BEHAVIOR OF GASES Gases have weight Gases take

BEHAVIOR OF GASES • Gases have weight • Gases take up space • Gases exert pressure • Gases fill their containers • Gases are mostly empty space (the molecules in a gas are separate, very small, and very far apart) Gases doing all of these things!
Kinetic Theory of Gases The basic assumptions of the kinetic molecular theory are: § Gases are mostly empty space § The molecules in a gas are separate, very small and very far apart

Kinetic Theory of Gases The basic assumptions of the kinetic molecular theory are: § Gas molecules are in constant, chaotic motion § Collisions between gas molecules are elastic (there is no energy gain or loss)
Kinetic Theory of Gases The basic assumptions of the kinetic molecular theory are: § The average kinetic energy of gas molecules is directly proportional to the absolute temperature § Gas pressure is caused by collisions of molecules with the walls of the container

Measurements of Gases § To describe a gas, its volume, amount, temperature, and pressure are measured. • Volume: measured in L, m. L, cm 3 (1 m. L = 1 cm 3) • Amount: measured in moles (mol), grams (g) • Temperature: measured in KELVIN (K) • K = ºC + 273 • Pressure: measured in mm Hg, torr, atm, etc. • P = F / A (force per unit area)
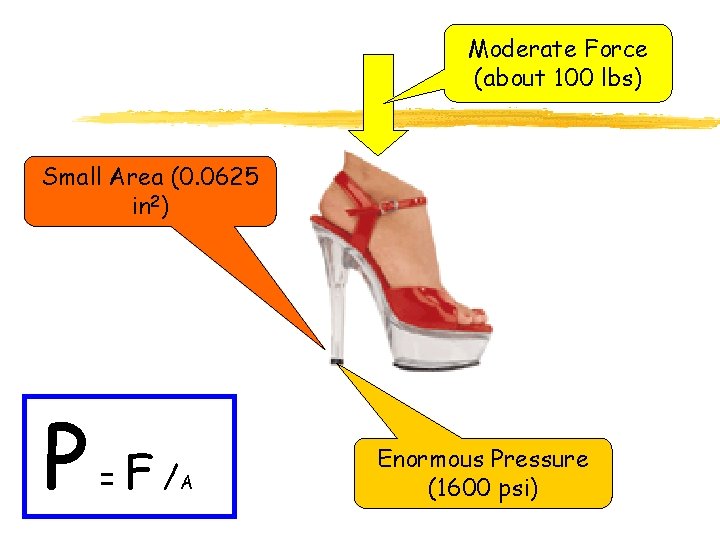
Moderate Force (about 100 lbs) Small Area (0. 0625 in 2) P = F /A Enormous Pressure (1600 psi)
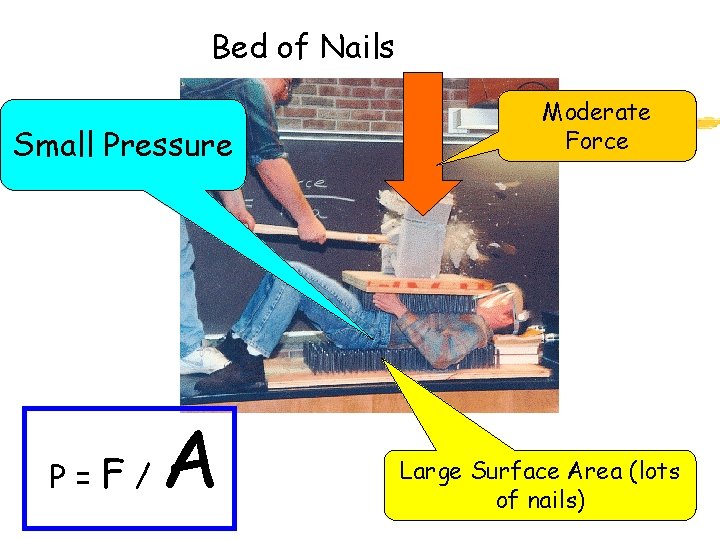
Bed of Nails Small Pressure P= F/ A Moderate Force Large Surface Area (lots of nails)

