Chemistry Midterm Review Regents Chemistry Sig Figs Round










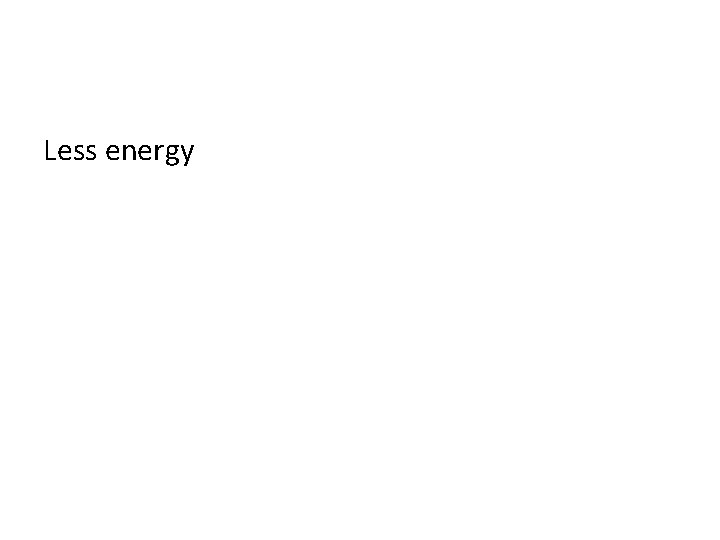

















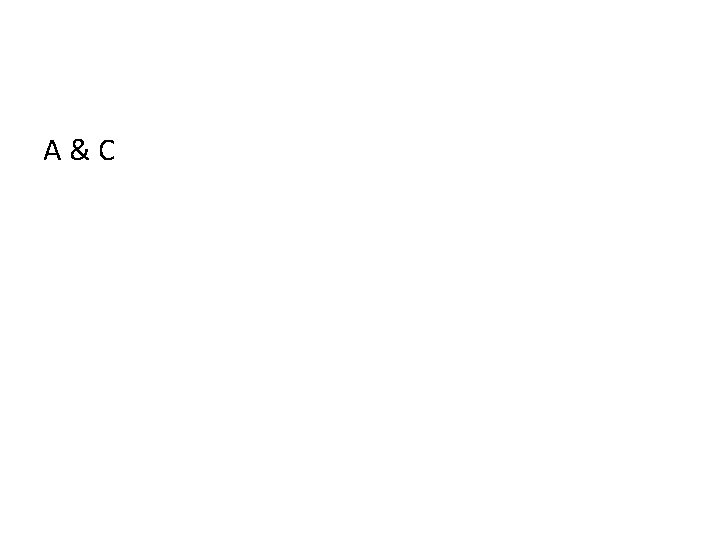

















































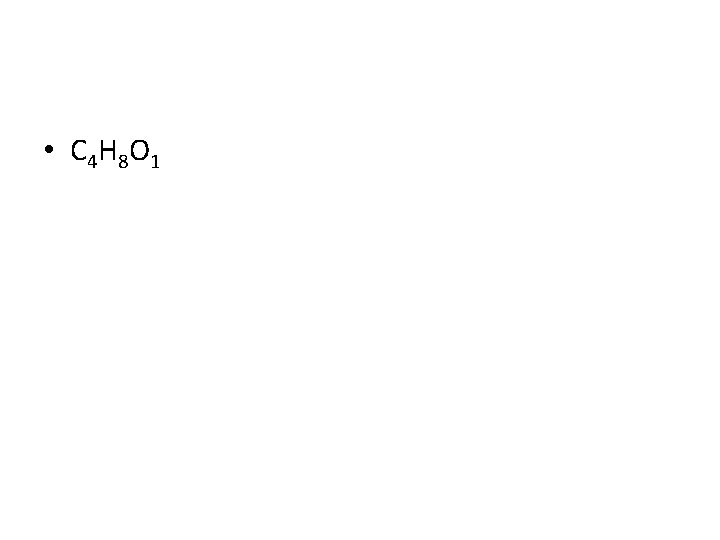






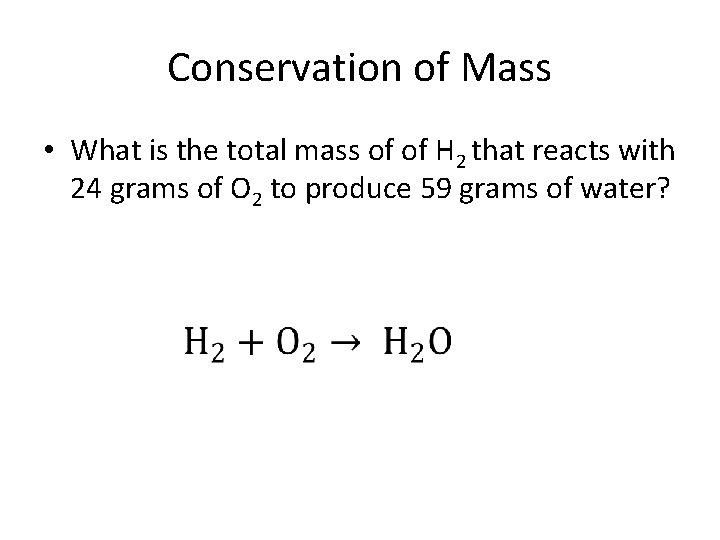


