Periodic Table Dmitri Mendeleev 1837 1907 In 1869

Periodic Table

Dmitri Mendeleev (1837 – 1907) In 1869 Mendeleev and Lothar Meyer (Germany) published nearly identical classification schemes for elements known to date. The periodic table is base on the similarity of properties and reactivities exhibited by certain elements. Later, Henri Moseley ( England, 1887 -1915) established that each elements has a unique atomic number, which is how the current periodic table is organized.
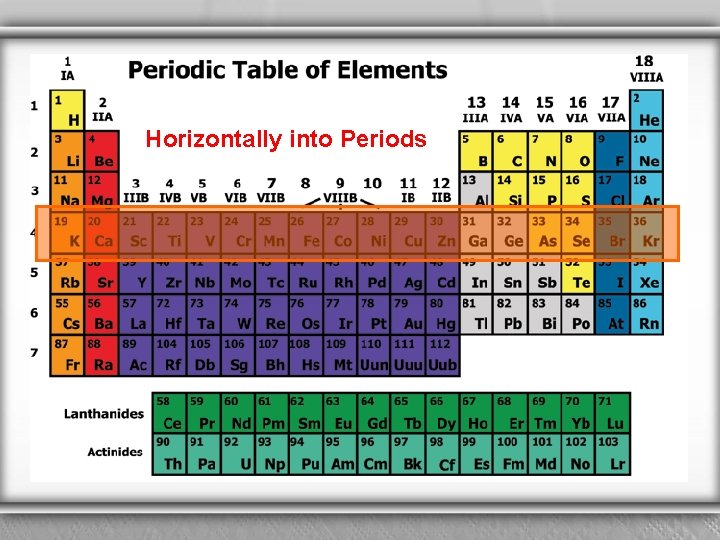
Horizontally into Periods
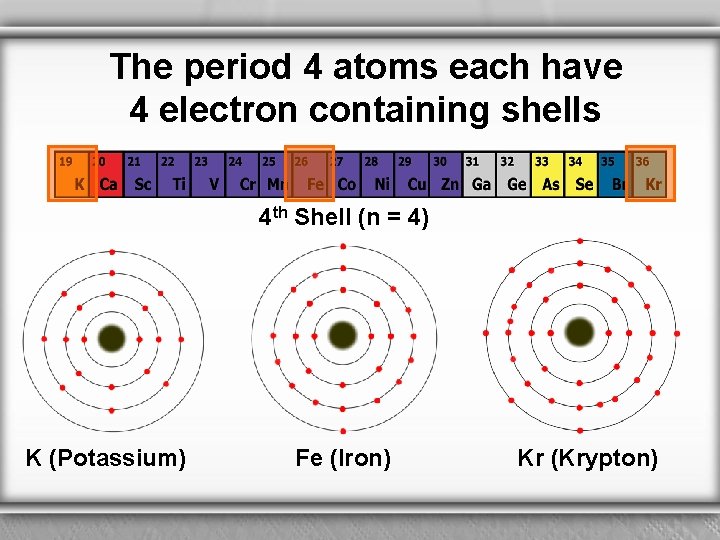
The period 4 atoms each have 4 electron containing shells 4 th Shell (n = 4) K (Potassium) Fe (Iron) Kr (Krypton)
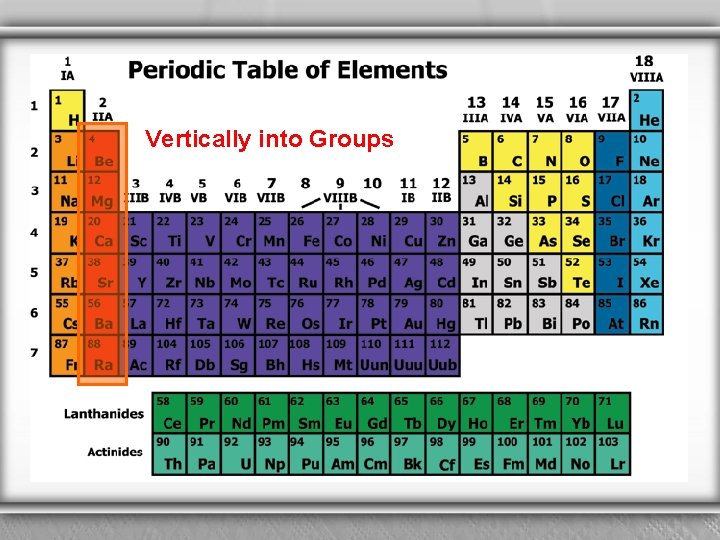
Vertically into Groups
![Each group has distinct properties ]The periodic Table is divided into several groups based Each group has distinct properties ]The periodic Table is divided into several groups based](http://slidetodoc.com/presentation_image_h/0e8f4db38af3e2af6fa7c709a54d5f41/image-6.jpg)
Each group has distinct properties ]The periodic Table is divided into several groups based on the properties of different atoms
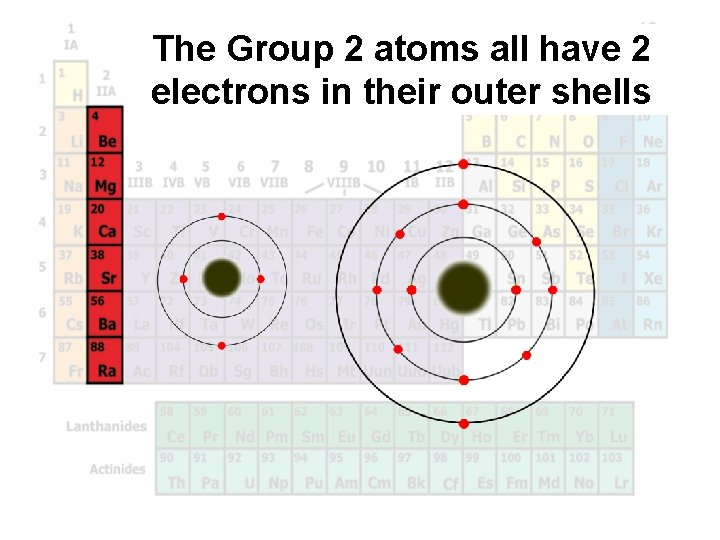
The Group 2 atoms all have 2 electrons in their outer shells
![Alkali Metals ]Soft, silvery coloured metals ]Easy to cut with knife ]Have low densities Alkali Metals ]Soft, silvery coloured metals ]Easy to cut with knife ]Have low densities](http://slidetodoc.com/presentation_image_h/0e8f4db38af3e2af6fa7c709a54d5f41/image-8.jpg)
Alkali Metals ]Soft, silvery coloured metals ]Easy to cut with knife ]Have low densities – All float on water ]Never found free in nature ]Very reactive
![Alkali Metals reacting with oxygen ] Slowly react with oxygen to form oxide ] Alkali Metals reacting with oxygen ] Slowly react with oxygen to form oxide ]](http://slidetodoc.com/presentation_image_h/0e8f4db38af3e2af6fa7c709a54d5f41/image-9.jpg)
Alkali Metals reacting with oxygen ] Slowly react with oxygen to form oxide ] Burn rapidly when heated in oxygen ] Produce coloured flames Lithium + Oxygen Lithium oxide Sodium + Oxygen Sodium oxide Potassium + Oxygen Potassium oxide

