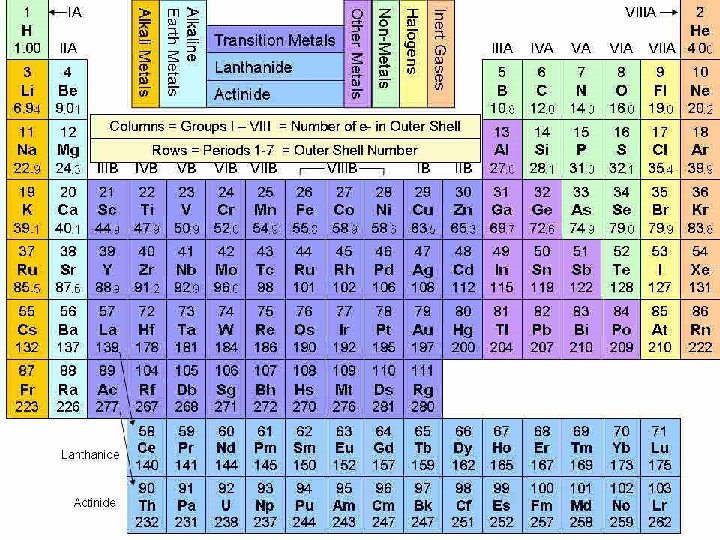
GROUPS OF THE PERIODIC TABLE Nonmetals Noble Gases

GROUPS OF THE PERIODIC TABLE Nonmetals, Noble Gases…
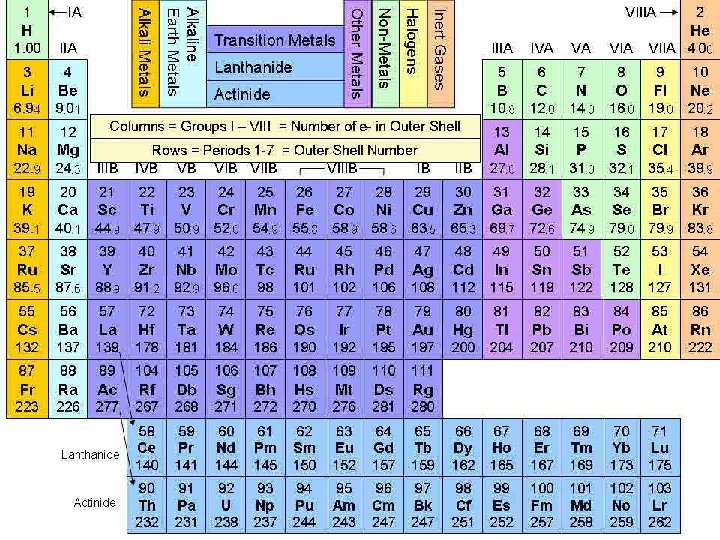
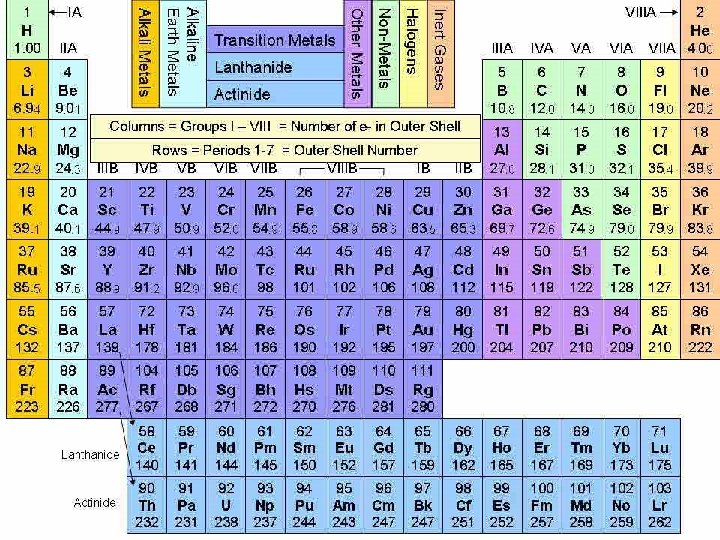
Periodic Table
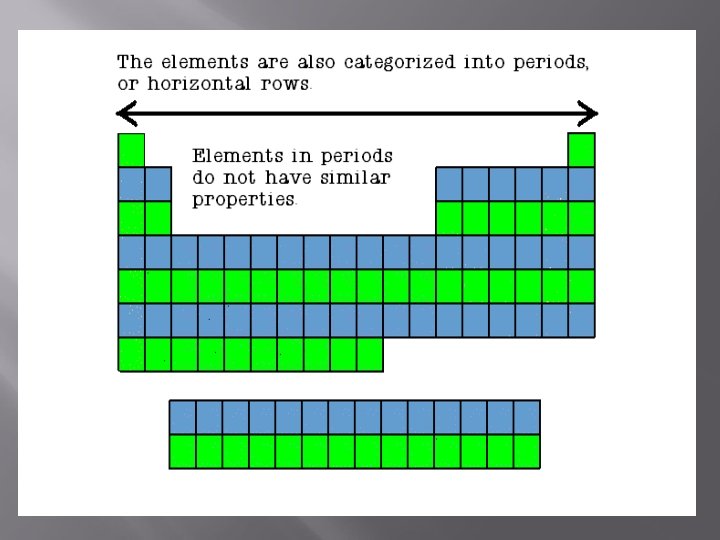
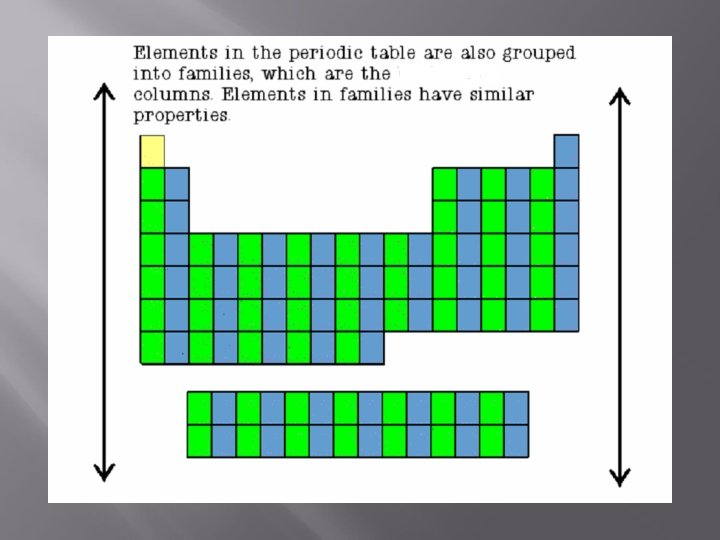

