Ch 12 Behavior of Gases Gases Gases expand

Ch. 12 Behavior of Gases

Gases • Gases expand to fill its container, unlike solids or liquids • Easily compressible: measure of how much the volume of matter decreases under pressure

Variables that describe a gas • Pressure (P) – Measured in kilopascals, k. Pa – Pressure and number of molecules are directly related v increase molecules = increase pressure – Gases naturally move from areas of high pressure to low pressure, due to the available space to move into

Variables that describe a gas • Volume (V) – Measured in Liters, L – Volume and pressure are inversely related • As volume decreases, the pressure increases • Smaller container = less room for movement, therefore molecules hit sides of container more often

Variables that describe a gas • Temperature (T) – Measured in Kelvin, K – The temperature and pressure are directly related • Increase in temp = increase in pressure • Volume must be held constant • Molecules hit the walls harder (due to increase in K. E. ) and more frequently. v. Think about a tire in hot weather…

Variables that describe a gas • Amount – Measured in moles, mol – Moles and pressure are directly related • Increase in # of moles = increase in pressure Ex: Inflating a balloon is adding more molecules. • Temperature must remain constant

Gas Laws • Describe how gases behave • Change can be calculated • Know the math and theory!!

Boyle’s Law (1662) • Gas pressure is inversely related to volume (as volume increases, pressure decreases) • Temperature is constant P 1 V 1= P 2 V 2

Ex: The pressure of a 2. 5 L of gas changes from 105 k. Pa to 40. 5 k. Pa. What will be the new volume?

Charles’s Law (1787) •

Ex: A sample of Nitrogen occupies a volume of 250 m. L at 25 o. C. What volume will the gas occupy at 95 o. C?

Gay-Lussac’s Law (1802) •

Ex: A gas has a pressure of 710 k. Pa at 227 o. C. What will the pressure be at 27 o. C, if the volume does not change?

Combined Gas Law •

Ex: 3. 0 L of Hydrogen gas has a pressure of 1. 5 atm at 20 o. C. What would the volume be if the pressure increased to 2. 5 atm at 30 o. C?

Ideal Gas Law •

Ideal Gas Law • A gas behaves “ideally” if it conforms to the gas laws – Gases do not usually do this – Real gases only behave this way at: 1. High temps (molecules move fast) 2. Low pressure (molecules are far apart) • This is because gases will stay a gas under these conditions – Molecules are not next to each other very long so attractive forces can’t play a role b/c molecules are moving too fast – Ideal Gases do no exist because: 1. Molecules do take up space 2. There attractive forces between molecules otherwise no liquid would form. (Molecules slow down to become liquids)
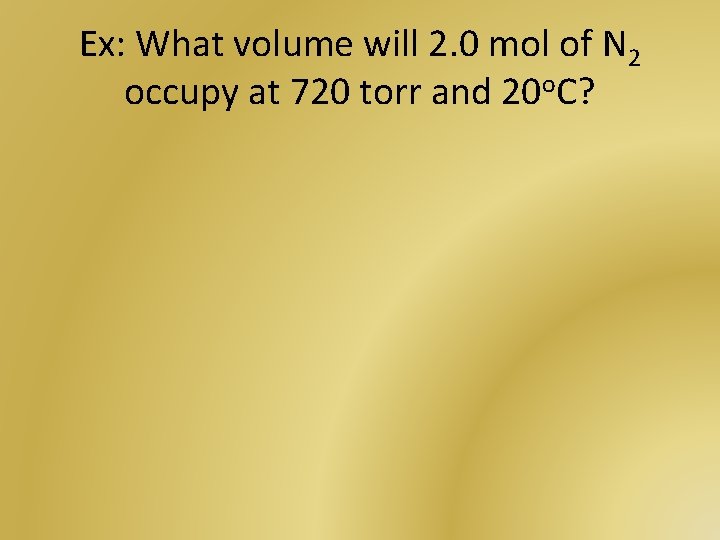
Ex: What volume will 2. 0 mol of N 2 occupy at 720 torr and 20 o. C?

Dalton’s Law of Partial Pressures • Used for mixture of gases in a container • If you know the P exerted by each gas in a mixture, you can calculate the total gas pressure • It is particularly useful in calculating pressure of gases collected over water. Ptotal = P 1 + P 2 + P 3… *P 1 represents the “partial pressure” or the contribution by the gas

Ex: Helium, Nitrogen, and Oxygen exist in a container. Calculate the total pressure of the mixture for the following partial pressures: He = 200 k. Pa N= 500 k. Pa O= 400 k. Pa

Graham’s Law of Effusion •

- Slides: 22