Noble Gases Noble Gases The noble gases helium

Noble Gases

Noble Gases • The noble gases Øhelium (He) Øneon (Ne) Øargon (Ar) Økrypton (Kr) Øxenon (Xe) Øradon (Rn) üform group 18 of the periodic table

Noble Gases Ooccurrence • Minor constituents of the atmosphere Ø Isolated first by Ramsay Ø Fractionation of liquid air • Helium occurs in radioactive mineral • Radon Ø Radioactive with short half-lives Ø Characterized in the decay series from uranium and thorium

Noble Gases • Electonic Configurations ØHe ØNe ØAr ØKr ØXe ØRn 1 s 2 [He] 2 s 2 2 p 6 [Ne] 3 s 2 3 p 6 [Ar] 3 d 10 4 s 2 4 p 6 [Kr] 4 d 10 5 s 2 5 p 6 [Xe] 4 f 14 5 d 10 6 s 2 6 p 6

Noble Gases • The chemical inertness ØThe stability of the noble gases with respect to loss or acceptance of electrons is due to their high ionization potentials and the highly positive values of their electron affinities. These effects are essentially responsible for the chemical inertness of these elements.

Noble Gases • Compounds Ø It was long believed that the noble gases were incapable of forming chemical compounds. Ø In 1962 three groups succeeded independently in preparing noble gas compounds. ü In June 1962 Bartlett prepared the orangeyellow, moisture-sensitive compound "xenon hexafluoroplatinate" by reaction of Xe with Pt. F 6. ü In July 1962 Hoppe obtained the first binary compound of xenon, Xe. F 2 , ü This was followed one month later by the synthesis of Xe. F 4 by Claasen, Selig, and Malm.

Noble Gas • Bonding in Compounds ØA surprising result üThe valence compounds of krypton and xenon do not involve a new type of chemical bonding. v. The atoms are bound in the same manner as in the long-known interhalogen compounds, such as IF 7 , and in Te. F 6.

Noble Gas • Bonding in Compounds ØFor the formation of noble gas compounds üAn electron must be promoted from the p shell to the d shell ü Formation of bonding electron pairs with another atom can occur with hybridization of the type s p d n (n = 1, 2, 3, 4).

Noble Gas • Types of Compounds ØNoble gas compounds can be divided into three general types : üShort-lived molecules containing noble gas atoms üValence compounds üInclusion compounds (clathrates and intercalation compounds)

Noble Gas • Types and Stability of Compounds ØValence compounds üOnly formed with the most electronegative elements (till now, F, Cl, Br, N, and C) üWhereby thermodynamically stable compounds with fluorine are formed only by xenon and radon.

Noble Gas • Types and Stability of Compounds Ø The thermodynamic stability of noble gas compounds, for example, the halides, follows the following general rules : ü The stability of the compounds EX 2 increases with increasing atomic number of the noble gas and with decreasing atomic number of the halogen : v. Ar. F 2 < Kr. F 2 < Xe. F 2 < Rn. F 2 v. Xe. F 2 > Xe. Cl 2 > Xe. Br 2 ü The stability of the compounds decreases as the oxidation state of the noble gas increases.

Noble Gas • Compounds Ø Up to now, attempts to react helium, neon, and argon with other elements have failed Ø The chemistry of krypton is limited to the detection of the ions Kr. F+, Kr 2 F 3+, Kr. F 2 radical, and the synthesis of Kr. F 2 and its complexes Kr. F 2· 2 Sb. F 5 and Kr. F 2· x As. F 5. A report of the detection of Kr. F 4 proved to be erroneous. The existence of a compound with a Kr – N bond is claimed

Noble Gas • Compounds ØCompounds of xenon are known in the oxidation states II – VIII, some of which are remarkably stable. Xenon (II) fluoride is even commercially available.

