Anion slow electron velocitymap imaging SEVI applications to

















- Slides: 47

Anion slow electron velocity-map imaging (SEVI): applications to spectroscopy and dynamics Columbus June, 2009

Motivation: spectroscopy and dynamics of transient species n Reactive free radicals play key role in combustion, planetary atmospheres, interstellar chemistry • Map out electronic and vibrational structure, with special focus on vibronic coupling between close-lying electronic states • Optical spectroscopy (LIF, infrared, microwave) are wellestablished probes n Our (complementary) approach: anion photoelectron spectroscopy (PES)and its variants • Slow electron velocity-map imaging (SEVI), a high resolution version of PES

Specific systems: n Open-shell radicals and reactive species (C 3 O, C 3 S, Cn. H, C 2 H 3 O, i. C 3 H 5 O, HCO 2) • Use SEVI to resolve low-frequency vibrational modes, fine-structure, lowlying electronic states • High resolution of SEVI particularly useful for probing vibronic coupling, Duschinsky mixing, internal rotations, …

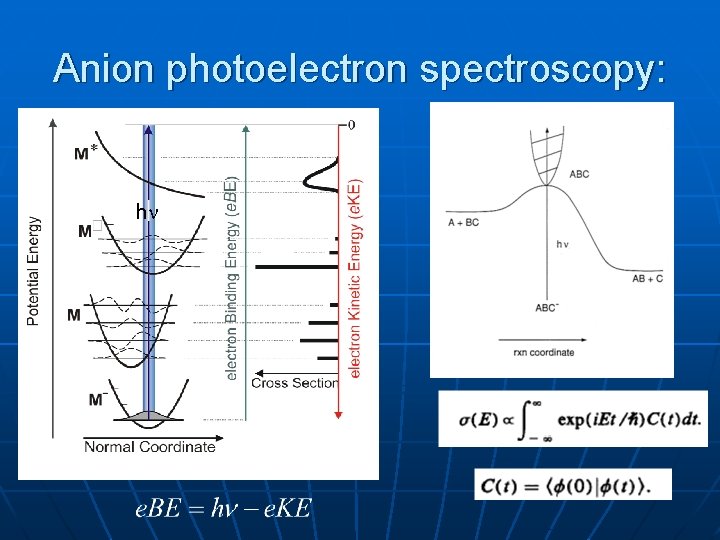
Anion photoelectron spectroscopy: h

Why negative ions? n n n Easy to mass-select Hard to make in large concentrations, but can usually photodetach at h <4 e. V Can access many interesting neutral species by anion photodetachment • Radicals, clusters, transition states…

Selection rules in PES Electronic: all “one-electron” transitions are allowed BN¯: X 2 + (… 2 p 2 p 4) Detachment from 2 p MO X 3 , b 1 states of BN Detachment from 2 p MO a 1 +, A 3 + states of BN Asmis, 1998 So can directly measure a-X singlet-triplet splitting (0. 031 e. V in BN)

Vibrational selection rules: Simplest possible expression-product of 1 -dim Franck-Condon factors No non-adiabatic effects, i. e. for anion, neutral Mode i totally symmetric(sym stretch in CO 2) : any i allowed. “Active” modes: large changes in normal coordinate upon photodetachment Mode i non-totally symmetric (CO 2 bend, antisym stretch) : only even i, but i =0 typically dominates

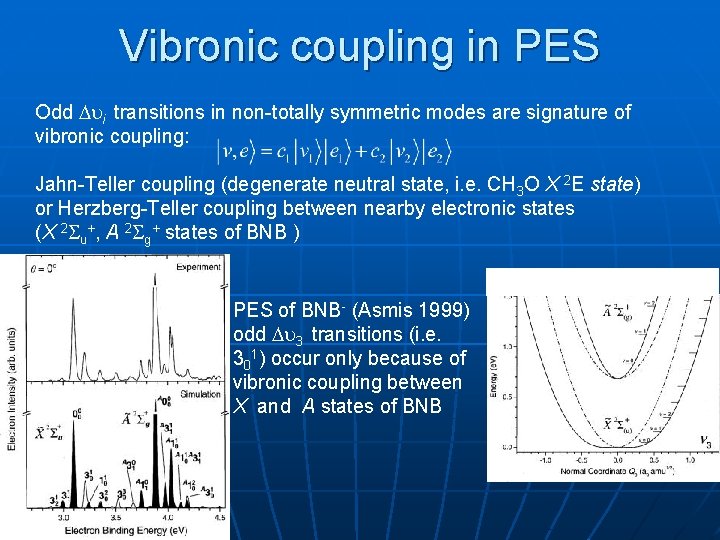
Vibronic coupling in PES Odd i transitions in non-totally symmetric modes are signature of vibronic coupling: Jahn-Teller coupling (degenerate neutral state, i. e. CH 3 O X 2 E state) or Herzberg-Teller coupling between nearby electronic states (X 2 u+, A 2 g+ states of BNB ) PES of BNB- (Asmis 1999) odd 3 transitions (i. e. 301) occur only because of vibronic coupling between X and A states of BNB

