Biology Midterm Review Introduction and Characteristics of Living

Biology Midterm Review

Introduction and Characteristics of Living Things

What is S C I E N C E ? ? ? • Science is a body of knowledge based on the study of nature. • The nature or essential characteristics, of science is scientific inquiry. • Scientific inquiry is both a creative process and a process rooted in unbiased observations and experimentation.

BIO - - LOGY ? ? ? • Biology comes from the Greek bio, meaning “life”, and from logos, meaning “study” • Whenever you see –ology, it means “the study of” • In Biology we study: • The origins and history of life, both past and present • The structures of living things • How living things interactive with one another • How living things function
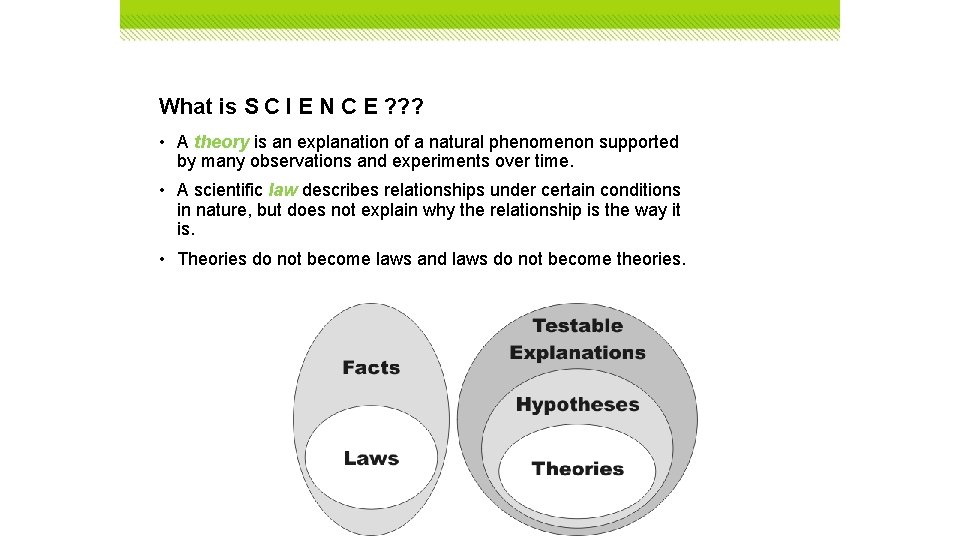
What is S C I E N C E ? ? ? • A theory is an explanation of a natural phenomenon supported by many observations and experiments over time. • A scientific law describes relationships under certain conditions in nature, but does not explain why the relationship is the way it is. • Theories do not become laws and laws do not become theories.

Characteristics of Life Organisms had or have all of the characteristics of life (8 Total): • 1. Made of one or more cells • Cells are the basic unit of all living things – the building blocks • One cell = unicellular; more than one cell = multicellular • 2. Displays organization • They arrange in an orderly way • Unicellular contain organized functional structures and often work together; multicellular have specialized cells organized into tissues, tissues organized into organ, organs organized into organ systems that work together to support life • 3. Grows and develops • Most everything starts as a single cell • Mass is then added (usually by added new cells) through cell growth, and have natural changes over organism’s lifetime that is called development • 4. Reproduces • Make offspring! • Species are organisms that can breed to produce fertile offspring

Characteristics of Life, cont. • 5. Responds to stimuli • Stimuli = anything that causes a reaction (response) by the organism • 6. Requires energy • Food provides energy • Most plants use light energy from the Sum to make their own (photosynthesis); organisms that don’t make their own get it by consuming others (that leads back to something that does) • 7. Maintains homeostasis • A balance and regulation of internal conditions • 8. Adaptations evolve over time • = inherited characteristic that allows species to survive more efficiently • Usually caused by a change in environment

The Scientific Process

A Systematic Approach • An observation is the act of gathering information. • Qualitative data is obtained through observations that describe color, smell, shape, or some other physical characteristic that is related to the 5 senses. • Quantitative data is obtained from numerical observations that describe how much, how little, how big, or how fast. • A hypothesis is a tentative explanation for what has been observed. • An experiment is a set of controlled observations that test the hypothesis.

A Systematic Approach • A variable is a quantity or condition that can have more than one value. • An independent variable is the variable you plan to change. • The dependent variable is the variable that changes in value in response to a change in the independent variable • EXAMPLE: If you were trying to determine if temperature affects bacterial growth, you would expose different petri dishes of the same bacteria to different temperatures • Temperature is your independent variable • Bacteria growth is your dependent variable Variables Song (Creepy guy)

Science literacy • Scientific inquiry begins with observation. • Scientific inquiry involves asking questions and processing information from a variety of reliable sources. • The process of combining what you know with what you have learned to draw logical conclusions is called inferring; the conclusions themselves are called inferences. • The methods scientists use to gather data and answer questions are referred to as scientific methods.

Scientific Method • Scientific Method is a step-bystep organized plan for gathering, organizing, and communicating information. STEPS 1. Make Observation 2. Ask Question 3. Develop Hypothesis 4. Experiment (include variables) 5. Analyze Data and Draw Conclusions - State if hypothesis is supported or not supported 6. Develop Theory
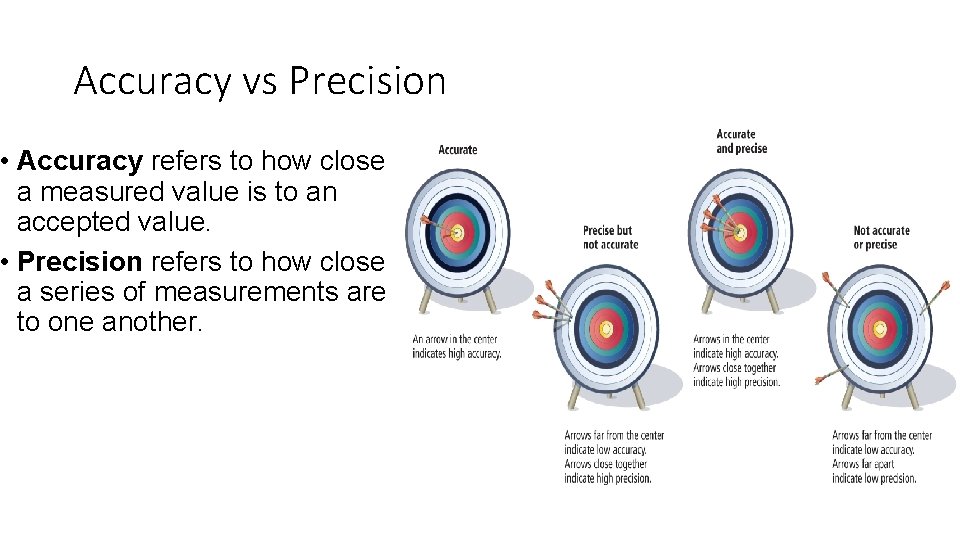
Accuracy vs Precision • Accuracy refers to how close a measured value is to an accepted value. • Precision refers to how close a series of measurements are to one another.

SI Units of Measure • All measurements need a number and a unit. • Example: 5 ft 3 in or 25ºF • The metric system uses units with divisions that are powers of ten (used in most of the world besides us – use the imperial system • Scientists usually do not use these units. They use a unit of measure called SI or International System of Units. • Base Units – more examples on following slide • Length- straight line distance between 2 points is the meter (m) • Mass- quantity of matter in an object or sample is the kilogram (kg)
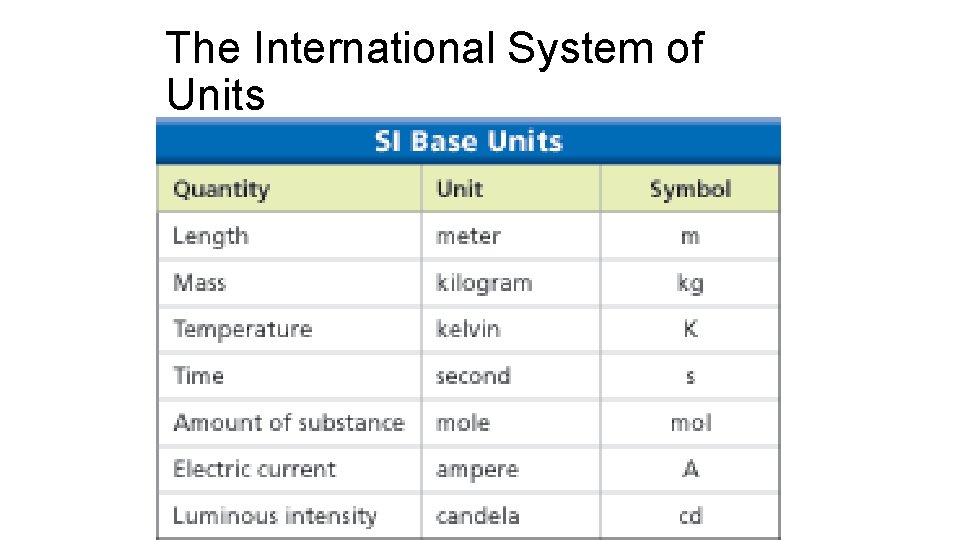
The International System of Units •