• Table 21 -2 Oxidat Compound Form ion state II Xe. F 2 colorless crystals IV Xe. F 4 colorless crystals VI Xe. F 6 colorless VI Xe. OF 4 colorless liquid VI Xe. O 3 colorless crystals n K+[Xe. O 3 F–]n colorless VI crystals VIII Xe. O 4 colorless gas VIII Xe. O 64– colorless salt mp, °C Structure 129 linear 117 square planar – 46 octahedral tetrahedral qpy Fbridges tetrahedral octahedral

Noble Gas • Preparation of Compounds • Xe + F 2 = Xe. F 2 • Xe + 2 F 2 = Xe. F 4 • Xe + 3 F 2 = Xe. F 6 400 ºC, 0. 1 M Pa, deficiency of F 2 600 ºC, 0. 6 M Pa Xe : F 2 = 1 : 5 300 ºC, 6 M Pa

Noble Gas • Properties of Compounds • Hydrolysis Xe. F 2 + 2 OH- = Xe + 1/2 O 2 + 2 F 2 + H 2 O Xe. F 4 + 6 H 2 O = Xe. O 3 + 2 Xe + 3/2 O 2 + 12 HF Xe. F 6 + 3 H 2 O = Xe. O 3 + 6 HF • Oxidation Na. Br. O 3 + Xe. F 2 + 2 H 2 O = Na. Br. O 4 + 2 HF + Xe • Fluoridation 2 Xe. F 6+ 3 Si. O 2 = 2 Xe. O 3 + 3 Si. F 4

Noble Gas • Molecular Structures of Compounds Ø The structures of the fluorides, oxyfluorides, and oxides of xenon follow the rules of the valence shell electron pair repulsion model (VSEPR) Ø When the lone pairs are taken into account ü Xe. F 2 , Xe. OF 2 , and Xe. O 2 F 2 have trigonal bipyramidal structures with a linear F – Xe – F axis in the gas phase.

Noble Gas • Molecular Structures of Compounds ü In Xe. F 4 and its oxyfluorides, the four fluorine atoms occupy equatorial positions, while the electron lone pairs or oxygen atoms occupy the axial positions of the octahedral structure.
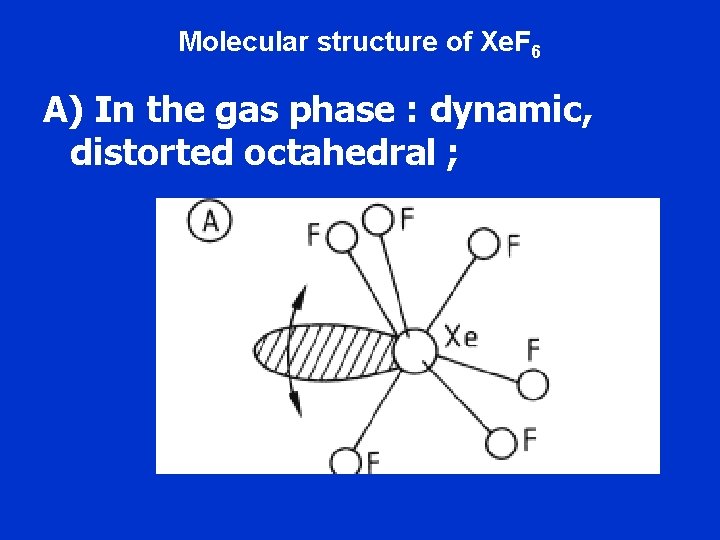
Molecular structure of Xe. F 6 A) In the gas phase : dynamic, distorted octahedral ;
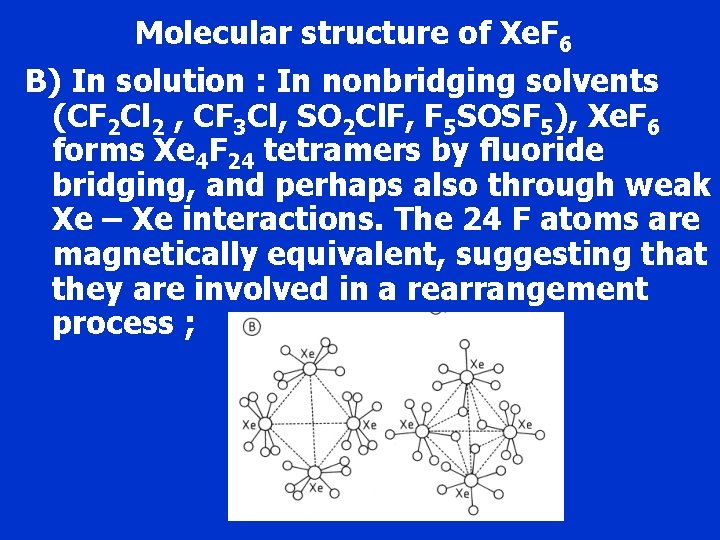
Molecular structure of Xe. F 6 B) In solution : In nonbridging solvents (CF 2 Cl 2 , CF 3 Cl, SO 2 Cl. F, F 5 SOSF 5), Xe. F 6 forms Xe 4 F 24 tetramers by fluoride bridging, and perhaps also through weak Xe – Xe interactions. The 24 F atoms are magnetically equivalent, suggesting that they are involved in a rearrangement process ;
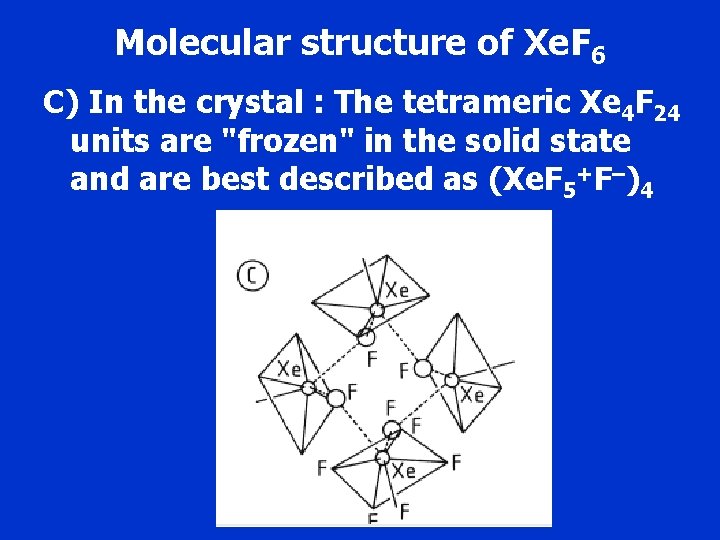
Molecular structure of Xe. F 6 C) In the crystal : The tetrameric Xe 4 F 24 units are "frozen" in the solid state and are best described as (Xe. F 5+F–)4