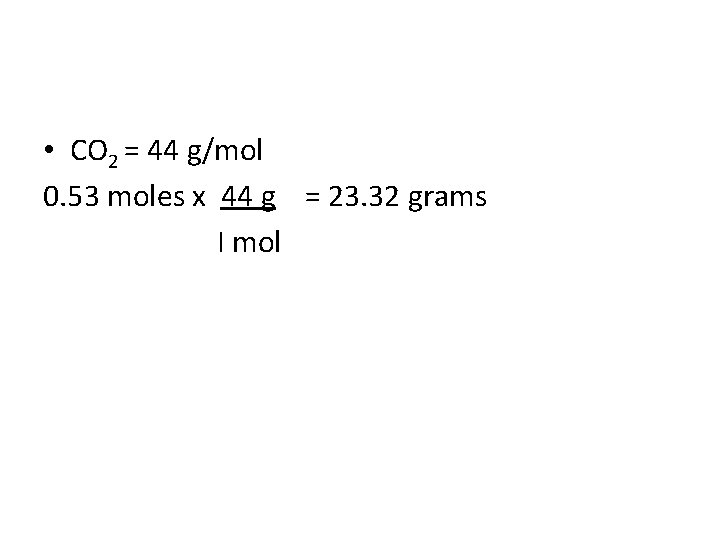



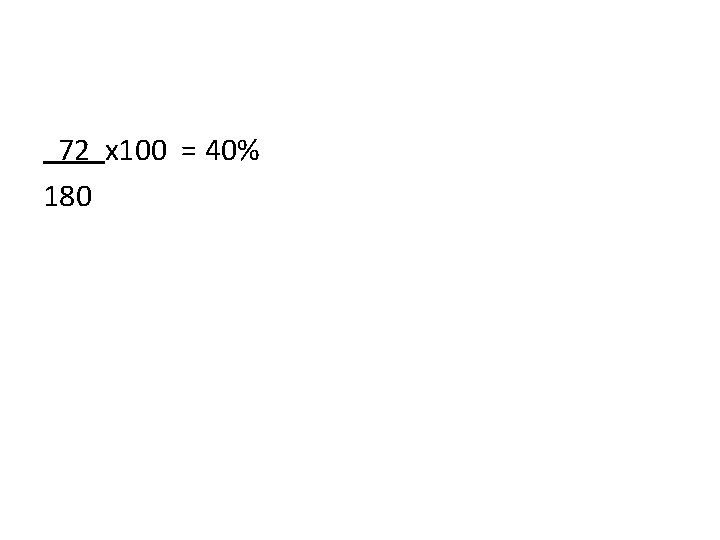
- Slides: 100

Chemistry Midterm Review Regents Chemistry

Sig Figs Round the following number to 3 sig figs: 0. 0067894

0. 00679

Average Atomic Mass Calculate the average atomic mass of Hydrogen given: H-1: (94. 0%) H-2: (4. 0%) H-3: (2. 0%)