Biochemistry
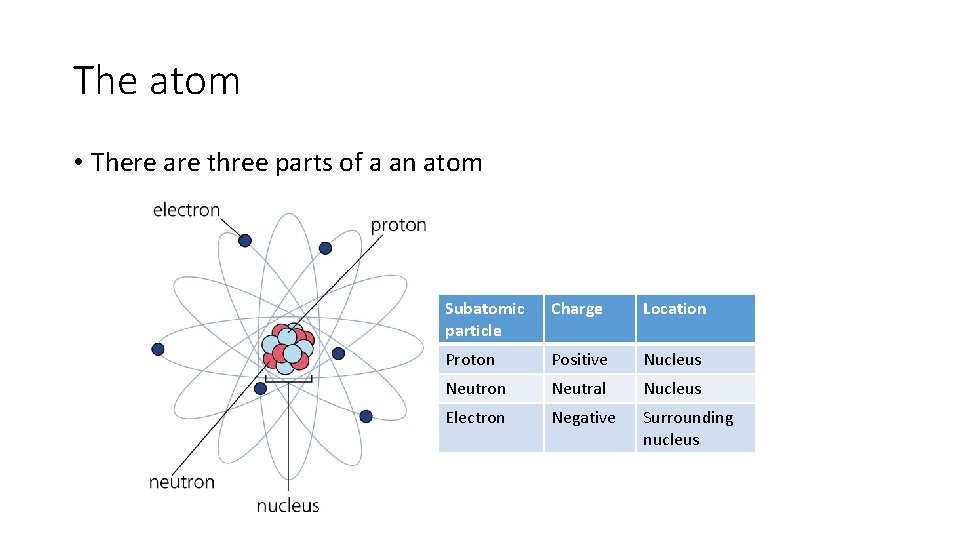
The atom • There are three parts of a an atom Subatomic particle Charge Location Proton Positive Nucleus Neutron Neutral Nucleus Electron Negative Surrounding nucleus
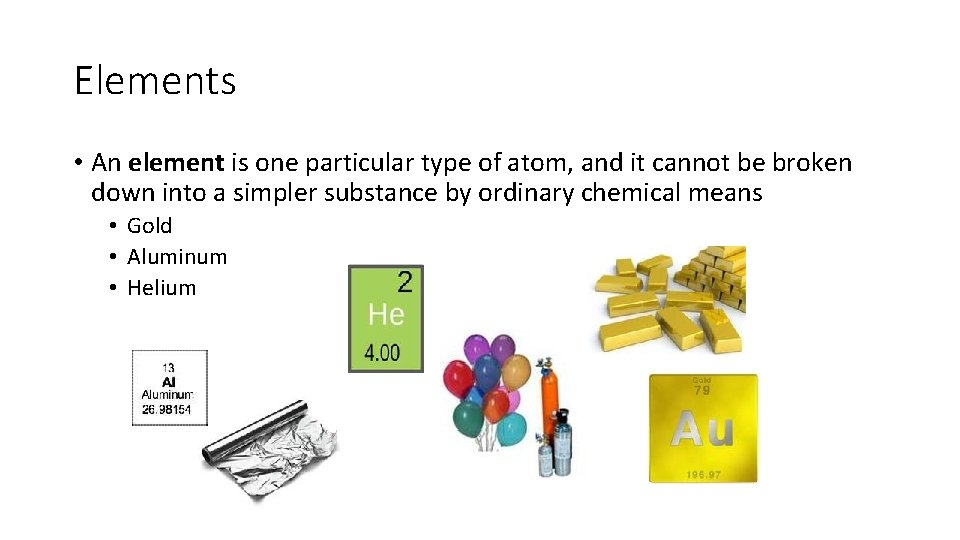
Elements • An element is one particular type of atom, and it cannot be broken down into a simpler substance by ordinary chemical means • Gold • Aluminum • Helium

Key Elements • In biology, there are SIX very important elements • • • Carbon Hydrogen Oxygen Phosphorus Sulfur Nitrogen N S P O H C
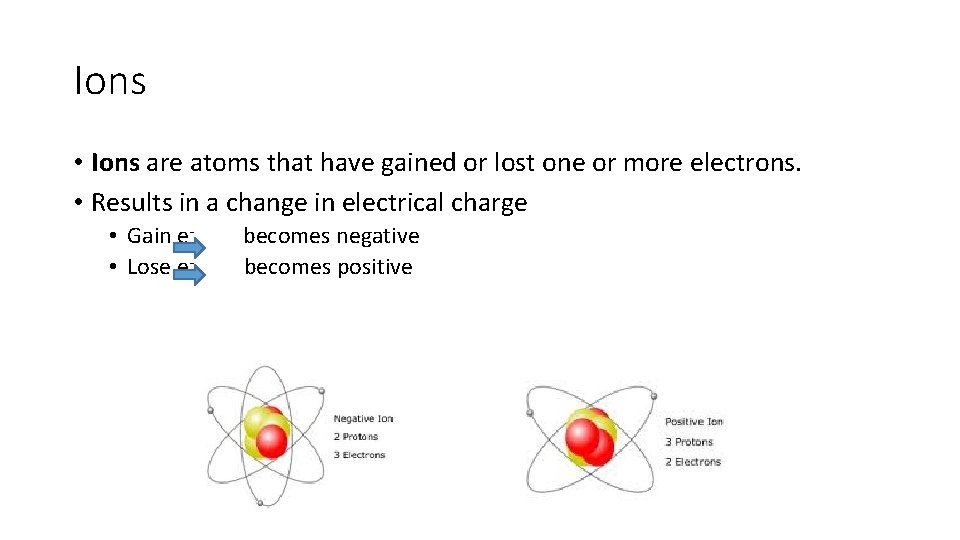
Ions • Ions are atoms that have gained or lost one or more electrons. • Results in a change in electrical charge • Gain e- becomes negative • Lose e- becomes positive
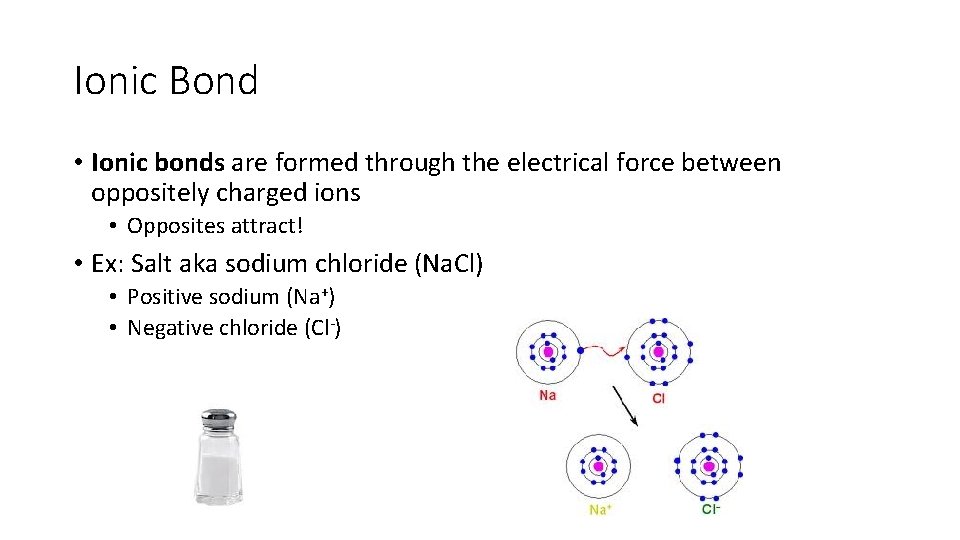
Ionic Bond • Ionic bonds are formed through the electrical force between oppositely charged ions • Opposites attract! • Ex: Salt aka sodium chloride (Na. Cl) • Positive sodium (Na+) • Negative chloride (Cl-)

Covalent Bond • Not all atoms easily gain or lose their electrons! • Some atoms share their electrons instead! • Covalent Bond: forms when atoms share a pair of electrons • Usually a very strong bond • Atoms may have several covalent bonds to share several electrons

Evidence of a chemical reaction Temperature change Formation of a solid (precipitate) Color change (sometimes) Odor (sometimes) Formation of gas