Units of Pressure § Units of Pressure: § § § 1 atm = 760 mm Hg 1 atm = 760 torr 1 atm = 1. 013 x 105 Pa 1 atm = 101. 3 k. Pa 1 atm = 1. 013 bar

Boyle’s Law For a given number of molecules of gas at a constant temperature, the volume of the gas varies inversely with the pressure. As P , V (when T and n are constant) and vice versa…. INVERSE RELATIONSHIP V 1/P P 1 V 1 = P 2 V 2

Example: A sample of gas occupies 12 L under a pressure of 1. 2 atm. What would its volume be if the pressure were increased to 3. 6 atm? (assume temp is constant) § P 1 V 1 = P 2 V 2 § (1. 2 atm)(12 L) = (3. 6 atm)V 2 § V 2 = 4. 0 L

Charles’ Law Jacques Charles (1746 -1828) The volume of a given number of molecules is directly proportional to the Kelvin temperature. As T , V (when P and n are constant) and vice versa…. DIRECT RELATIONSHIP V T

Example: A sample of nitrogen gas occupies 117 m. L at 100. °C. At what temperature would it occupy 234 m. L if the pressure does not change? (express answer in K and °C) § V 1 / T 1= V 2 / T 2 § (117 m. L) / (373 K) = (234 m. L) / T 2 § T 2 = 746 K § T 2 = 473 ºC

Combined gas law Combining Boyle’s law (pressure-volume) with Charles’ Law (volume-temp): This is for one gas undergoing changing conditions of temp, pressure, and volume.

Example 1: A sample of neon gas occupies 105 L at 27°C under a pressure of 985 torr. What volume would it occupy at standard conditions? § P 1 = 985 torr § V 1 = 105 L § T 1 = 27 °C = 300. K § P 2 = 1 atm = 760 torr § V 2 = ? § T 2 = 0 °C = 273 K P 1 V 1 T 2 = P 2 V 2 T 1 (985 torr)(105 L)(273 K) = (760 torr)(V 2)(300 K) V 2= 124 L

Example 2: A sample of gas occupies 10. 0 L at 240°C under a pressure of 80. 0 k. Pa. At what temperature would the gas occupy 20. 0 L if we increased the pressure to 107 k. Pa? § P 1 = 80. 0 k. Pa § V 1 = 10. 0 L § T 1 = 240 °C = 513 K § P 2 = 107 k. Pa § V 2 = 20. 0 L § T 2 = ? P 1 V 1 T 2 = P 2 V 2 T 1 (80. 0 k. Pa)(10. 0 L)(T 2) = (107 k. Pa)(20. 0 L)(513 K) T 2= 1372 K≈ 1370 K

Example 3: A sample of oxygen gas occupies 23. 5 L at 22. 2 °C and 1. 3 atm. At what pressure (in mm Hg) would the gas occupy 11. 6 L if the temperature were lowered to 12. 5 °C? § P 1 = 1. 3 atm § V 1 = 23. 5 L § T 1 = 22. 2 °C = 295. 2 K § P 2 = ? § V 2 = 11. 6 L § T 2 = 12. 5 °C = 285. 5 K P 1 V 1 T 2 = P 2 V 2 T 1 P 2= P 1 V 1 T 2/V 2 T 1 P 2=(1. 3 atm x (760 mm Hg/1 atm))(23. 5 L)(285. 5 K) (11. 6 L)(295. 2 K) = P 2= 1936 mm Hg ≈ 1900 mm. Hg

Gases: Standard Molar Volume & The Ideal Gas Law § Avogadro’s Law: at the same temperature and pressure, equal volumes of all gases contain the same # of molecules (& moles). § Standard molar volume = 22. 4 L @STP § This is true of “ideal” gases at reasonable temperatures and pressures , the behavior of many “real” gases is nearly ideal.