(1)(. 94) + (2)(. 04) + (3)(. 02) = 1. 08 amu

Rutherford’s Gold Foil • What two conclusions did Rutherford make about the structure of an atom from his gold foil experiment?

1. Dense, positively charged nucleus 2. Atom is made mostly of empty space

Excited Electron Config. Write an excited electron configuration for an atom of chlorine.

2 -7 -8 OR 1 -8 -8 OR 2 -8 -6 -1

Energy of Electrons found in the first shell (compared to electrons found in the last shell) have _________ (more/less) energy?
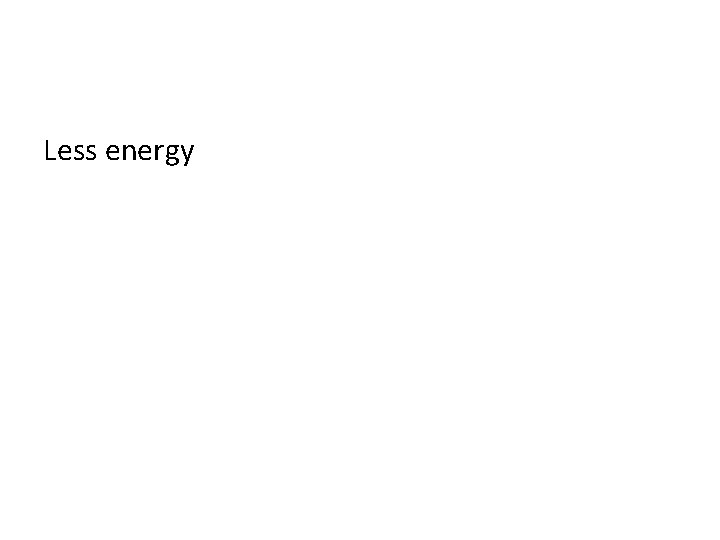
Less energy

Mass of Subatomic Particles • The mass of a proton and neutron is…….

1 amu

Mass of an electron The mass of an electron is …………

0 amu

Charge of subatomic particles What are the charges of a proton, neutron and electron?

Proton = +1 Neutron = 0 Electron = -1

Finding # of p+, e- and n 0 (neutral atom) • How many protons, neutrons and electrons are there in a neutral atom of potassium?

Protons = 19 Neutrons = 20 (39 amu- 19 = 20) Electrons = 19

Finding # of p+, e- and n 0 (ion) How many protons, neutrons and electrons are there in an ion of bromine?

Protons = 35 Neutrons = 45 (80 - 35 = 45) Electrons = 36

Finding # of p+, e- and n 0 (isotope) How many protons, neutrons and electrons are in an isotope of carbon-14?

Protons = 6 Neutrons = 8 (14 -6 = 8) Electrons = 6

Occupied Principal Energy Levels If the electron configuration of an atom is 2 -8 -3, how many occupied principal energy levels are there?

3

Energy of Excited Electrons When an excited electron returns back down to ground state, energy is _________.