� Reactants are the substance changed during a chemical reactions �Oxygen (O 2) & Glucose (C 6 H 12 O 6) � Products reaction are the substances made by a chemical �Carbon Dioxide (CO 2) & Water (H 2 O) 6 O 2 + C 6 H 12 O 6 Reactants 6 CO 2 + 6 H 2 O Products
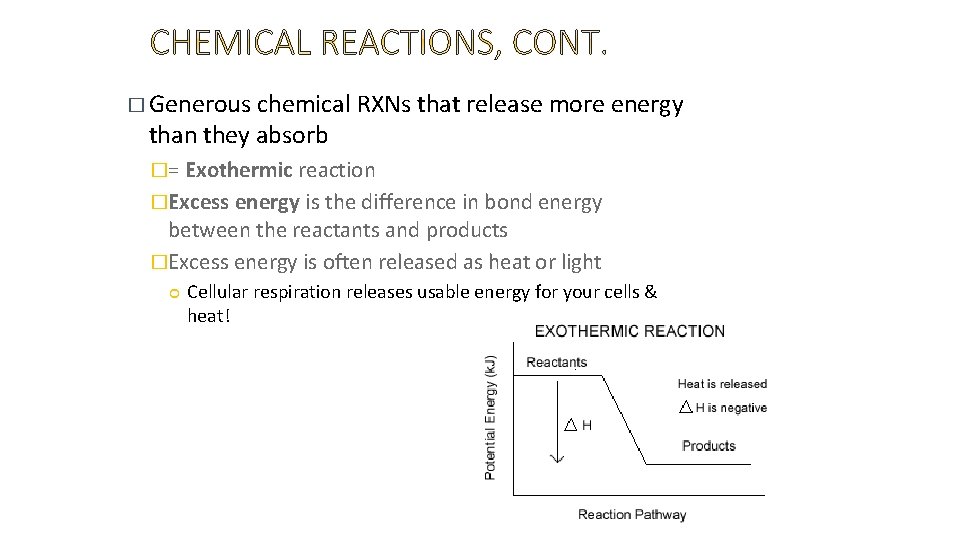
� Generous chemical RXNs that release more energy than they absorb �= Exothermic reaction �Excess energy is the difference in bond energy between the reactants and products �Excess energy is often released as heat or light Cellular respiration releases usable energy for your cells & heat!

� Greedy chemical RXNs that absorb more energy than they release �= Endothermic reaction In photosynthesis, plants absorb energy from sunlight and use that to make sugars and carbohydrates

But how does a rxn start? • Some energy must first be absorbed by the reactants in ANY chemical reaction • The amount of energy needed will vary • Activation energy is the amount of energy that needs to be absorbed for a chemical reaction to start • Push a rock up a hill

Catalysts & Enzymes • A catalyst is a substance that lowers the activation energy needed to start a chemical reaction. • Special proteins called enzymes are the biological catalysts that speed up the rate of chemical reactions in biological processes.

___________ 1. HIGH SPECIFIC HEAT ___________ 2. COHESION ___________ 3. ADHESION

Cohesion: the attraction among _________ of the same MOLECULES substance. Cohesion from hydrogen bonds makes water molecules ___________. STICK TOGETHER Cohesion produces _________, SURFACE TENSION ( “skin on water” )

Adhesion: the attraction among _________ MOLECULES of _______ DIFFERENT substances. For example, water molecules stick to other things. Water in a test tube, (water is attracted to the ______) GLASS

Mixtures Heterogeneous Homogeneous • A mixture with easily separated parts with their distinct properties • A mixture that it the SAME throughout and hard to tell individual characteristics
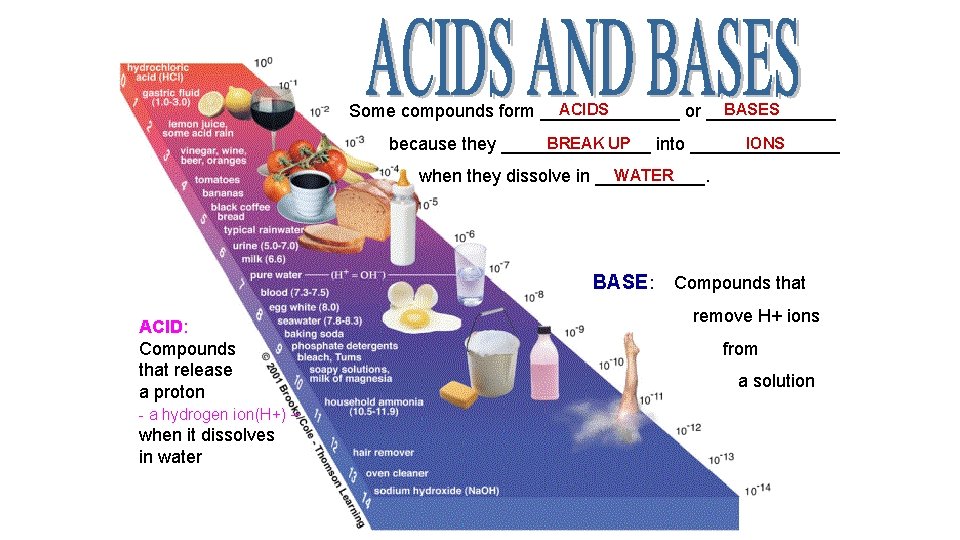
ACIDS BASES Some compounds form _______ or _______ BREAK UP IONS because they ________ into ________ WATER when they dissolve in ______. BASE : Compounds that ACID: Compounds that release a proton - a hydrogen ion(H+) – when it dissolves in water remove H+ ions from a solution
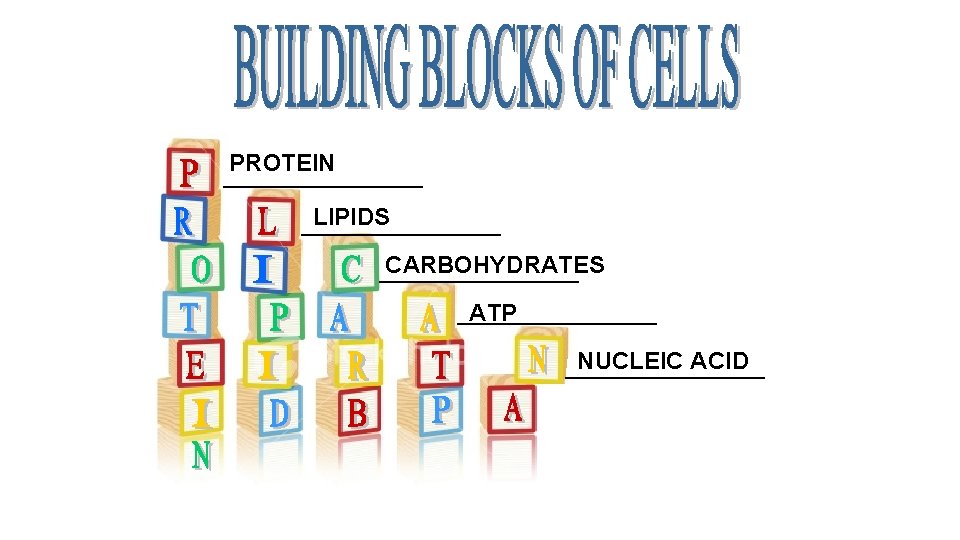
PROTEIN __________ LIPIDS __________ CARBOHYDRATES __________ ATP __________ NUCLEIC ACID __________

• USED BY CELLS FOR _____ ENERGY ______ SOURCE AND _____ ENERGY______. STORAGE SIMPLE CARBS = ________________ ONE OR TWO SUGAR MOLECULES CHAINS OF SUGAR COMPLEX CARBS =LONG _______________ MOLECULES _______________ Ex) Starches such as potato, ________________ pasta, bread ________________
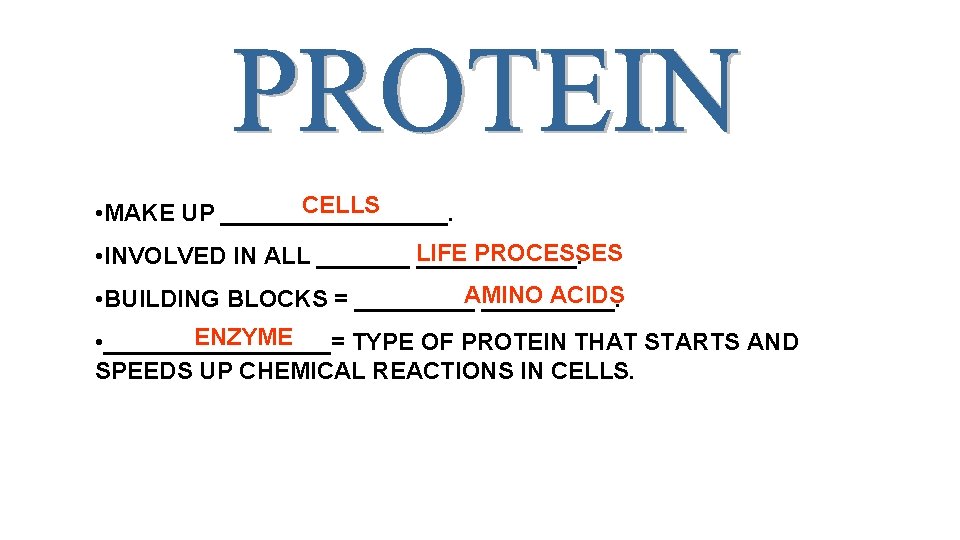
CELLS • MAKE UP _________. PROCESSES • INVOLVED IN ALL _______ LIFE ______. ACIDS • BUILDING BLOCKS = _____AMINO _____. ENZYME • _________= TYPE OF PROTEIN THAT STARTS AND SPEEDS UP CHEMICAL REACTIONS IN CELLS.