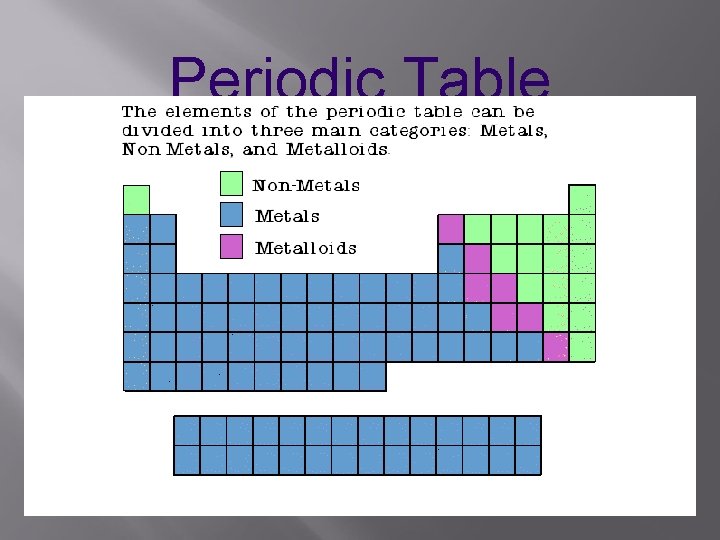
Metals
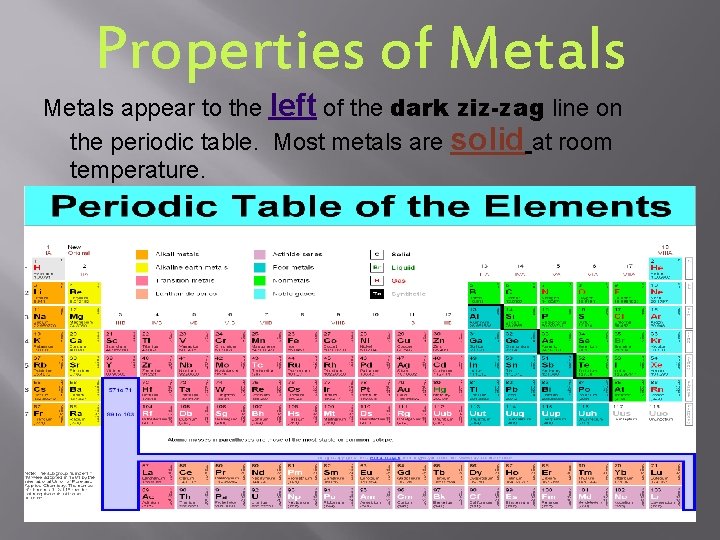
Properties of Metals appear to the left of the dark ziz-zag line on the periodic table. Most metals are solid at room temperature.