Released

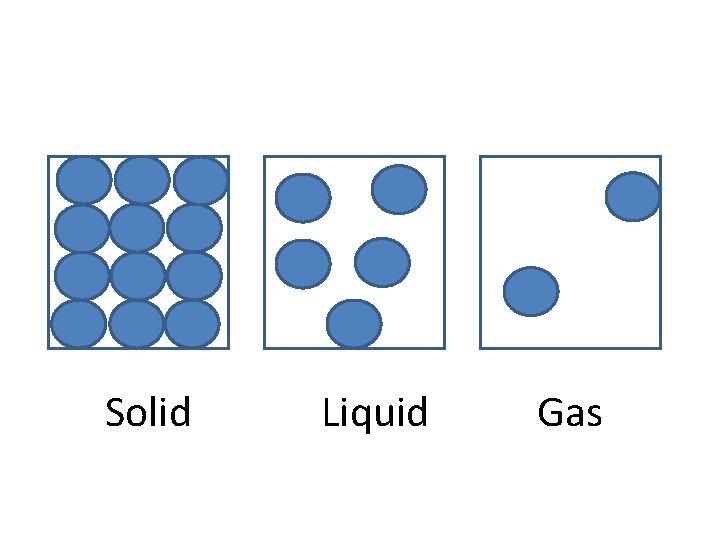
Particle Diagrams Draw a particle diagram for a solid, liquid and gas.

Solid Liquid Gas

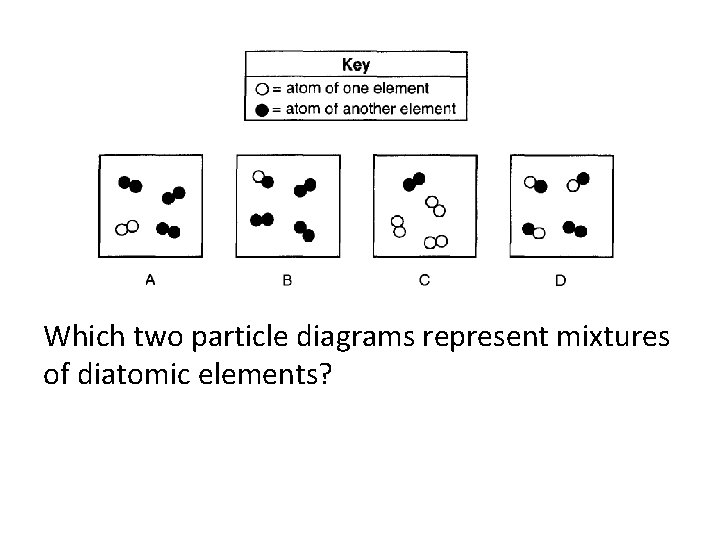
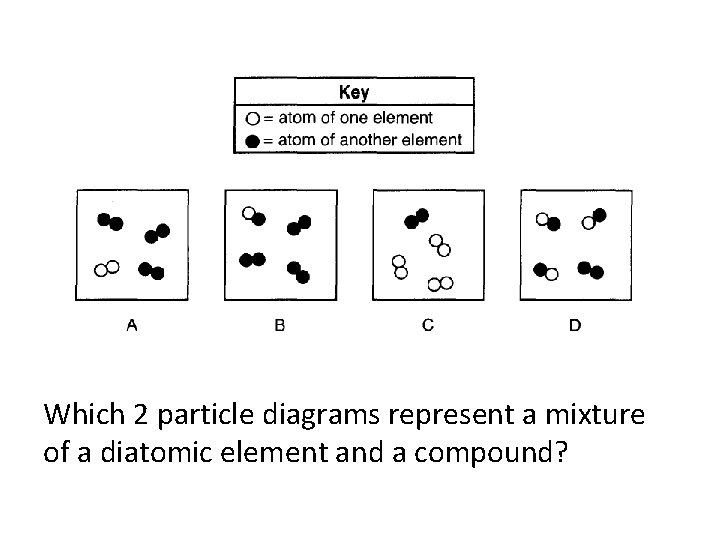
Which two particle diagrams represent mixtures of diatomic elements?
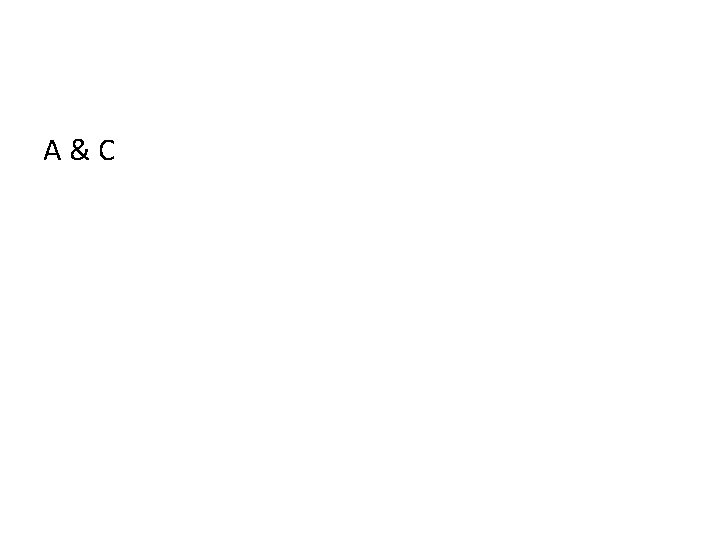
A&C