WATER • DON’T MIX WITH _______. CELL MEMBRANE • PHOSPHOLIPIDS – MAKE UP ______________. STORE ______. ENERGY • FATS AND OILS THAT _____ • ORGANISMS USE FATS AND OILS WHEN THEY HAVE CARBOHYDRATES USED UP __________. FATS • _____SOLID AT ROOM TEMPERATURE. OILS • _____LIQUID AT ROOM TEMPERATURE.

ENERGY CARRYING • MAJOR ___________ MOLECULE IN CELL. CARBOHYDRATES AND _____ LIPIDS • ENERGY IN _________ AND PROTEINS ___________ MUST BE TRANSFERRED TO ATP IN CELL TO BE USED.

PROTEIN • HAVE ALL INFO NEEDED TO MAKE _____. BLUE PRINT • “________” OF LIFE. • BUILDING BLOCKS OF NUCLEOTIDES _______. DNA RNA • TWO TYPES: ____ AND _______.
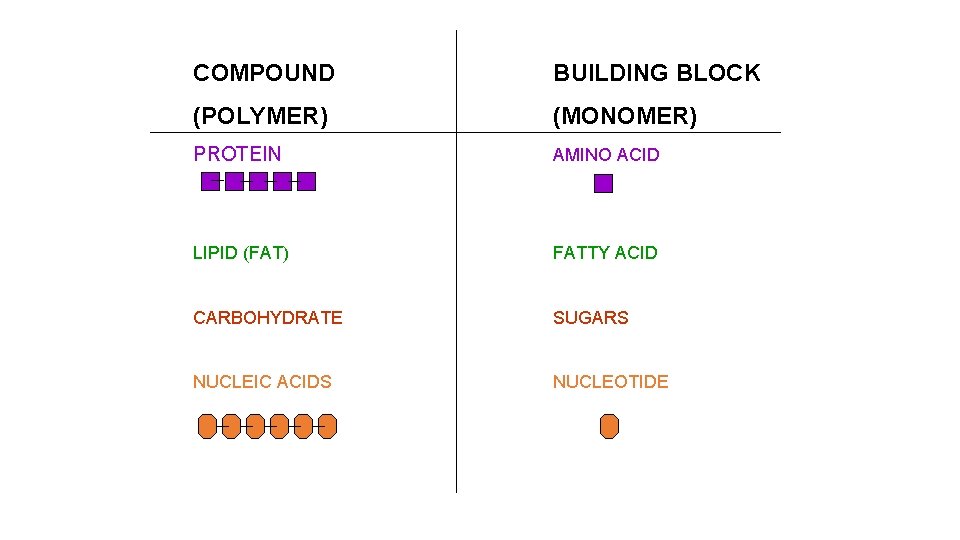
COMPOUND BUILDING BLOCK (POLYMER) (MONOMER) PROTEIN AMINO ACID LIPID (FAT) FATTY ACID CARBOHYDRATE SUGARS NUCLEIC ACIDS NUCLEOTIDE

Cells

Microscope Technology Compound light microscopes • Use a series of glass lenses and visible light to magnify images • Magnify images up to ~1000× actual size Electron microscopes • Create an image by illuminating a sample with a beam of electrons and collecting the electrons that are reflected back from the sample • Magnify images up to 500, 000× actual size Cell Discovery and Theory
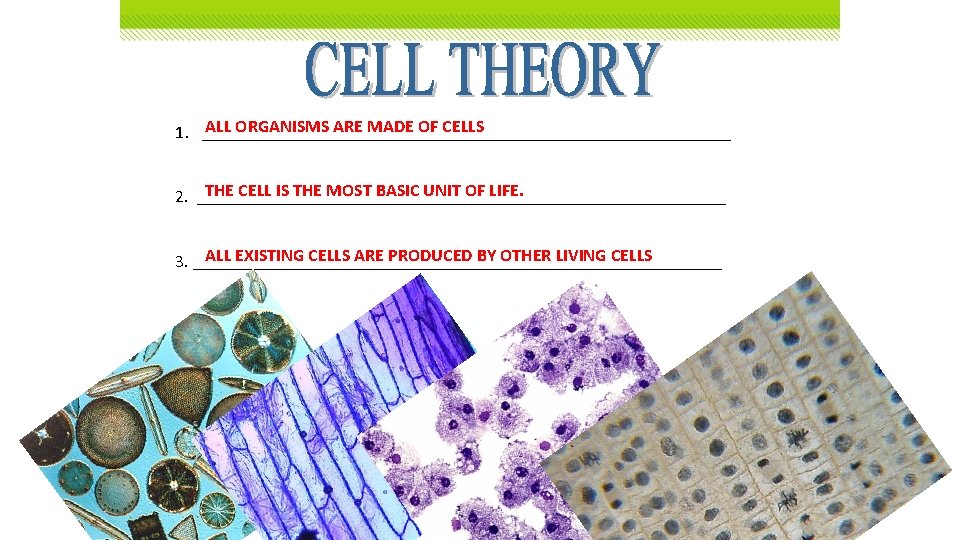
ALL ORGANISMS ARE MADE OF CELLS 1. ______________________________ THE CELL IS THE MOST BASIC UNIT OF LIFE. 2. ______________________________ ALL EXISTING CELLS ARE PRODUCED BY OTHER LIVING CELLS 3. ______________________________

HOOKE _______ CORK LEEUWENHOEK _______ ANIMALCULES Made better The first to LENSES _______ IDENTIFY ______ cells. and observed cells in Responsible for greater NAMING ______ them DETAIL _______. First to observe NUCLEUS _______ SCHLEIDEN _______ SCHWANN _______ VIRCHOW _______ RESPONSIBLE FOR CELL THEORY The first to note Concluded that all LIVING THINGS ______ that ______ were _______ PLANTS made up of were made up of CELLS ___________ Proposed that all cells come from OTHER CELLS ______

