Building Atoms AP Biology On your paper Label

Building Atoms! AP Biology

On your paper • Label Notes 2. 1

Categories • smarties – Electrons • Old candy – Protons • Green beads - neutrons

Build a Helium Atom # protons = atomic number # neutrons = atomic mass rounded – protons # electrons = # protons in stable atoms

Suppose…. What is it called now? • The number of protons change? • The number of electrons change? Ion • The number of neutrons change? Isotope • What is a radioisotope? Radioactive; unstable nucleus
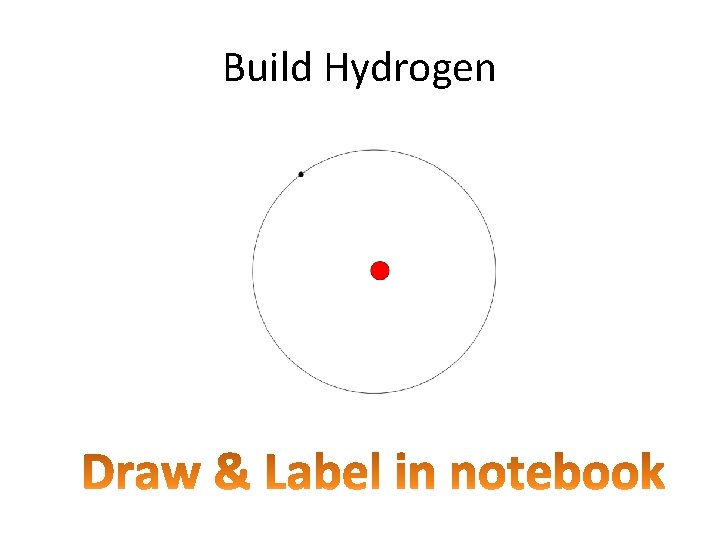
Build Hydrogen

Predict in your notebook… • If Hydrogen gained a proton, what would it be called…. Helium • If hydrogen lost an electron, what would it be called… Hydrogen ion “oxidized” • If hydrogen gained a neutron? Isotope deuterium
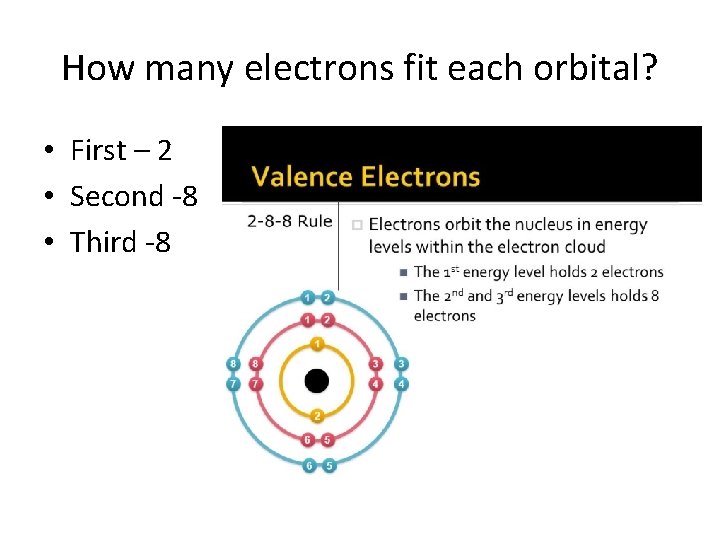
How many electrons fit each orbital? • First – 2 • Second -8 • Third -8
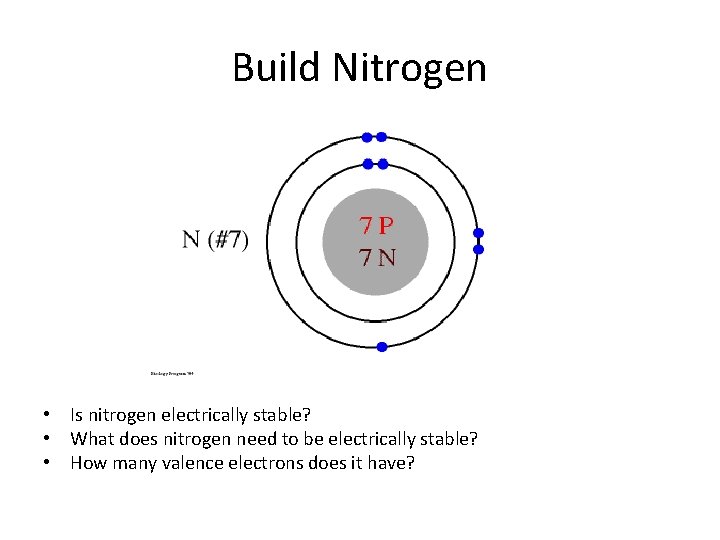
Build Nitrogen • Is nitrogen electrically stable? • What does nitrogen need to be electrically stable? • How many valence electrons does it have?
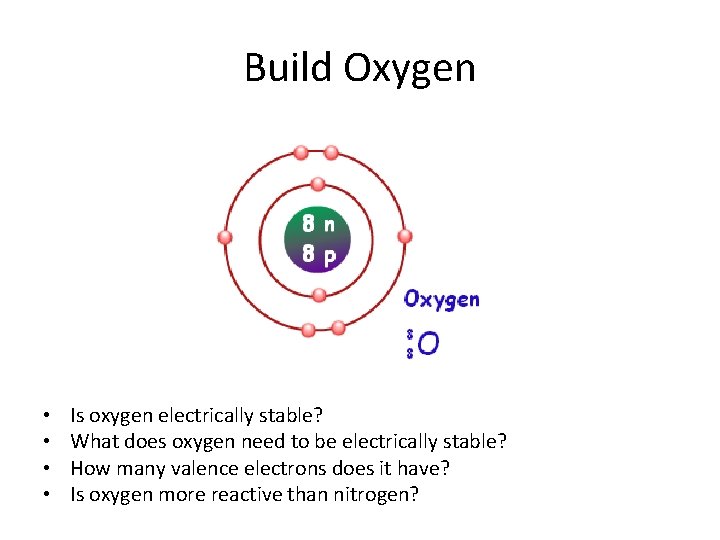
Build Oxygen • • Is oxygen electrically stable? What does oxygen need to be electrically stable? How many valence electrons does it have? Is oxygen more reactive than nitrogen?

