bicarbonates, can be removed by boiling l l Precipitation of Ca. CO 3 Ca(HCO 3)2 = Ca. CO 3 + H 2 O + CO 2 Ø Permanent hardness - sulfates, chlorides, other anions

Salinity Ø Concentrations of Ca 2+ Mg 2+ Na+ K+ and HCO 3 - CO 32 - SO 42 - ClØ Plus other ionized components of other elements