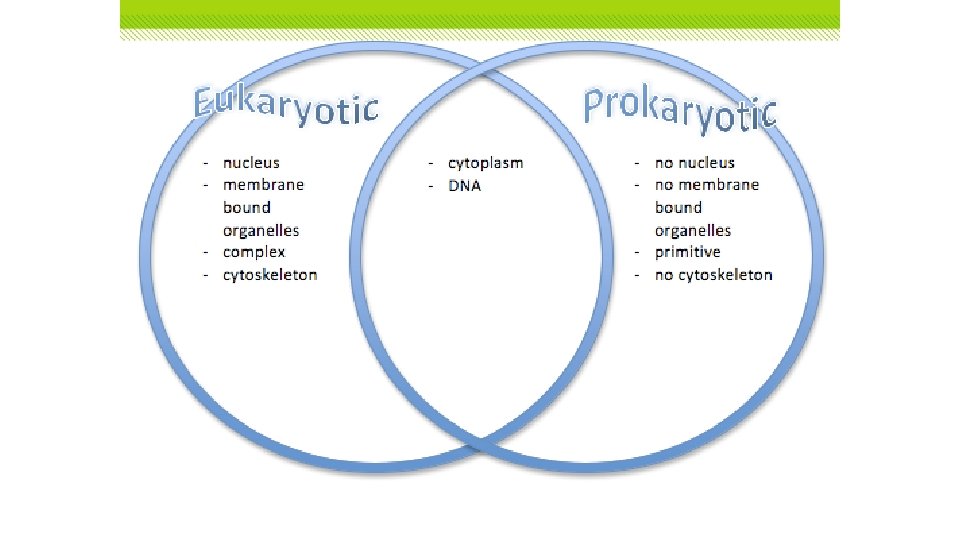
Basic Cell Types • Eukaryotic cells contain a nucleus and other organelles. • Organelles are specialized structures that perform specific cell functions. • The nucleus is the distinct central organelle that contains the cell’s genetic material. • Prokaryotic cells are cells without a nucleus or other membrane-bound organelles. • Smaller and simpler than eukaryotes • Probably similar to first organisms that lived on earth Cell Discovery and Theory
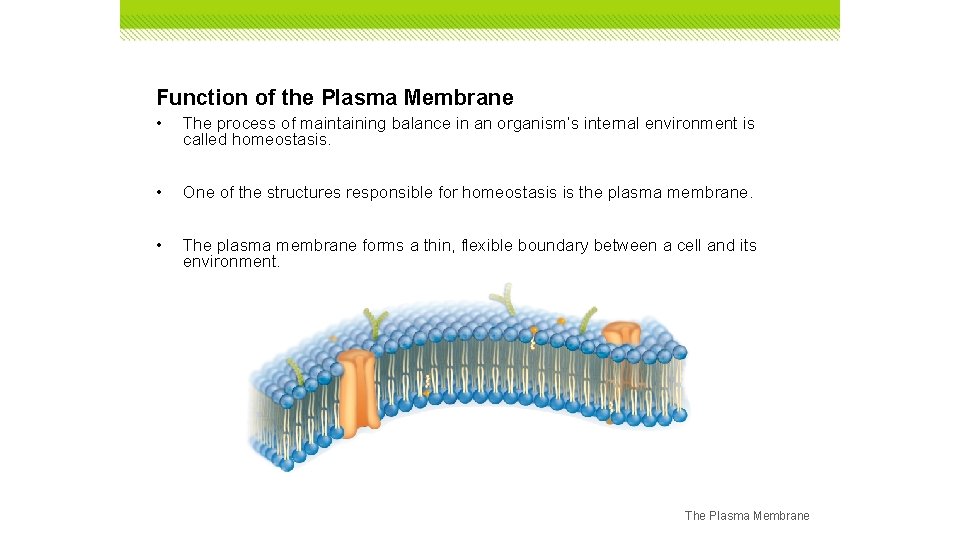
Function of the Plasma Membrane • The process of maintaining balance in an organism’s internal environment is called homeostasis. • One of the structures responsible for homeostasis is the plasma membrane. • The plasma membrane forms a thin, flexible boundary between a cell and its environment. The Plasma Membrane
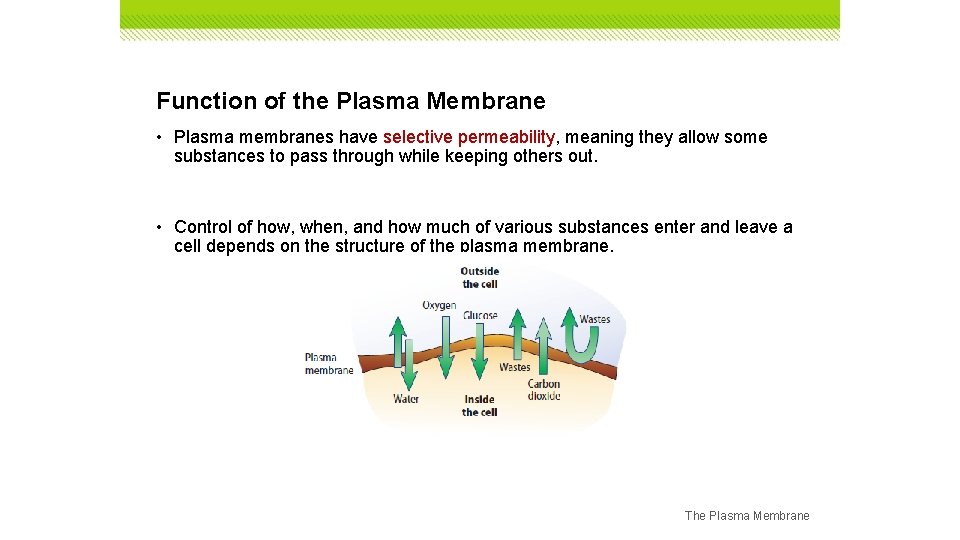
Function of the Plasma Membrane • Plasma membranes have selective permeability, meaning they allow some substances to pass through while keeping others out. • Control of how, when, and how much of various substances enter and leave a cell depends on the structure of the plasma membrane. The Plasma Membrane