Which 2 particle diagrams represent a mixture of a diatomic element and a compound?

B&D

Mixtures Which of the following represents a mixture? (1) Li. Cl (s) (2) Li. Cl (g) (3) Li. Cl (l) (4) Li. Cl (aq)

Li. Cl (aq)

Distillation A mixture is separated by distillation. What is distillation?

When a mixture is separated by difference in boiling points.

Metalloid Which element has both properties of metals and non metals? (1) Germanium (2) Sodium (3) Carbon (4) Aluminum

Germanium

Name That Category • • The following properties describe what? Good conductor of heat & electricity Malleable Ductile

• Metals

Name That Category • These group forms colored ions in solution…

• Transition Metals

Trends: Atomic Radius As you go across a period on the periodic table, the atomic radius of each atom_______, due to ______.

Decreases, due to increased nuclear charge

Trends: Atomic Radius

As you go down a group on the periodic table, the atomic radius of each atom _______, due to _____.

Increases, increases of shells/orbitals

Trends: I. E. As you go down a group on the periodic table, the ionization energy of each atom _______, due to _____.

Decreases, increased shielding effect

Trends in I. E. As you go across a period on the periodic table, the ionization energy of each atom _______, due to _____.

Increases, increased nuclear charge

Trends: Electronegativity As you go down a group on the periodic table, the electronegativity of each atom _______, due to _____.

Decreases (think in relation to F!)

Trends: Electronegativity As you go across a period on the periodic table, the atomic radius of each atom _______, due to _____.

Increases (in relation to F!)

Types of Bonding What type of bonding does Ca 3(PO 4)2 have?

• Both ionic and covalent

Types of Bonding What type of bonding does Strontium have?

• Metallic

Molecular Compound • Which is classified as a molecular compound? (a) Na. Cl (b) Br 2 (c) KBr (d) Mg(OH)2


Name That Category • What kind of compound has the following properties: • Good conductor of electricity in aqueous solutions • Crystalline structure • High melting point

• Ionic Compounds

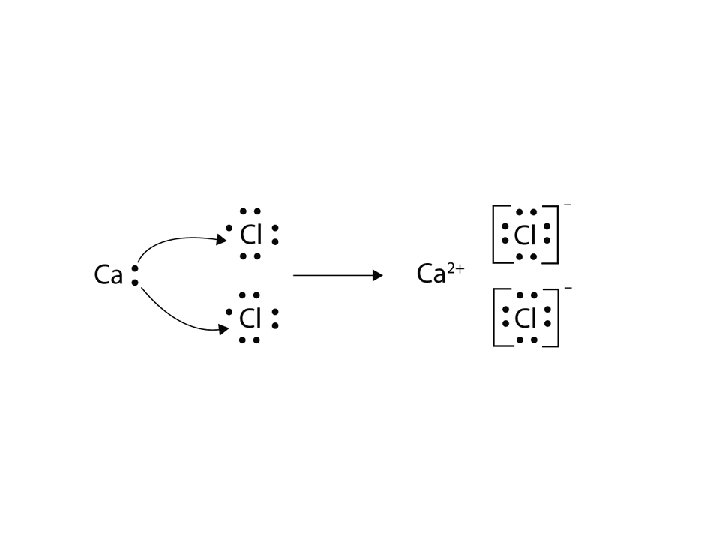
Draw a Dot Diagram • Draw an electron dot diagram for Ca. Cl 2


Draw a Dot Diagram • Draw an electron dot diagram for a sulfide ion.