Uses ØArgon and helium are used in the welding, cutting, and spraying of metals; used in metallurgy as a protective gas. ØNeon: high-voltage tubular lamps ØArgon: mixture with nitrogen, used as filler gas for conventional light bulbs

Uses ØKrypton: used as a better filler gas for high-quality light bulbs, also in halogen lamps ØXenon: gas-discharge lamps, are used as filler gases for lamps, sometimes as constituents of gas mixtures üHigh-purity gases are required for these applications.

Uses • Helium: (1997 Europe) Ø Low-temperature technology 36 % Ø Welding, cutting 14 % Ø Optical fibers 8% Ø Breathing mixtures, diving 6% Ø Analysis 14 % Ø Leak detection 9% Ø Balloons 7% Ø Other uses 6%
Group 18—The Noble Gases • The Group 18 elements are called the noble gases. • This is because they rarely combine with other elements and are found only as uncombined elements in nature. • Their reactivity is very low.

Group 18—The Noble Gases • Helium is less dense than air, so it’s great for all kinds of balloons. • Helium balloons lift instruments into the upper atmosphere to measure atmospheric conditions.

Group 18—The Noble Gases • Even though hydrogen is lighter than helium, helium is preferred for these purposes because helium will not burn.

Uses for the Noble Gases • The “neon” lights you see in advertising signs can contain any of the noble gases, not just neon. • Electricity is passed through the glass tubes that make up the sign.

Uses for the Noble Gases • The electricity causes the gas to glow. • Each noble gas produces a unique color. • Helium glows yellow, neon glows red-orange, and argon produces a bluish-violet color.

Uses for the Noble Gases • Argon, the most abundant of the noble gases on Earth, was first found in 1894. • Krypton is used with nitrogen in ordinary lightbulbs because these gases keep the glowing filament from burning out. • Krypton lights are used to illuminate landing strips at airports, and xenon is used in strobe lights and was once used in photographic flash cubes.

Uses for the Noble Gases • At the bottom of the group is radon, a radioactive gas produced naturally as uranium decays in rocks and soil. • If radon seeps into a home, the gas can be harmful because it continues to emit radiation. • When people breathe gas over a period of time, it can cause lung cancer.

Group 18/0 – The Noble gases Some facts… 1) All of the noble gases have a full outer shell, so they are very _______ 2) They all have low melting and boiling points 3) They exist as single atoms rather then diatomic molecules 4) Helium is lighter then air and is used in balloons and airships (as well as for talking in a silly voice) 5) Argon is used in light bulbs (because it is so unreactive) and argon , krypton and neon are used in fancy lights
- Slides: 32