Alkali Metals reacting with water • Li (Lithium) • Na (Sodium) • K (Potassium)
![Alkali Metals reacting with water ] React with water to form hydroxide and hydrogen Alkali Metals reacting with water ] React with water to form hydroxide and hydrogen](http://slidetodoc.com/presentation_image_h/0e8f4db38af3e2af6fa7c709a54d5f41/image-11.jpg)
Alkali Metals reacting with water ] React with water to form hydroxide and hydrogen gas Lithium + Water Lithium hydroxide + H 2 Sodium + Water Sodium hydroxide + H 2 Potassium + Water Potassium hydroxide + H 2

Alkali Metals uses • Lithium is used to make alloys and is an anode material for batteries. Used as medication for bipolar disorders. • Potassium is used in fertilizers. • Sodium is used in the food industry as sodium chloride or table salt (Na. Cl) and is used in street lighting to produce a soft orange glow.

Alkaline Earth Metals Silvery-White Metals Fairly reactive Many are found in rocks in the earth’s crust
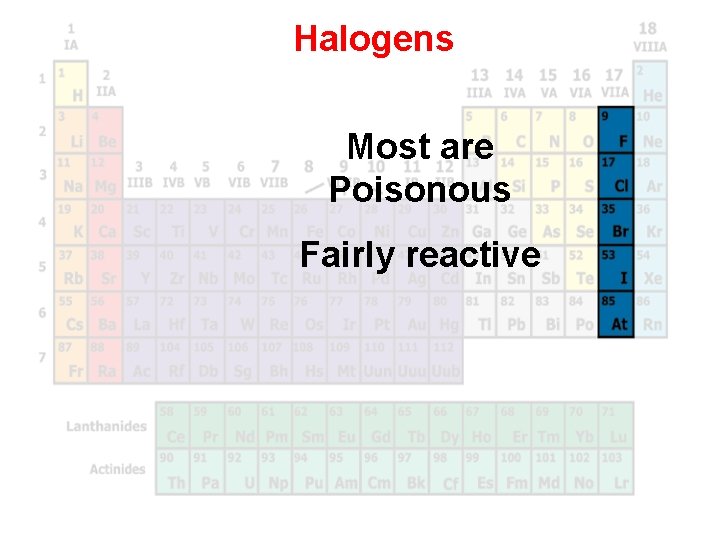
Halogens Most are Poisonous Fairly reactive