Properties of Metals have luster. This means they are shiny

Properties of Metals Ductile metals can be drawn into wire.

Properties of Metals Malleable metals can be hammered into sheets

Properties of Metals have a high also very dense. melting point. They are

Properties of Metals Conductors Metals are good conductors of electricity and heat

When you leave a spoon in a cup of hot drink, the bit poking out of the drink gets hot. Why? Conduction! METALS are the best conductors of heat. This is because the electrons in metals move more freely than in non-metals, allowing the heat energy to travel across the metal. For example, when the spoon touches the hot drink, the heat from the drink excites the electrons in the metal, and the electrons transfer the energy from one electron to another, carrying the heat all the way up the spoon quickly. Best conductors: silver and copper

Physical Properties of METALS Metals are good conductors of electricity. Copper, silver, and gold are good electrical conductors. In a conductor, electric current can flow freely. Since metals have free electrons, they can carry a charge easily. Copper Wiring

Properties of Metals A chemical property of metal is its reaction with water and oxygen. This results in corrosion and rust.
Nonmetals

Properties of Nonmetals occur to the right of the dark zig-zag on the periodic table. Although Hydrogen is in family 1, it is also a nonmetal. Many nonmetals are gases at room temperature.

Properties of Nonmetals do not have luster; they are dull.

Properties of Nonmetals Brittle Nonmetals are brittle so they break easily. This means nonmetals ARE NOT ductile or malleable.

Properties of Nonmetals have low density.
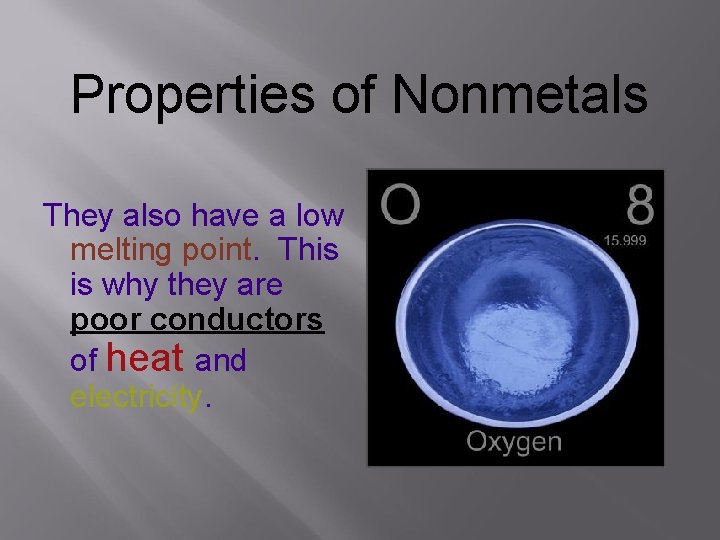
Properties of Nonmetals They also have a low melting point. This is why they are poor conductors of heat and electricity.

METALLOIDS Elements that have properties of metals and nonmetals

These elements are found along the stair-step line that separates the transition metals from the nonmetals

Metalloids

Properties of Metalloids (metal-like) have properties of both metals and nonmetals.

Properties of Metalloids are solids that can be shiny or dull.

Properties of Metalloids They conduct electricity and heat better than nonmetals but not as well as metals.

Properties of Metalloids are malleable and ductile

The most common metalloid is silicon (Si). Silicon combines with oxygen to form silicon dioxide (Si. O 2). Ordinary sand, which is mostly Si. O 2, is the main component of glass. A compound of boron (B) and oxygen is added during the process of glassmaking to make heat-resistant glass. Compounds of boron are also used in some cleaning materials.

The most useful property of the metalloids is their varying ability to conduct electricity. For this reason, metalloids such as silicon, germanium (Ge), and arsenic (As) are used to make semiconductors. Semiconductors are substances that can conduct electricity under some conditions but not under other conditions. Semiconductors are used to make computer chips, transistors, and lasers.

A silicon computer chip is dwarfed by an ant, but the chip’s properties as a semiconductor make it a powerful part of modern computers.