Build Fluorine. • How many valence electrons does it have? • Is Fluorine electrically stable? • What does Fluorine need to be electrically stable? • Is fluorine more reactive than nitrogen? Oxygen?

What is electronegativity? • Electronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons • What is the trend of electronegativity on the periodic table? • Electronegativity increases from left to right and decreases from top to bottom down a column • Which elements are the most stable? • Which elements are the most reactive?

PDQ 2. 1

1. Atom vs. Element Atom Element • Basic unit of matter • Composed of protons, neutrons and electrons • Pure substance • Contains only 1 kind of atom • Has unique characteristics that distinguish it from other elements • 94 elements in nature • More than 24 have been made in laboratories

2. 6 elements common in bio • SPONCH – Sulfur – found in certain proteins – Phosphorus – found in DNA – Oxygen – required for aerobic organisms – Nitrogen – found in DNA and protein – Carbon – basic atom in all organic molecules – Hydrogen – also in all organic molecules

3. Describe atom structure • Composed of – Protons – in nucleus, positive charge – Neutrons – in nucleus, no charge – Electrons – in orbitals/electron cloud, negative charge • Most atoms are electrically neutral, which means the number of electrons = number of protons
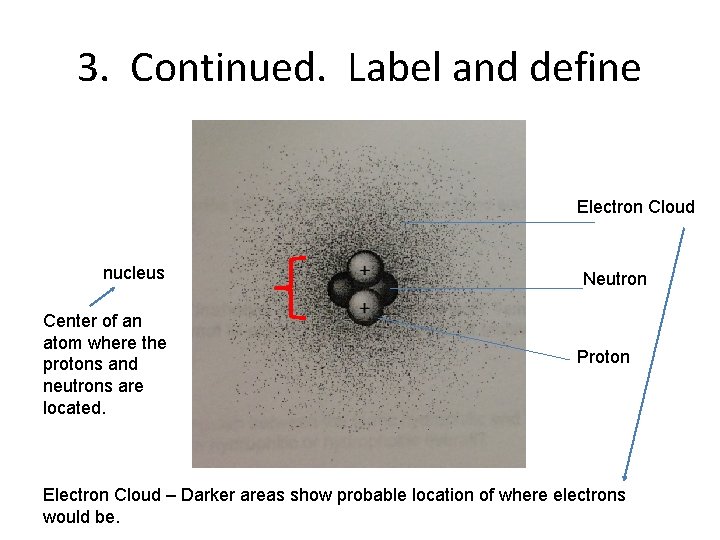
3. Continued. Label and define Electron Cloud nucleus Center of an atom where the protons and neutrons are located. Neutron Proton Electron Cloud – Darker areas show probable location of where electrons would be.