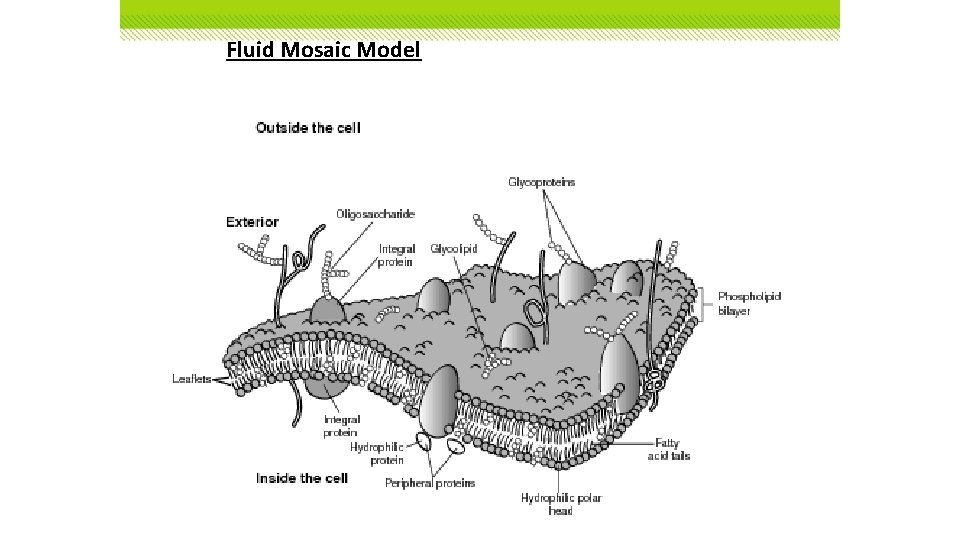
Fluid Mosaic Model
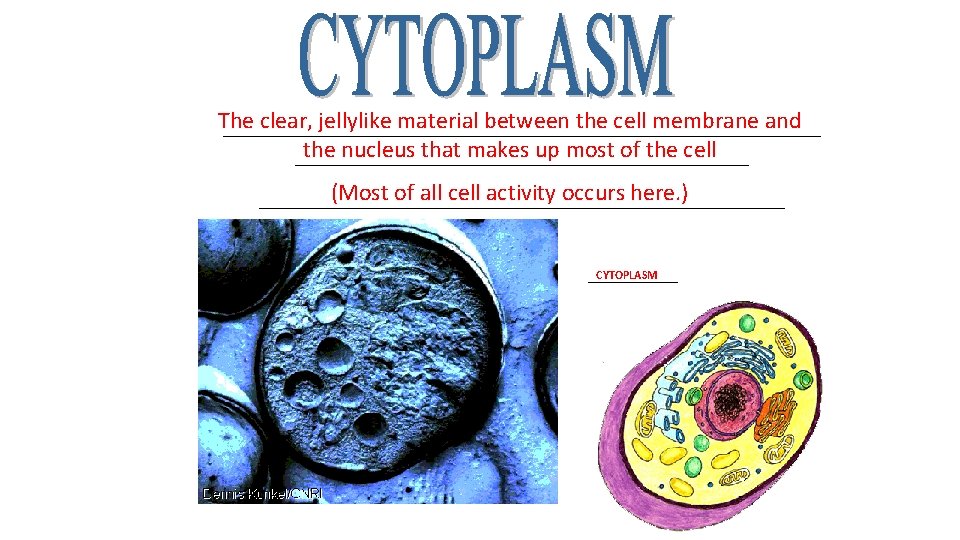
The clear, jellylike material between the cell membrane and _________________________ the nucleus that makes up most of the cell ___________________ (Most of all cell activity occurs here. ) ______________________ CYTOPLASM ________
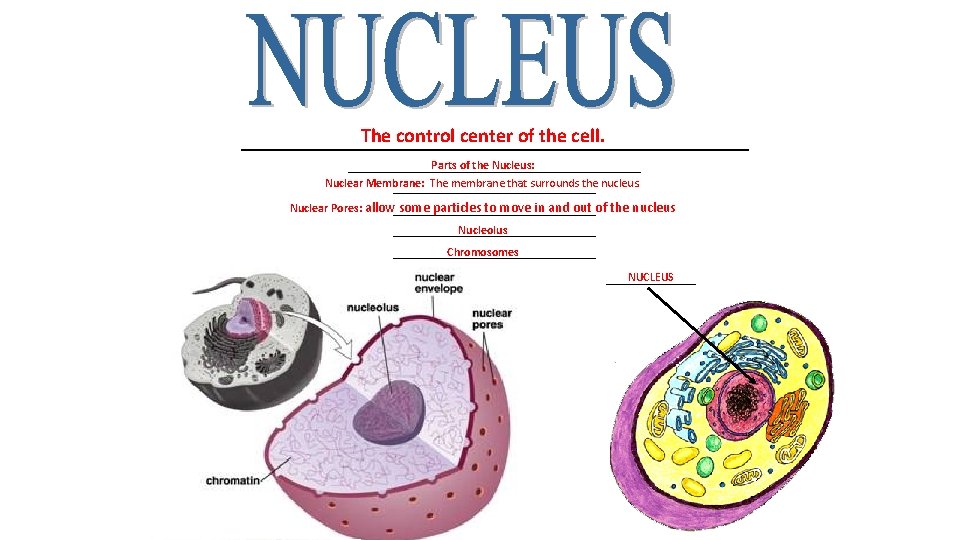
The control center of the cell. __________________________ Parts of the Nucleus: _________________________ Nuclear Membrane: The membrane that surrounds the nucleus. _________________ Nuclear Pores: allow some particles to move in and out of the nucleus _________________ Nucleolus _________________ Chromosomes _________________ NUCLEUS ________
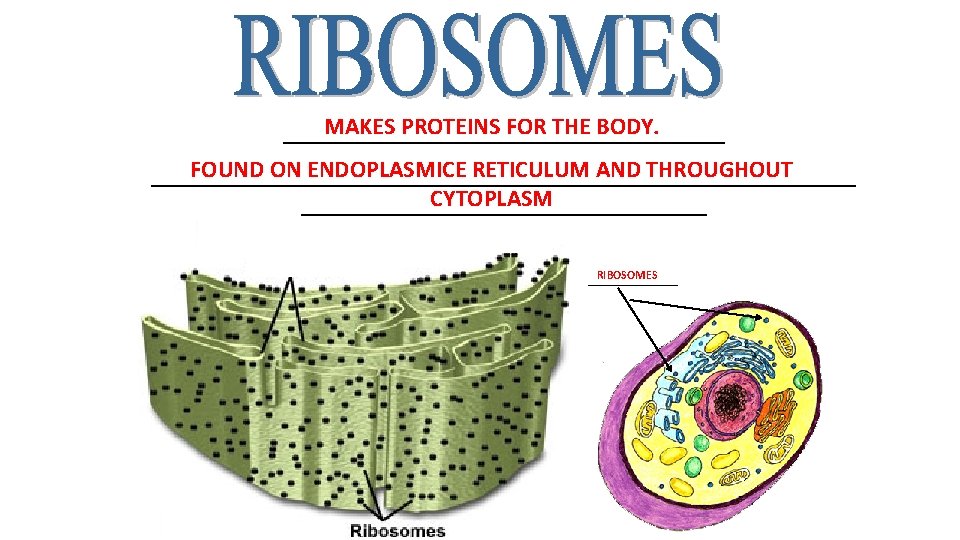
MAKES PROTEINS FOR THE BODY. ___________________ FOUND ON ENDOPLASMICE RETICULUM AND THROUGHOUT ______________________________ CYTOPLASM _________________ RIBOSOMES ________
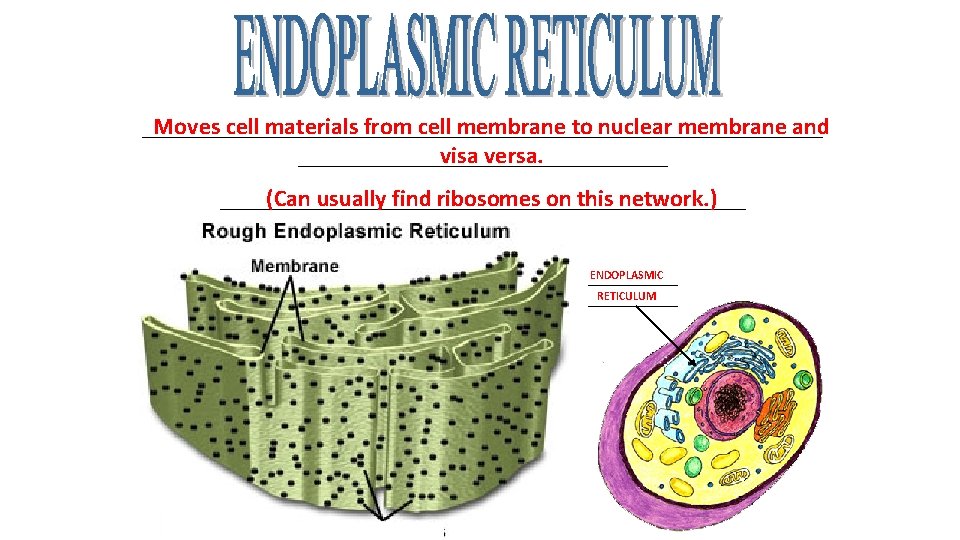