Metalloids can be conductors (like metals) or insulators (like nonmetals). The ability to become either a conductor or an insulator has made metalloids the basis of the computer chip industry and “Silicone Valley. ”

NONMETALS Hydrogen, Halogens, Noble Gases

Diatomic Molecule Def: Elements that are found in nature as two atoms of the same element Examples O 2 N 2 H 2

Hydrogen Atomic number 1 on the periodic table Makes up 90% of all the atoms in the universe. Diatomic molecules– consists of two atoms of the same element Ex. Hydrogen is found in nature as H 2

Hydrogen Alone in the upper left corner of the periodic table is hydrogen—the element with the simplest and smallest atoms. Because the chemical properties of hydrogen differ very much from those of the other elements, it really cannot be grouped into a family. Although hydrogen makes up more than 90 percent of the atoms in the universe, it makes up only 1 percent of the mass of Earth’s crust, oceans, and atmosphere. Hydrogen is rarely found on Earth as a pure element. Most hydrogen is combined with oxygen in water (H 2 O).

Importance of Hydrogen Water is a compound of hydrogen and oxygen. Without liquid water, life on Earth would be impossible.

USES OF HYDROGEN

rocket fuel liquid H 2 is important in cryogenics and in the study of superconductivity since its melting point is only just above absolute zero

Superconducting magnets are some of the most powerful electromagnets known. They are used in : • MRI/NMR machines • massspectrometers • the beam-steering magnets used in particle accelerators

welding

Hydrochloric acid is used in the production of batteries, photoflash bulbs and fireworks. It's even used to process sugar and make gelatin.

hydrogenation of fats and oils

The most abundant element in the universe!

HALOGENS “salt-former. ”

Halogens Found in group 17 of the periodic table Can bond to only one other atom Often bond to Group 1 elements (Alkali Metals) This combination of a metal (alkali - group 1) and a nonmetal is called a salt. All the elements in Group 17 are nonmetals except for astatine, which is a radioactive metalloid

Halogen compounds Silver halides (e. g. silver chloride, silver bromide etc) These are used in photographic paper. They are reduced by light and x-ray radiation to leave a silver photographic image. Hydrogen halides (e. g. hydrogen chloride, hydrogen fluoride) When these dissolve in water they make acids and will turn universal indicator red.

USES OF HALOGENS

FLUORINE F


CHLORINE Cl




BROMINE Br

Flame-Retardant Clothing

IODINE I

NOBLE GASES A group of inert gases that almost never form a compound “inert”

The Noble Gases The elements in Group 18 are known as the noble gases. They do not ordinarily form compounds because atoms of noble gases do not usually gain, lose, or share electrons. As a result, the noble gases are usually unreactive. Even so, scientists have been able to form some compounds of the heavy noble gases (Kr, Xe) in the laboratory.

All the noble gases exist in Earth’s atmosphere, but only in small amounts. Because they are so unreactive, the noble gases were not discovered until the late 1800 s. Helium was discovered by a scientist who was studying not the atmosphere but the sun.

Have you made use of a noble gas? You have if you have ever purchased a floating balloon filled with helium. Noble gases are also used in glowing electric lights. These lights are commonly called neon lights, even though they are often filled with argon, xenon, or other noble gases.

USES OF NOBLE GASES

HELIUM He

Representative Elements 2 Group 18—The Noble Gases • Helium is less dense than air, so it’s great for all kinds of balloons. • Helium balloons lift instruments into the upper atmosphere to measure atmospheric conditions.

Representative Elements 2 Group 18—The Noble Gases • Even though hydrogen is lighter than helium, helium is preferred for these purposes because helium will not burn.

NEON Ne

Representative Elements 2 Uses for the Noble Gases • The “neon” lights you see in advertising signs can contain any of the noble gases, not just neon. • Electricity is passed through the glass tubes that make up the sign.

Representative Elements 2 Uses for the Noble Gases • The electricity causes the gas to glow. • Each noble gas produces a unique color. • Helium glows yellow, neon glows red-orange, and argon produces a bluish-violet color.

ARGON Ar Comes from the Greek word for “lazy, ” as its discoverers were amazed at how they couldn’t get it to do anything…

Representative Elements 2 Uses for the Noble Gases • Argon, the most abundant of the noble gases on Earth, was first found in 1894. • Krypton is used with nitrogen in ordinary lightbulbs because these gases keep the glowing filament from burning out. • Krypton lights are used to illuminate landing strips at airports, and xenon is used in strobe lights and was once used in photographic flash cubes.

Representative Elements 2 Uses for the Noble Gases • At the bottom of the group is radon, a radioactive gas produced naturally as uranium decays in rocks and soil. • If radon seeps into a home, the gas can be harmful because it continues to emit radiation. • When people breathe gas over a period of time, it can cause lung cancer.

Used in containers to replace oxygen, which is highly reactive with certain elements (which ones? ? ? )

Incandescent bulbs are filled with argon to prevent the filaments from oxidizing

THE END!
- Slides: 80