3. Continued. Label and Define electron proton neutron Orbital/ Electron Shell Paths where electrons travel have varying distances from nucleus

4. Atomic Mass vs. Atomic Number Atomic Mass Atomic Number • Protons + Neutrons • Equal to the number of protons • Example – Oxygen has an atomic mass of 16. Because • Unique to each element it has 8 protons and 8 • Oxygen is number 8 and neutrons. therefore has 8 protons. • The mass written on periodic tables refers to the average atomic mass. You would round this number to determine number of neutrons

5. Atomic mass vs. Atomic Weight Atomic Mass Atomic Weight • Mass of a single atom or an individual isotope • Is the average mass of all naturally occuring isotopes of an element • Also called average atomic mass. • This is what is on the periodic table.

6. How to calculate number of neutrons • Number of neutrons = atomic mass (rounded) – number of protons • • For example, nitrogen. Protons = 7 Atomic mass is 14. Number of neutrons = 14 -7 = 7.

7. Isotopes • Have a different number of neutrons than it’s element • Each isotope has a different mass number. • Radioactive isotopes are radioactive and have an unstable nucleus. (They have excess nuclear energy)

8. What determines interactions between atoms? • Number of electrons
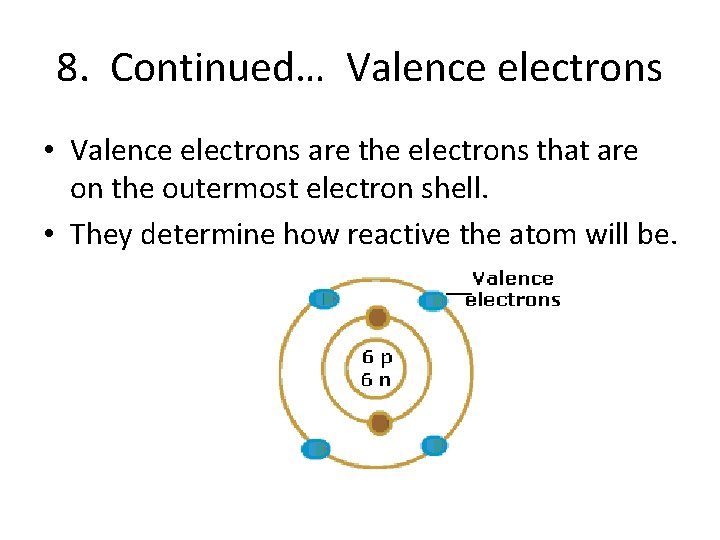
8. Continued… Valence electrons • Valence electrons are the electrons that are on the outermost electron shell. • They determine how reactive the atom will be.
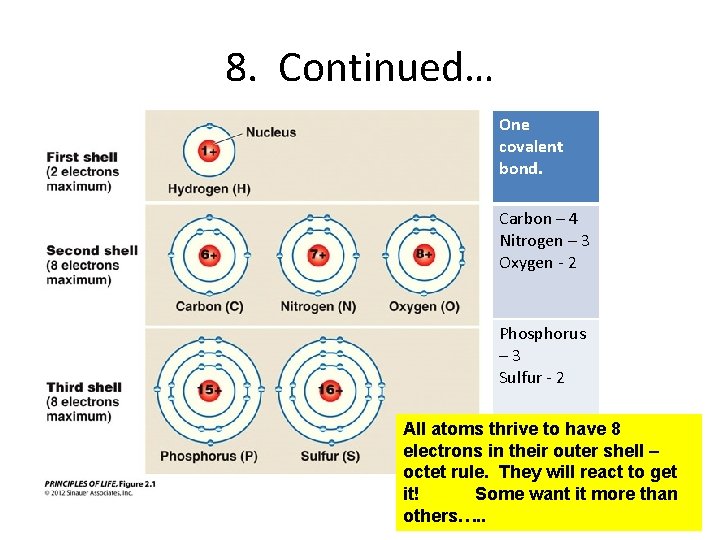
8. Continued… One covalent bond. Carbon – 4 Nitrogen – 3 Oxygen - 2 Phosphorus – 3 Sulfur - 2 All atoms thrive to have 8 electrons in their outer shell – octet rule. They will react to get it! Some want it more than others…. .

Homework • Finish Reading Guide 2. 2
- Slides: 26