Noble Gases Noble Gases He H Li Be

Noble Gases
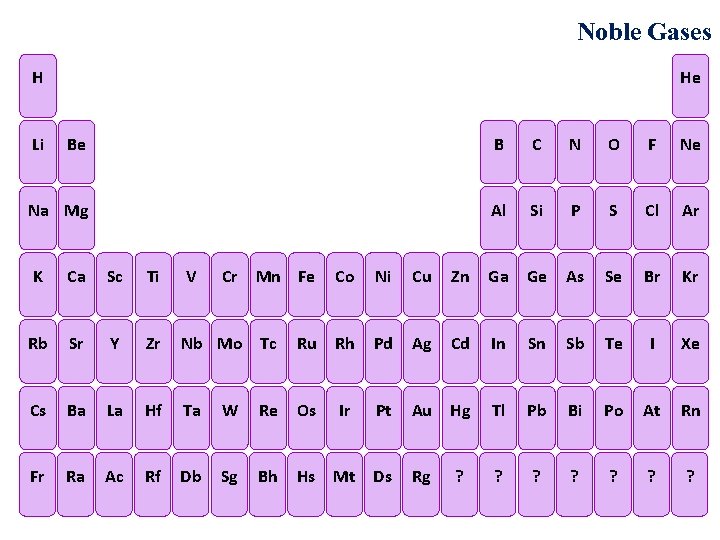
Noble Gases He H Li Be B C N O F Ne Na Mg Al Si P S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Fr Ra Ac Rf Db Sg Bh Hs Mt Ds Rg ? ?
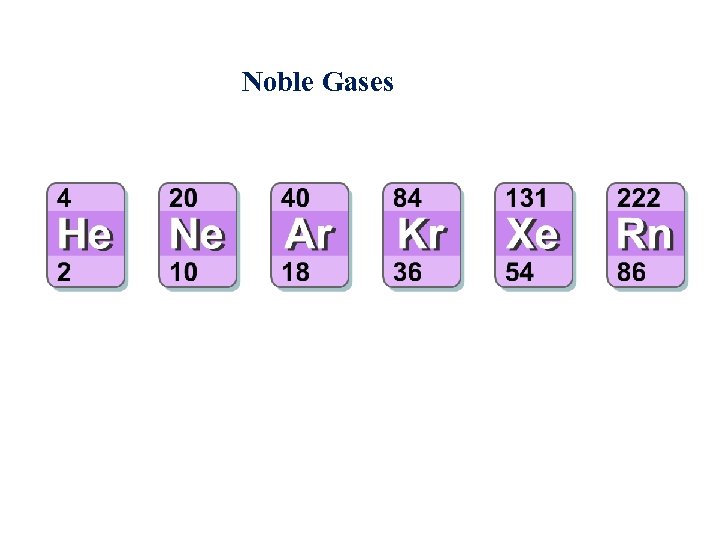
Noble Gases

Why are noble gases so unreactive?

Why NOBLE GASES are inert in nature? Chemical Inertness of these gases is due to following reasons: I. The atoms have stable completely field electronic shells II. They have high ionization energies III. The noble have almost zero electron affinities. Therefore, they do not have any tendency to gain, lose or share electrons with other atoms.
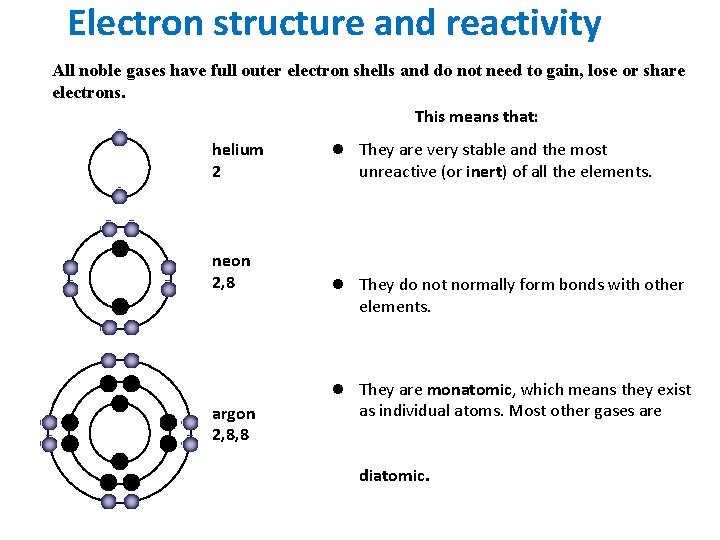
Electron structure and reactivity All noble gases have full outer electron shells and do not need to gain, lose or share electrons. This means that: helium 2 neon 2, 8 argon 2, 8, 8 l They are very stable and the most unreactive (or inert) of all the elements. l They do not normally form bonds with other elements. l They are monatomic, which means they exist as individual atoms. Most other gases are diatomic.

Group 8 becomes group 0 Why is group 0 not called group 8, even though it comes after group 7? It used to be called group 8, and still is in some cases. 8 0 He In the rest of the periodic table, the number of the group is the same as the number of outer shell electrons in the elements of that group. Ne Ar Kr Xe Rn However, this is not true for the noble gases. Helium only has 2 electrons in its outer shell, while the others all have 8. The group’s number was changed to 0 because of this.

Compounds of noble gases • Xe. F 2 • Xe. F 4 • Xe. F 6 • Xe. O 3 • Xe. O 2 F 2 • Xe. OF 4 • Xe. O 4 • Xe. O 3 F 2

Uses of noble gases Although noble gases are unreactive, they are still very useful elements. Many uses of noble gases depend on their ability to prevent other, undesirable, reactions taking place.

Uses of helium Helium is used as: l The gas for inflating balloons and airships, because it is less dense than air and inflammable. l A component of breathing gas (with oxygen) for deep-sea divers, because it is unreactive, insoluble and prevents divers getting ‘the bends’. l A protective gas for growing silicon crystals in silicon chip manufacture, because it is unreactive. l A super-coolant for high-performance magnets, e. g. in body scanners, because it has a very low boiling point (-269 °C).

Uses of neon Neon is used: l In ‘neon’ advertising signs, because it glows red when an electric current is passed through it. l In TV tubes. l In certain types of lasers. l As a cryogenic refrigerant (when liquid).

Uses of argon Argon is used: l In normal light bulbs, because it is unreactive and prevents the tungsten filament from burning. l In energy-efficient fluorescent light bulbs. l As a ‘gas blanket’ for arc welding, because it is unreactive and prevents the hot welding metal from oxidizing.

Uses of other noble gases Krypton is used: l In lasers for eye surgery, to stop bleeding on the retina. l In lighthouses and other types of lamps. Xenon is used: l In various types of electron tubes, lamps and lasers. Radon is used: l To treat cancer by radiotherapy, because it is radioactive. However, because radon is radioactive, it is also an environmental hazard.
- Slides: 13