Ionic Bonding The Octet Rule Noble gases are

Ionic Bonding The Octet Rule • Noble gases are unlike any other group of elements on the periodic table because of their extreme stability ( they don’t want to interact with other elements) • Each noble gas has eight valence electrons, except for helium, which has two. • The octet rule says that atoms can become stable by having eight electrons in their outer energy level.

Example • Sodium is in Group 1, so it has 1 valence electron. • Chlorine is in Group 7 A and has 7 valence electrons. What will happen if the two want to combine?

Ionic bonding • How can the valence electrons of atoms rearrange to give each atom a stable configuration of valence electrons? • If the one valence electron of sodium is transferred to the chlorine atom, chlorine becomes stable with an octet of electrons

Electrons Can Be Transferred • Because the chlorine atom now has an extra electron, it has a negative charge. • Also, because sodium lost an electron, it now has an unbalanced proton in the nucleus and therefore has a positive charge.

• Now that each atom has an octet of outerlevel electrons, they are no longer neutral atoms; they are charged particles called ions. • An ion is an atom or group of combined atoms that has a charge because of the loss or gain of electrons.
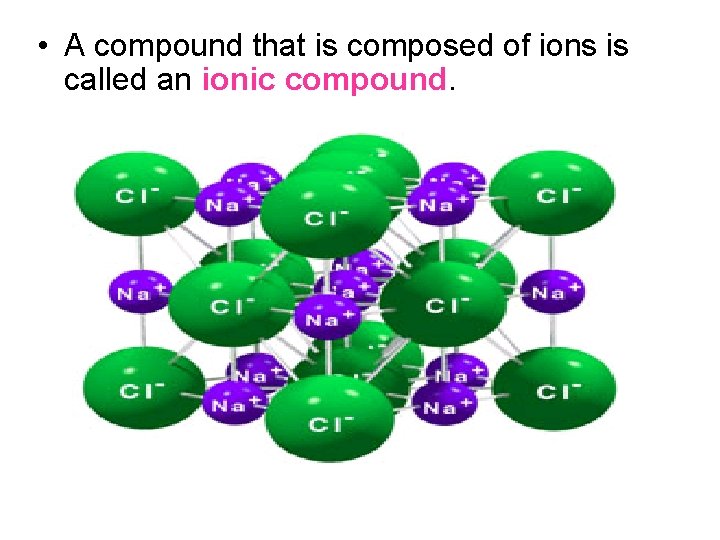
• A compound that is composed of ions is called an ionic compound.
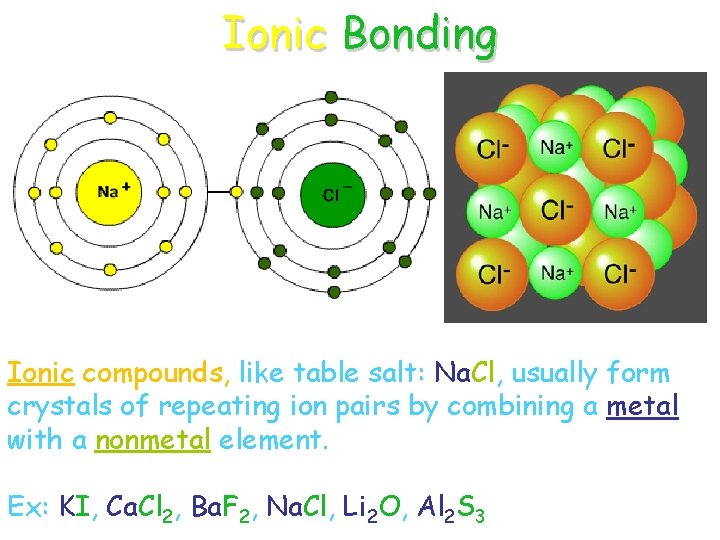
Ionic Bonding Ionic compounds, like table salt: Na. Cl, usually form crystals of repeating ion pairs by combining a metal with a nonmetal element. Ex: KI, Ca. Cl 2, Ba. F 2, Na. Cl, Li 2 O, Al 2 S 3

Ionic bonding • NACL crystals form repeating patterns of positive and negative ions to maximize the distance between like charges • When ionic compounds melt or dissolve in water, their three-dimensional structure breaks apart, and the ions are released from the structure.

Covalent bonding • Now what if the following atoms want to combine? • Cl-Cl • N-N • O-O • C-O
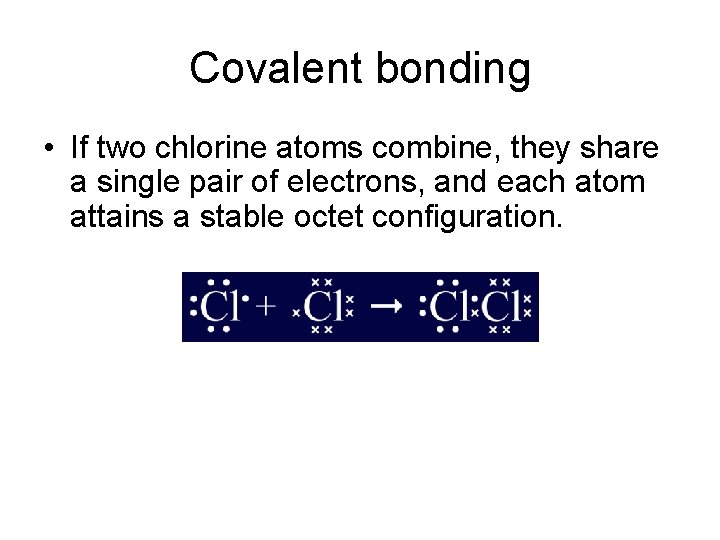
Covalent bonding • If two chlorine atoms combine, they share a single pair of electrons, and each atom attains a stable octet configuration.

Covalent bonding • Two oxygen atoms share two pairs of electrons to form O 2, and two nitrogen atoms share three pairs of electrons to form N 2.

Electron Sharing Produces Molecules • The attraction of two atoms for a shared pair of electrons is called a covalent bond. • Notice that in a covalent bond, atoms share electrons and neither atom has an ionic charge.
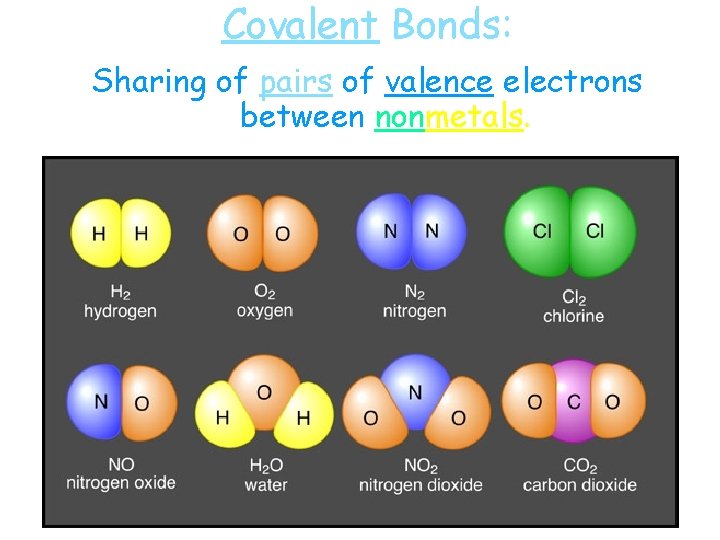
Covalent Bonds: Sharing of pairs of valence electrons between nonmetals.
- Slides: 13