Chemistry Midterm Review Question One n Describe the

Chemistry Midterm Review

Question One n Describe the atomic model for each scientist listed. Be sure to mention information about the experiment and the location of the subatomic particles. 1. 2. 3. 4. 5. Dalton Thomson Rutherford Bohr Modern View Answer

Answer 1 n n n Dalton – indivisible, solid sphere (no subatomic particles) Thomson- “plum pudding model” electrons are evenly distributed throughout a mass of protons. Model based on his cathode ray experiment Rutherford- discovered mostly empty space surrounding a dense positively charged nucleus using through the gold foil experiment Bohr- believed in a small positively charged nucleus containing the protons and neutrons surrounded by electrons in ORBITS of specific energy levels Modern View- Also called the wave- mechanical model and the quantum model similar to Bohr but electrons are in regions of high probability based on energy level, and orbital.

Question 2 n. Compare the charge, size, and location of a proton, neutron, and electron. Answer 2

Answer 2 Type of Size in subatomic amu particle Charge Location in an atom Proton 1 +1 nucleus Neutron 1 0 nucleus Electron 1/2000 -1 Electron clouds surrounding nucleus

Question 3 n Explain why the following two elements are isotopes. State the number of protons, electrons, and neutrons each isotope possesses.

Answer 3 n n n They are isotopes because each have a different # of neutrons but the same number of protons and electrons. Sn -121 has 50 p+, 50 e-, 71 n 0 Sn- 119 has 50 p+, 50 e-, 69 n 0

Question 4 n. List the seven metalloids

Answer 4 B Si, Ge, As Sb, Te At

Question 5 n Why is the formation of a solution a physical change while the ability to react with a chemical considered a chemical change?

Answer 5 n n “reacting” indicates a change in the physical properties of an element resulting in a new compound. Formation of a solution is a physical property because a new compound has not been formed. Evaporation of the water would leave the original compound behind. (ex. salt dissolving in water)

Question 6 n. List Properties of a metal and a nonmetal.

Answer 6 n Metals n n n n Form cations Shiny Malleable Ductile Good conductors Low electronegativites and IE Solids except Hg n Nonmetals Brittle n Dull n Poor conductors n High electronegativities and IE n

Question 7 n n n A solid substance was tested in the laboratory. The test results are listed below. • dissolves in water • is an electrolyte • melts at a high temperature A)Na B) Na. Cl C) Cl D) NH 3

Answer 7 n A)Na B) Na. Cl C) Cl D) NH 3

Question 8 n. What is the correct formula for Cobalt(III) bromide?
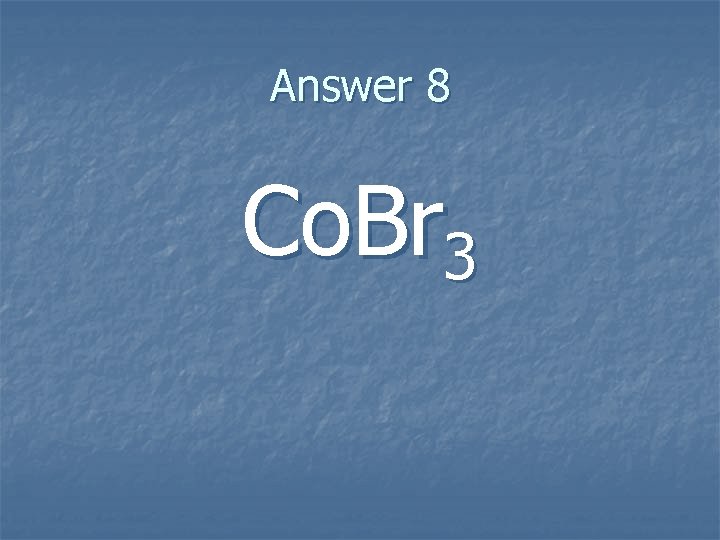
Answer 8 Co. Br 3

Question 9 n. What form of matter cannot be decomposed?

Answer 9 Element

Question 10 n What happens to a nonmetal ion when it becomes a negative ion? n. Mention difference in # of electrons n. Changes in radius

Answer 10 n. Gains electrons n. Radius increases

Question 11 n What is the name of the property that indicates an element’s desire to attract electrons and which element has the greatest attraction?

Answer 11 n. Electronegativit y n. Fluorine

Question 12 n What are the terms used to identify a pure substance? n What are the terms used to identify a mixture?

Answer 12 n. Element and compound n. Heterogeneous and Homogeneous

Question 13 n. What is the difference between an excited state electron and a ground state electron?

Answer 13 n n Ground state electrons are in the lowest energy state Excited state electrons have absorbed energy allowing them to jump to a higher energy level.

Question 14 n Which substance can be decomposed by chemical change? n n beryllium boron methanol magnesium

Answer 14 n Which substance can be decomposed by chemical change? n n beryllium boron methanol magnesium

Question 15 n What is the name for each type of phase change? 1. 2. 3. 4. 5. Solid liquid Liquid gas Gas liquid Liquid solid Solid gas

Answer 15 1. 2. 3. 4. 5. Melting Boiling or vaporization Condensation Freezing / solidification Sublimation

Question 16 n What is the volume of a liquid with a density of 1. 5 g/ml and a mass of 78 g?