Draw a Dot Diagram • Draw an electron dot diagram for CO 2


Bond & Energy • Fill in the blank: When a bond is broken, energy must be ________________.

• Absorbed (BARF)

IMF’s A Van der Waals force in responsible of the different phases of matter in group 17. Fill in the blank: When size decreases, Van der Waal force ___________.

• Decreases (think of your gases at the top of the group).

Bond Polarity • Does H 2 O have polar or nonpolar bonds? • Why?

• Polar bonds because the EN difference between H and O is greater than 0. 4.

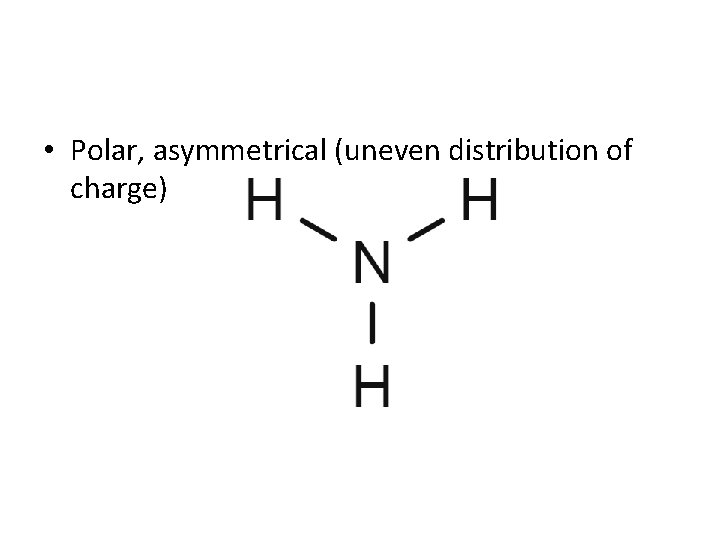
Molecular Polarity • Draw the structural formula of NH 3. • Is this a polar or nonpolar molecule? And why?

• Polar, asymmetrical (uneven distribution of charge)

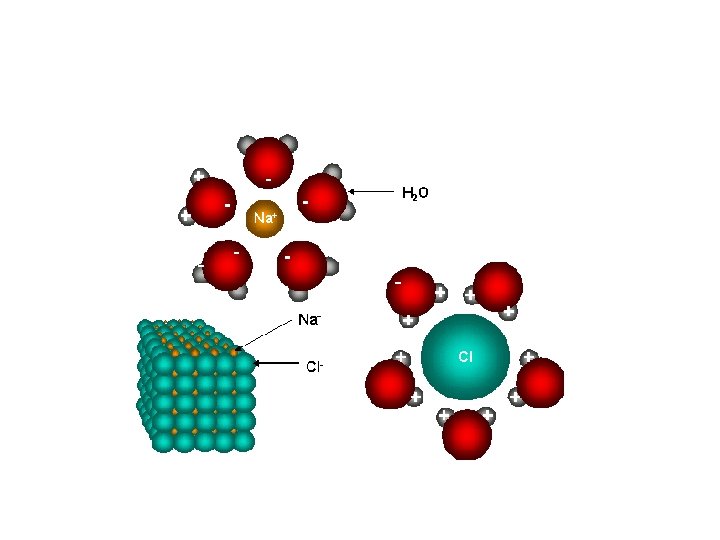
Draw a Picture • Draw a picture of a molecule ion attraction between Na. Cl and H 2 O.