Noble Gases Unreactive Gases at room temperature

Jellyfish lamps made with noble gases

Colours noble gases produce in lamp tubes: • He (Helium): pale peach • Ne (Neon): orange-red • Ar (Argon): pale lavender • Kr (Krypton): pale silver • Xe (Xenon): pale, deep blue
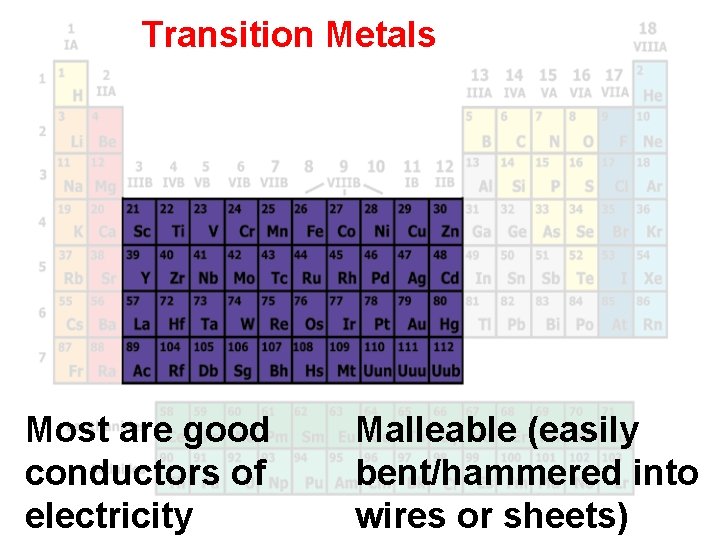
Transition Metals Most are good conductors of electricity Malleable (easily bent/hammered into wires or sheets)


Metalloids lie on either side of these “stairsteps” They share properties with both metals and non-metals Si (Silicon) and Ge (Germanium) are very important “semi-conductors”

What are semiconductors used in?
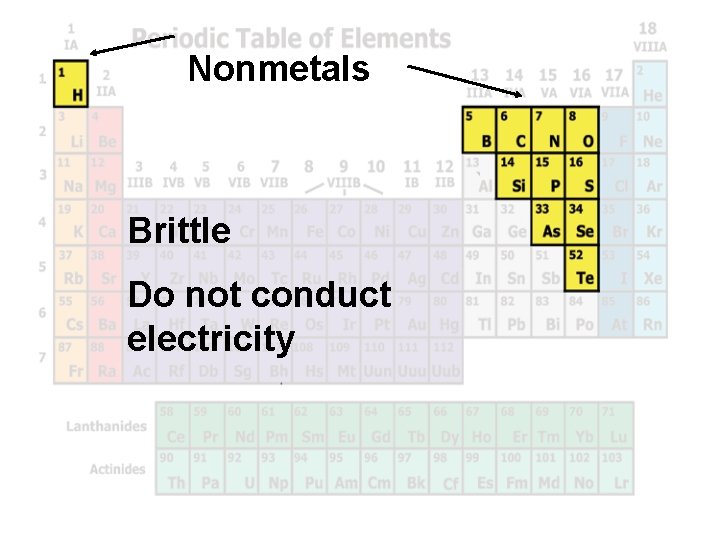
Nonmetals Brittle Do not conduct electricity
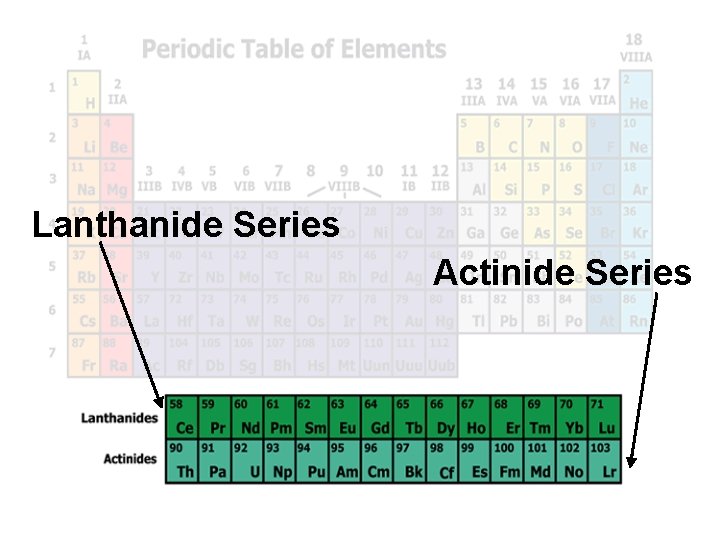
Lanthanide Series Actinide Series
- Slides: 23