The IDEAL GAS LAW § Shows the relationship among the pressure, volume, temp. and # moles in a sample of gas. § P = pressure (atm) The units of R depend on the units used for P, § V = volume (L) V&T § n = # moles § T = temp (K) § R = universal gas constant = 0. 0821

Example 1: What volume would 50. 0 g of ethane, C 2 H 6, occupy at 140 ºC under a pressure of 1820 torr? § § P = (1820 torr)(1 atm/760 torr) = 2. 39 atm V=? n = (50. 0 g)(1 mol / 30. 08 g) = 1. 66 mol T = 140 °C + 273 = 413 K PV = n. RT V = n. RT/P V = (1. 66 mol) (0. 0821 L·atm/mol·K)(413 K) (2. 39 atm) V = 23. 6 L

Example 2: Calculate (a) the # moles in, and (b) the mass of an 8. 96 L sample of methane, CH 4, measured at standard conditions. (a) § § P = 1. 00 atm V = 8. 96 L n=? T = 273 K PV = n. RT n = PV/RT n = (1 atm)(8. 96 L)/(0. 0821 L·atm/mol·K)(273 K) n = 0. 400 mol

Example 2: Calculate (a) the # moles in, and (b) the mass of an 8. 96 L sample of methane, CH 4, measured at standard conditions. (a) § Or the easier way…

Example 2: Calculate (a) the # moles in, and (b) the mass of an 8. 96 L sample of methane, CH 4, measured at standard conditions. (b) § Convert moles to grams…

Example 3: Calculate the pressure exerted by 50. 0 g ethane, C 2 H 6, in a 25. 0 L container at 25 ºC? § § P=? V = 25. 0 L n = (50. 0 g)(1 mol / 30. 08 g) T = 25 °C + 273 = 298 K PV = n. RT P = n. RT/V P= (1. 66 mol)(0. 0821 L·atm/mol·K)(298 K) (25. 0 L) P = 1. 62 atm

Warm-up: z Identify which law (name and equation) that you would use for each of the following problems: z A sample of gas occupies 10. 0 L at 240°C under a pressure of 80. 0 k. Pa. At what temperature would the gas occupy 20. 0 L if we increased the pressure to 107 k. Pa? z What volume would 50. 0 g of ethane, C 2 H 6, occupy at 140 ºC under a pressure of 1820 torr? z A sample of gas occupies 12 L under a pressure of 1. 2 atm. What would its volume be if the pressure were increased to 3. 6 atm? (assume temp is constant) z A sample of neon gas occupies 105 L at 27°C under a pressure of 985 torr. What volume would it occupy at standard conditions? z A sample of nitrogen gas occupies 117 m. L at 100. °C. At what temperature would it occupy 234 m. L if the pressure does not change? (express answer in K and °C)

z. DALTON’S LAW OF PARTIAL PRESSURES

Partial Pressures and Mole Fractions § In a mixture of gases each gas exerts the pressure it would exert if it occupied the volume alone. § The total pressure exerted by a mixture of gases is the sum of the partial pressures of the individual gases: § Ptotal = P 1 + P 2 + P 3 + …

Example: If 100. 0 m. L of hydrogen gas, measured at 25 C and 3. 00 atm, and 100. 0 m. L of oxygen, measured at 25 C and 2. 00 atm, what sould be the pressure of the mixture of gases? Notice the two gases are measured at the same temp. and vol. § Ptotal = P 1 + P 2 + P 3 + … § PT = 3. 00 atm + 2. 00 atm § PT = 5. 00 atm