Bright Line Spectra • Identify the elements in the mixture:

• Strontium & Lithium

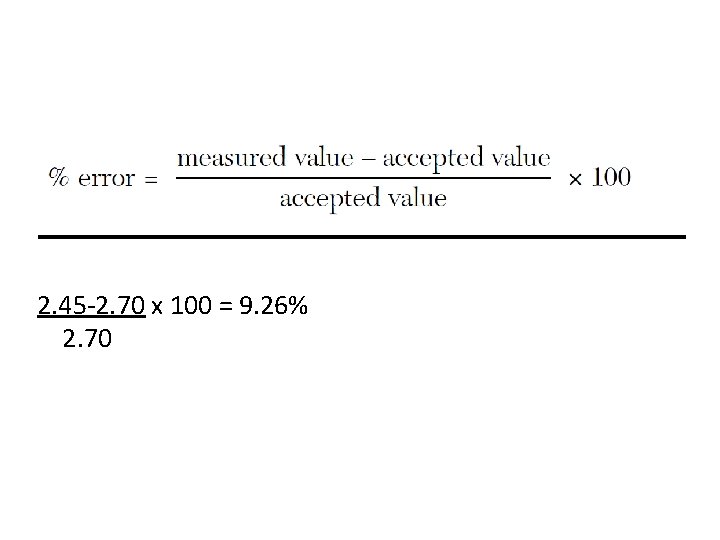
Percent Error • A student measured the density of Aluminum to be 2. 45 g/cm 3. Calculate the student’s percent error.

2. 45 -2. 70 x 100 = 9. 26% 2. 70

Empirical Formula • What is the empirical formula of the following compound? • C 8 H 16 O 2
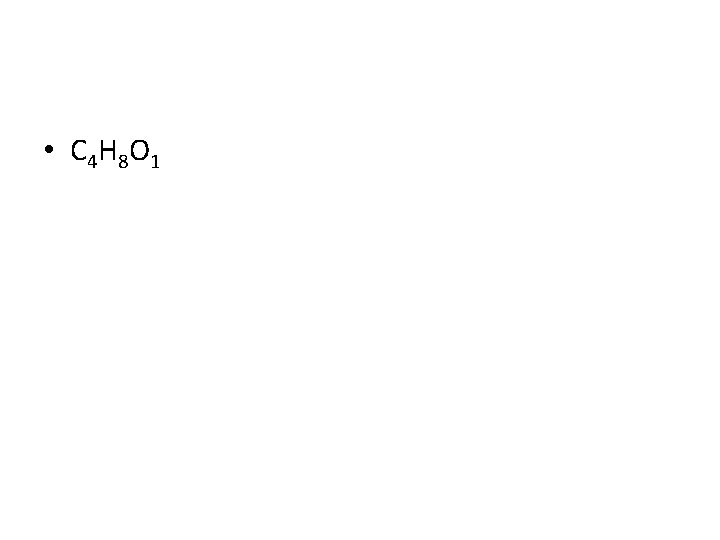
• C 4 H 8 O 1

Types of Reactions • What type of reaction is represented by the equation below:

• Synthesis

Types of Reactions • What type of reaction is represented by the equation below:

• Single Replacement

Balancing Equations What are the sum of the coefficients after balancing the following equation:

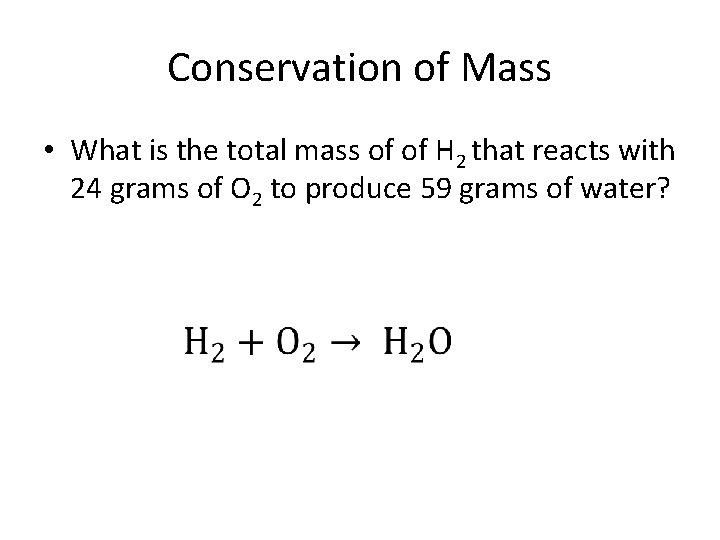
Conservation of Mass • What is the total mass of of H 2 that reacts with 24 grams of O 2 to produce 59 grams of water?

• 35 grams

Mole Conversions • What is the total mass in grams of 0. 53 moles of CO 2?
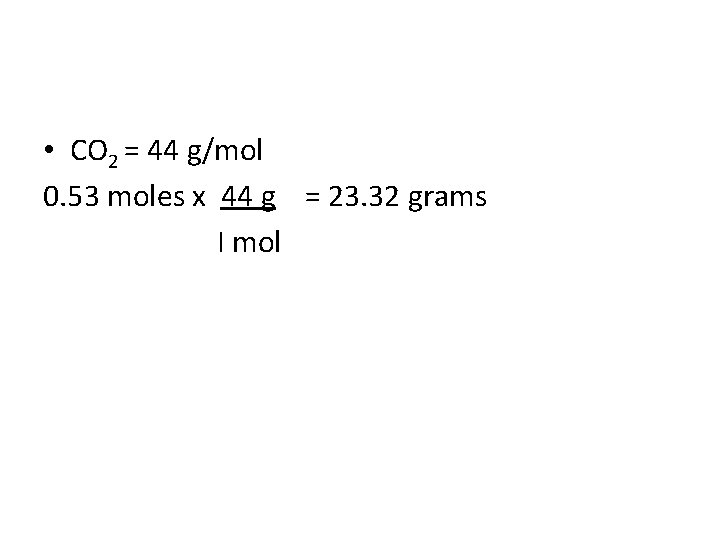
• CO 2 = 44 g/mol 0. 53 moles x 44 g = 23. 32 grams I mol

Molecular Formulas • What is the molecular formula of a compound with an empirical formula of CH 4 and a molecular mass of 128 g/mol?

• CH 4 = 16 g/mol • 128 = 8 16 C 8 H 32

Percent Composition • Show a correct numerical setup for calculating the percent by mass of Carbon in C 6 H 12 O 6
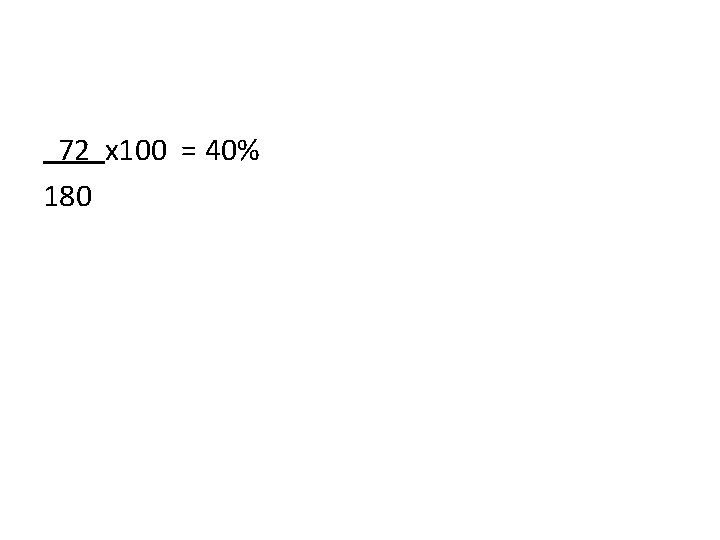
72 x 100 = 40% 180