_____________________________ Moves cell materials from cell membrane to nuclear membrane and ________________ visa versa. ______________________ (Can usually find ribosomes on this network. ) ENDOPLASMIC ________ RETICULUM ________
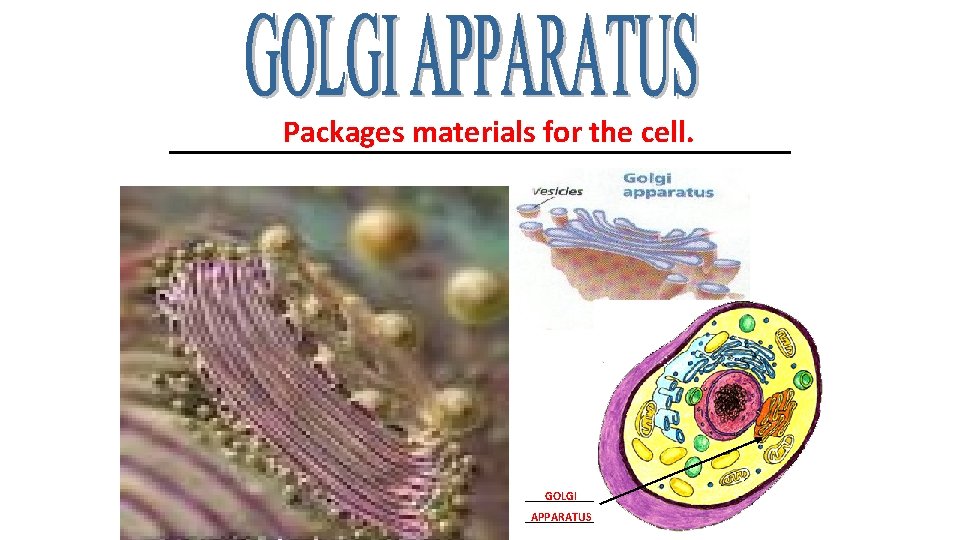
Packages materials for the cell. ____________________ GOLGI _______ APPARATUS
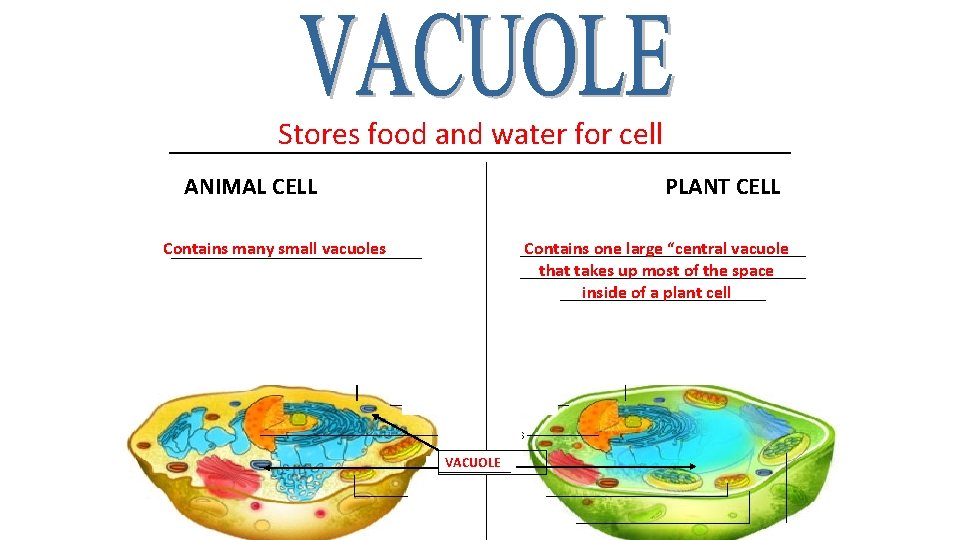
Stores food and water for cell ____________________ ANIMAL CELL PLANT CELL ________________ Contains one large “central vacuole ________________ that takes up most of the space ____________ inside of a plant cell Contains many small vacuoles ______________ VACUOLE ____
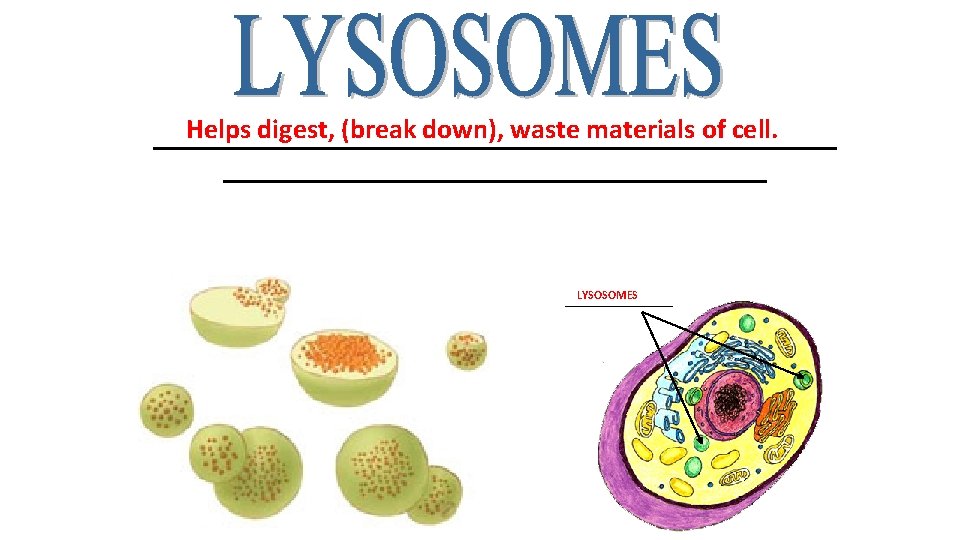
Helps digest, (break down), waste materials of cell. _________________________ LYSOSOMES ______
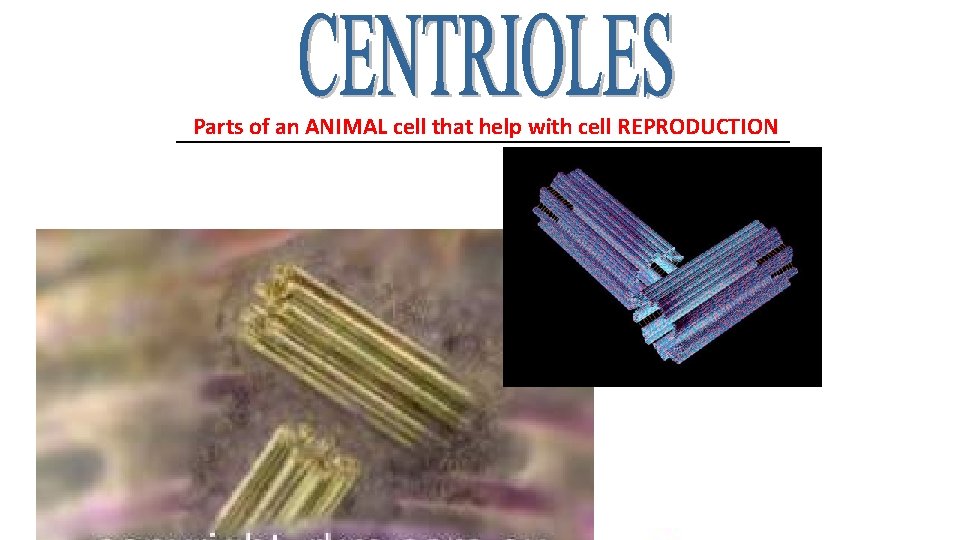
Parts of an ANIMAL cell that help with cell REPRODUCTION ______________________
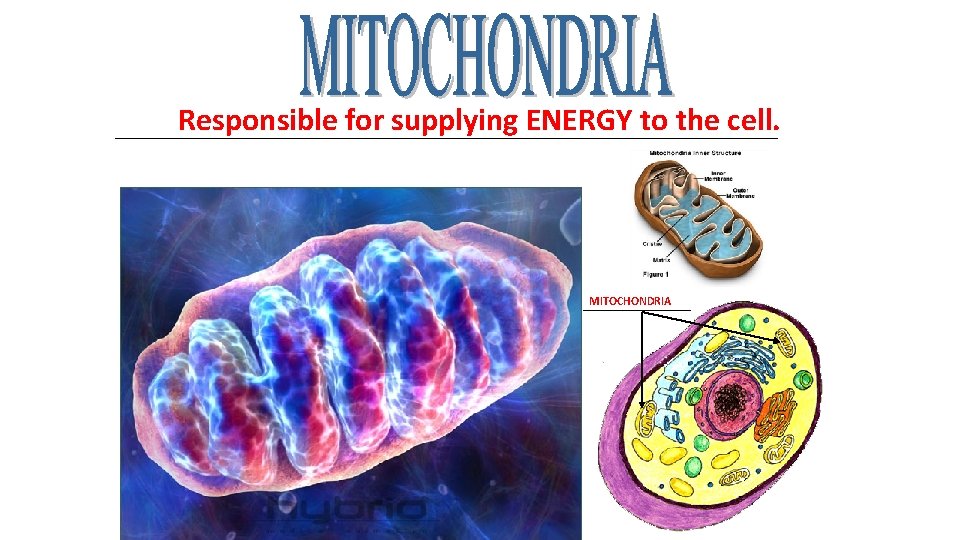
Responsible for supplying ENERGY to the cell. _____________________________________ MITOCHONDRIA ______