Vapor Pressure of a Liquid § The pressure exerted by its gaseous molecules in equilibrium with the liquid; increases with temperature
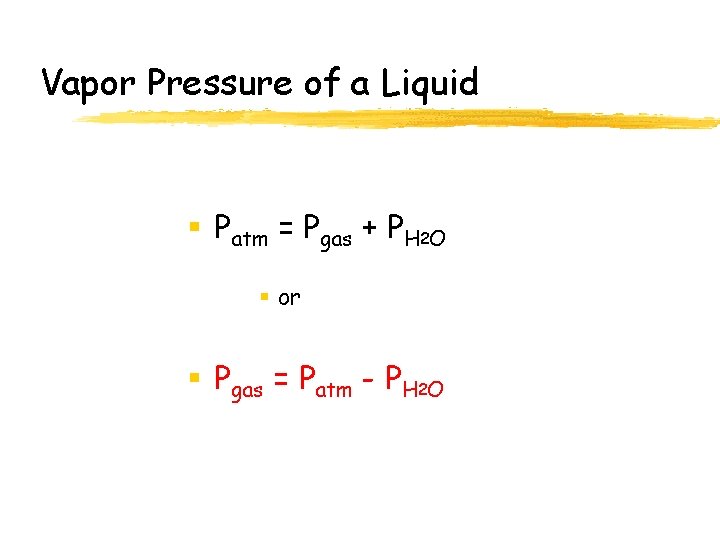
Vapor Pressure of a Liquid § Patm = Pgas + PH 2 O § or § Pgas = Patm - PH 2 O
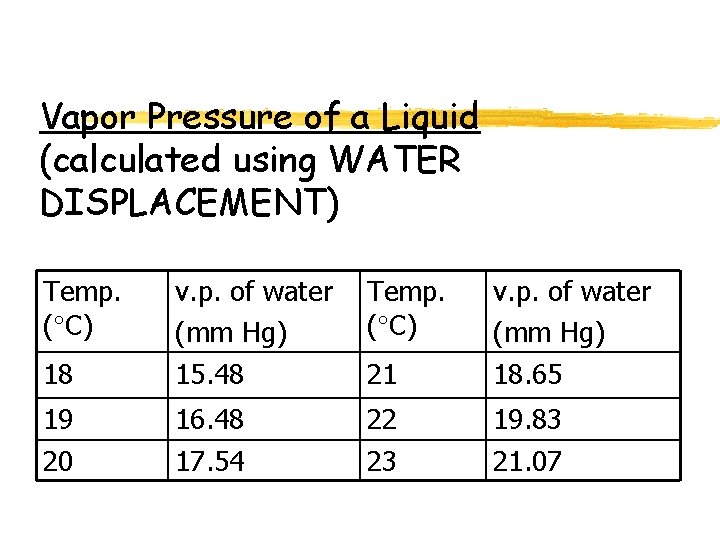
Vapor Pressure of a Liquid (calculated using WATER DISPLACEMENT) Temp. ( C) 18 v. p. of water (mm Hg) 15. 48 21 v. p. of water (mm Hg) 18. 65 19 20 16. 48 17. 54 22 23 19. 83 21. 07

Example 1: A sample of hydrogen gas was collected by displacement of water at 25 C (vapor pressure of water at 25 C is 23. 76 mm Hg). The atmospheric pressure was 748 mm Hg. What pressure would the dry hydrogen exert in the same conditions? § § PH 2 = Patm - PH 2 O PH 2 =748 mm Hg – 23. 76 mm Hg PH 2 = 724. 24 mm Hg PH 2 724 mm Hg

Example 2: A sample of oxygen gas was collected by displacement of water. The oxygen occupied 742 m. L at 27 C (the vapor pressure of water at 27 C is 26. 74 mm Hg). The atmospheric pressure was 753 mm Hg. What volume would the dry oxygen occupy at STP? § PO 2 = Patm - PH 2 O § PO 2 =753 mm Hg – 26. 74 mm Hg § PO 2 = 726 mm Hg § P 1 V 1 T 2 = P 2 V 2 T 1 V 2 = P 1 V 1 T 2/P 2 T 1 § V 2 = (726 mm Hg)(742 m. L)(273 K)/(760 mm Hg)(300. K) § V 2 = 645 m. L

Example 3: A student prepares a sample of hydrogen gas by electrolyzing water at 25 C (the vapor pressure of water at 25 C is 23. 76 mm Hg). She collects 152 m. L of H 2 at a total pressure of 758 mm Hg. Calculate: (a) the partial pressure of hydrogen, and (b) the number of moles of hydrogen collected. § PH 2 = Patm - PH 2 O § PH 2 =758 mm Hg – 23. 76 mm Hg § PH 2 = 734 mm Hg