Answer 16 n. V=m/D n 78 g/4. 5 g/m. L n=52 m. L

Question 17 n When a bond is formed, energy is (released/absorbed) n When a bond is broken, energy is (released/absorbed)

Answer 17 n When a bond is formed, energy is (released/absorbed) n When a bond is broken, energy is (released/absorbed)

Question 18 n What is the mass number of an atom which contains 28 protons, 28 electrons and 34 neutrons? n n 28 56 62 90

Answer 18 n What is the mass number of an atom which contains 28 protons, 28 electrons and 34 neutrons? n n 28 56 62 90

Question 19 n. What is the % composition by mass of carbon in carbon dioxide [gram-formula mass 44 grams/mole]?

Answer 19 27 % C

Question 20 n. Why does calcium have similar chemical properties to barium?

Answer 20 n. Both are Group 2 elements n. Both have the same number of valence electrons

Question 21 n 1. 2. 3. 4. 5. Which of the following terms indicate a chemical property and which indicate a physical property? Flatten Conduct Corrode Drawn into a wire React

Answer 21 n Chemical n. Physical n Corrode n react n. Flatten n. Drawn n. Conduct

Question 22 n. What is the charge on a I-127 nucleus?

Answer 22 +53

Question 23 Name the type of element as it appears in the order below. Ge K Ar Br

Answer 23 Ge K Ar Br Metalloid, metal, noble gas, nonmetal

Question 24 How many electrons are contained in an 2+ Cu ion?

Answer 24 27 electrons

Question 25 Which electron configuration represents the electrons of atom of sodium in the excited? 1. 2 -8 -1 2. 2 -8 -2 3. 2 -7 -3 4. 2 -7 -2

Answer 25 4. 2 -7 -2

Question 26 n. If an element is shiny and a good conductor, what type of element is it?

Answer 26 Metal

Question 27 Compare the number of subatomic particles that compose N-14 and F-19.

Answer 27 N-14 n F-19 n Protons 7 n Protons 9 n Electrons 7 n Electrons 9 n Neutrons 7 n Neutrons 10 F-19 has two protons, two more electrons, and three more protons than N-14 n

Question 28 Which of the following is a molecular formula? CH 4 C 3 H 6

Answer 28 C 3 H 6

Question 29 In what ratio, will a calcium ion combine with a phosphide ion?

Answer 29 Ca 3 P 2 3: 2

Question 30 n Two atoms with an electronegativity difference of 0. 4 form a bond that is n n ionic, because electrons are shared ionic, because electrons are transferred covalent, because electrons are shared covalent, because electrons are transferred

Answer 30 n Two atoms with an electronegativity difference of 0. 4 form a bond that is n n ionic, because electrons are shared ionic, because electrons are transferred covalent, because electrons are shared covalent, because electrons are transferred

Question 31 Describe the difference between a particle diagram of a solid, liquid, and gas.

Answer 31 n Solid particles are packed tightly n Liquids are close but have some space for movement n Gases are far apart

Question 32 Compare the atomic radius of Ca to Mg and Sr.

Answer 32 The atomic radius falls in the middle of Mg and Sr.

Question 33 n State the trend for each of the following properties across a period. n. Ionization Energy n. Electronegativity n. Atomic radius

Answer 33 n. IE and Electronegativity increases across the period n. Atomic Radius decreases

Question 34 n State the trend for each of the following properties down a group. n. Ionization Energy n. Electronegativity n. Atomic radius

Answer 34 n. IE and Electronegativity decreases across the period n. Atomic Radius increases

Question 35 n When the equation __C 8 H 16 + __O 2 → __CO 2 + __H 2 O is correctly balanced using the smallest whole number coefficient, the coefficient of O 2 is n n n 1 8 12

Answer 35 n When the equation __C 8 H 16 + __O 2 → __CO 2 + __H 2 O is correctly balanced using the smallest whole number coefficient, the coefficient of O 2 is n n n 1 8 12

Question 36 Is this a endothermic or exothermic reaction? C B 380 k 280 k 1 A 2 3

Question 36 n What is the gram formula mass of Li 2 SO 4? n n 54 g 55 g 110 g 206 g

Answer 36 n What is the gram formula mass of Li 2 SO 4? n n 54 g 55 g 110 g 206 g

Question 37 n What is the total mass in grams of 0. 75 mole of SO 2? n n 16 g 24 g 32 g 48 g

Answer 37 n What is the total mass in grams of 0. 75 mole of SO 2? n n 16 g 24 g 32 g 48 g

Question 38 n. Calculate the number moles in 48 grams of CO.

Answer 38 1. 7 moles

Question 39 n Which electron-dot formula represents a polar molecule?

Answer 39

Question 40 What is the relationship between Celsius and Kelvin?

Answer 40 Kelvin = Celsius temp + 273
- Slides: 82