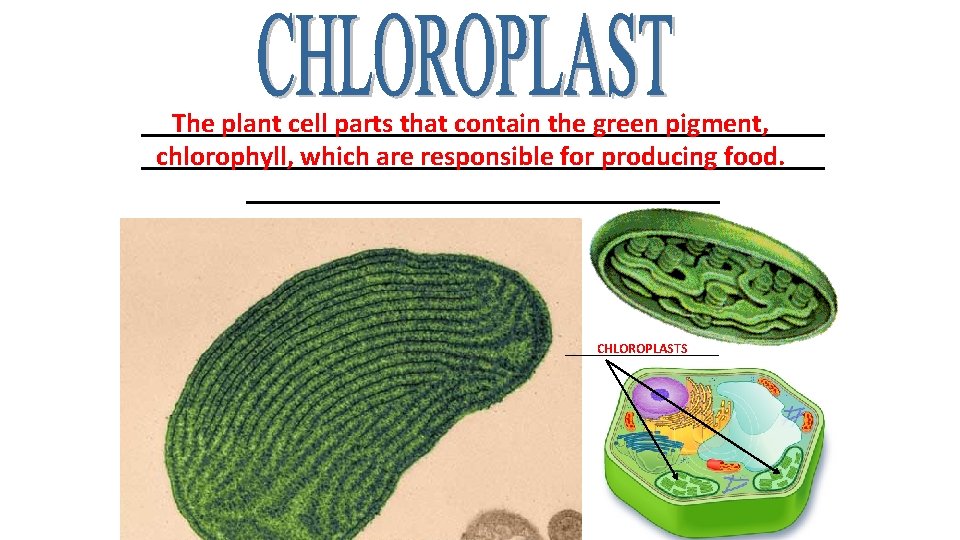
_________________________ The plant cell parts that contain the green pigment, _________________________ chlorophyll, which are responsible for producing food. _________________ CHLOROPLASTS
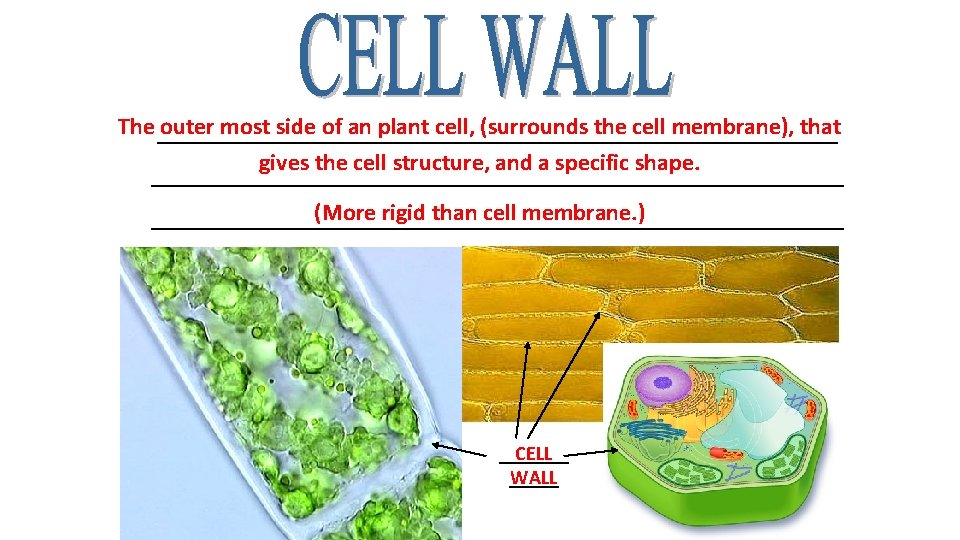
The _____________________________ outer most side of an plant cell, (surrounds the cell membrane), that gives the cell structure, and a specific shape. _____________________________ (More rigid than cell membrane. ) _____________________________ CELL _____ WALL
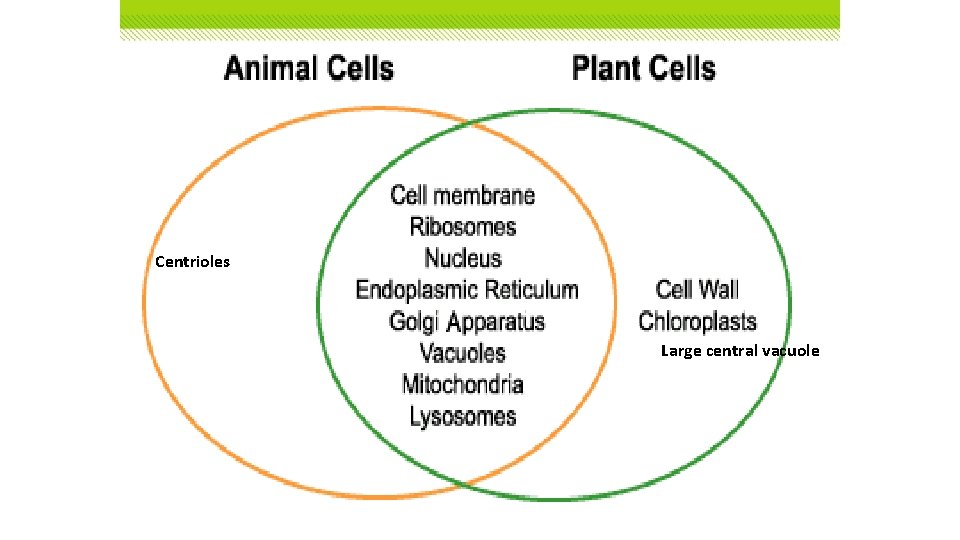
Centrioles Large central vacuole
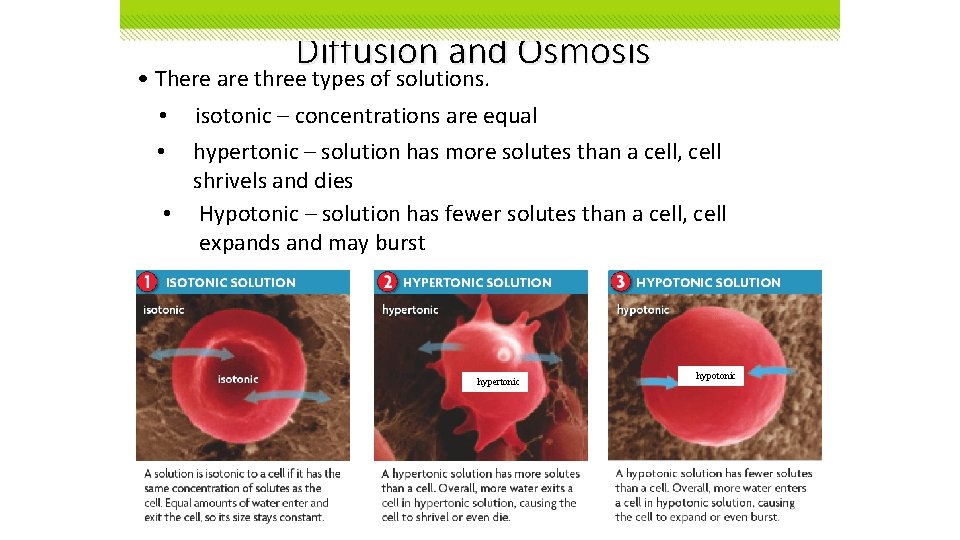
Diffusion and Osmosis • There are three types of solutions. • • isotonic – concentrations are equal hypertonic – solution has more solutes than a cell, cell shrivels and dies • Hypotonic – solution has fewer solutes than a cell, cell expands and may burst hypertonic hypotonic
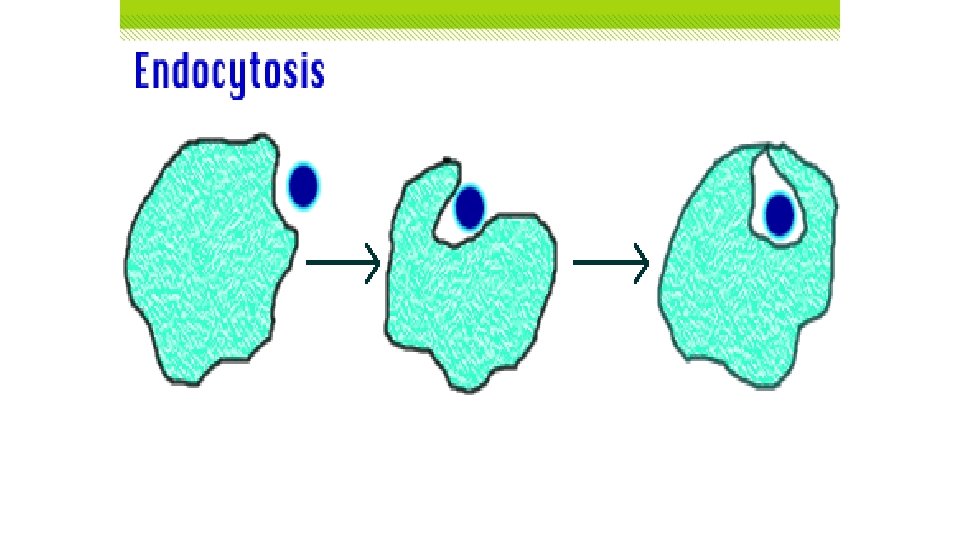
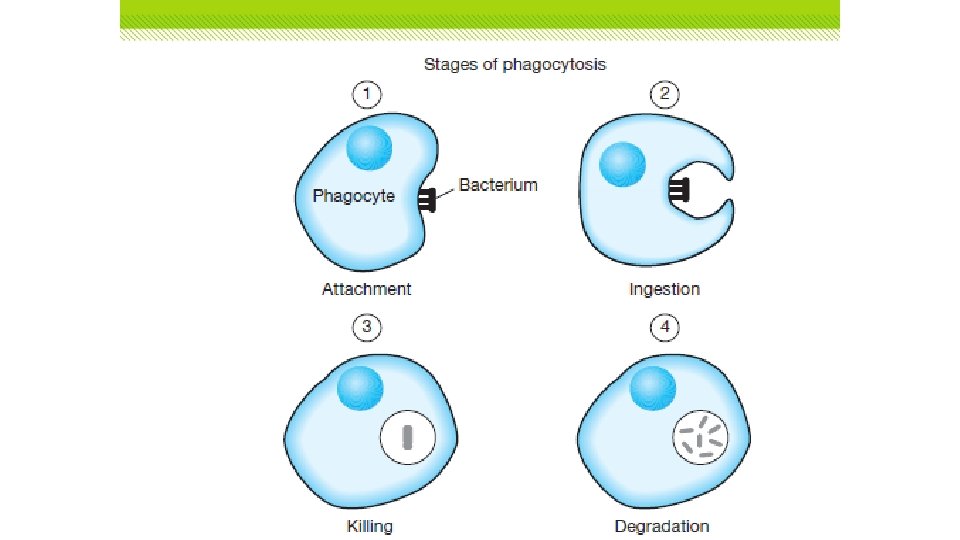
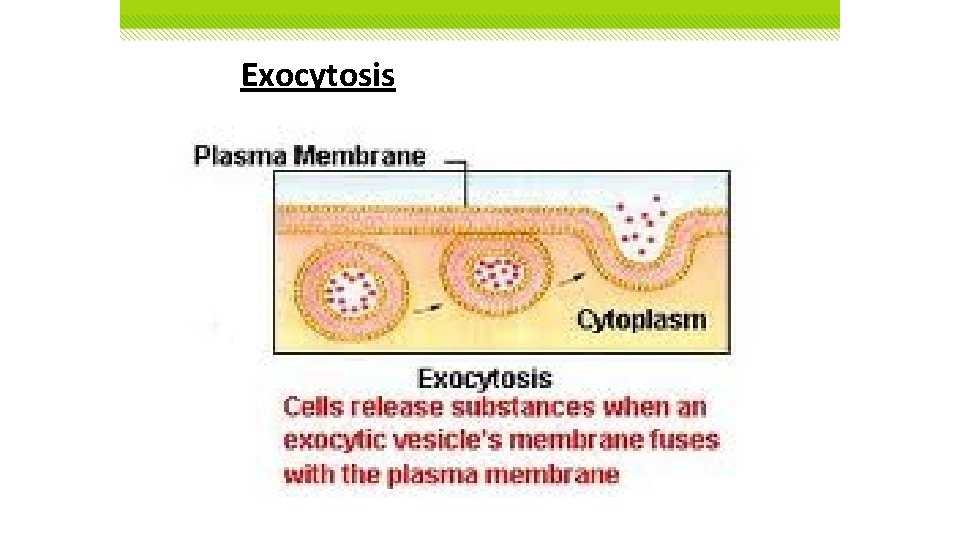
Exocytosis
- Slides: 66