Example 3: A student prepares a sample of hydrogen gas by electrolyzing water at 25 C. She collects 152 m. L of H 2 at a total pressure of 758 mm Hg. Calculate: (a) the partial pressure of hydrogen, and (b) the number of moles of hydrogen collected. PV = n. RT n = PV/RT n = (734 mm. Hg x(1 atm/760 mm. Hg))(0. 152 L) (0. 0821 L·atm/mol·K)(298 K) n = 0. 00600 mol H 2

Mole Fraction Moles of Substance x Total Pressure Total Moles = Partial Pressure of that substance

Graham’s Law of Diffusion & Effusion § Where, § Rate = rate of diffusion or effusion § MM=molar mass

WS: Graham’s Law z 1. Under the same conditions of temperature and pressure, how many times faster will hydrogen effuse compared to carbon dioxide?
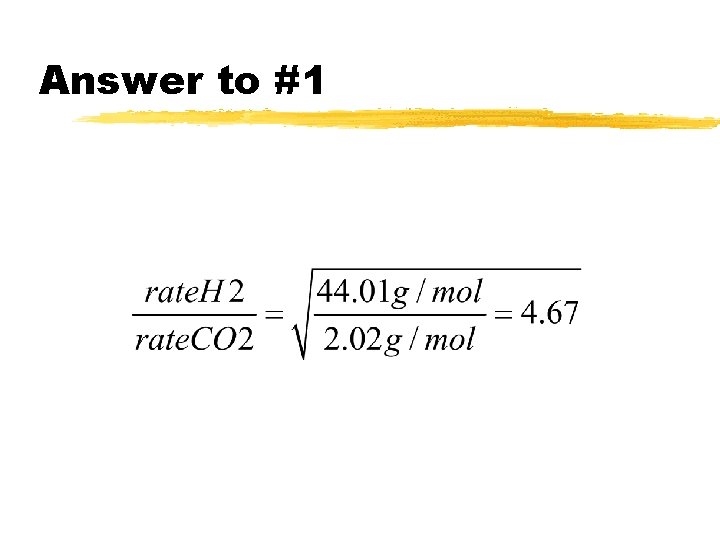
Answer to #1

Extra Question z. Rank the following gases in terms of fastest to slowest effusion: Kr, He, Rn, N 2
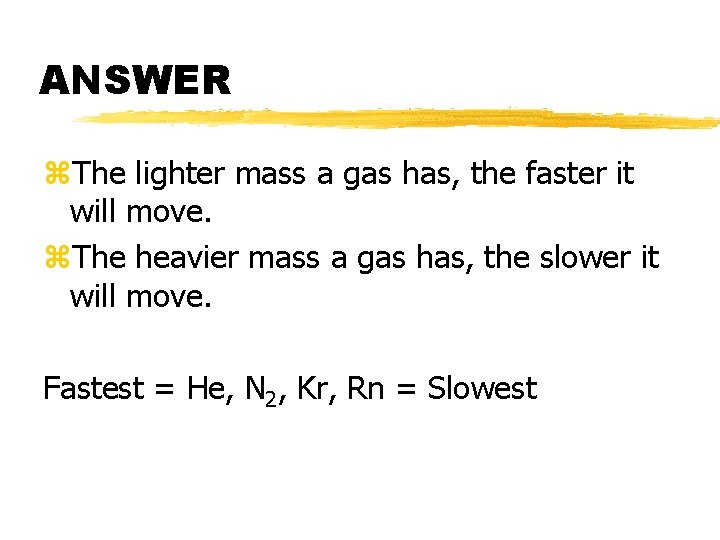
ANSWER z. The lighter mass a gas has, the faster it will move. z. The heavier mass a gas has, the slower it will move. Fastest = He, N 2, Kr, Rn = Slowest
- Slides: 40