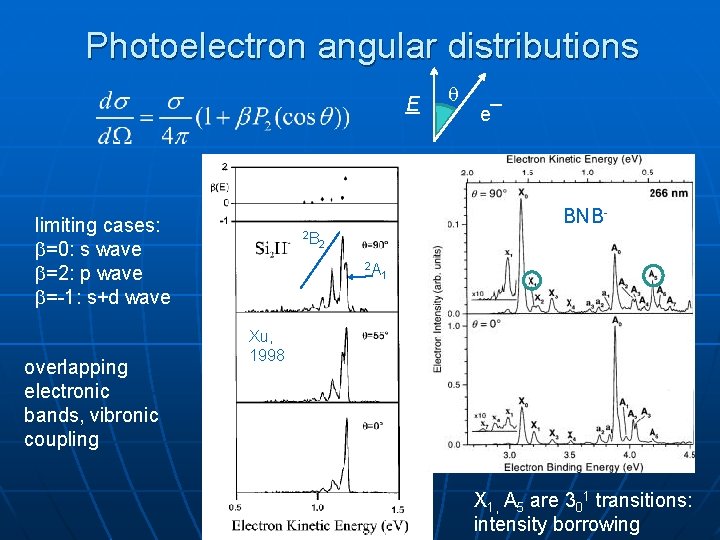
Photoelectron angular distributions E e¯ BNB- limiting cases: =0: s wave =2: p wave =-1: s+d wave overlapping electronic bands, vibronic coupling 2 B 2 2 A 1 Xu, 1998 X 1, A 5 are 301 transitions: intensity borrowing

How to improve resolution? n Photoelectron spectroscopy • Very general, limited to 5 -10 me. V ZEKE SEVI n ZEKE (zero electron kinetic energy) spectroscopy • High resolution (0. 1 -0. 2 me. V) • Experimentally challenging n Fixed h Tunable h SEVI • Resolution comparable to ZEKE without expt’l complications

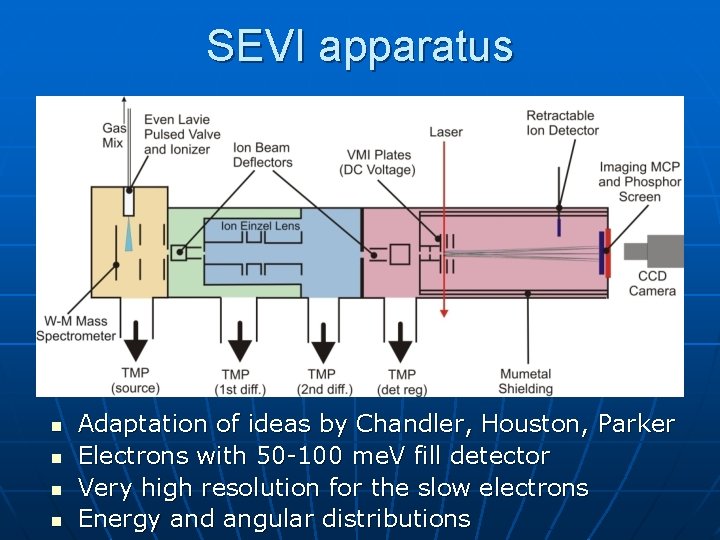
SEVI apparatus n n Adaptation of ideas by Chandler, Houston, Parker Electrons with 50 -100 me. V fill detector Very high resolution for the slow electrons Energy and angular distributions

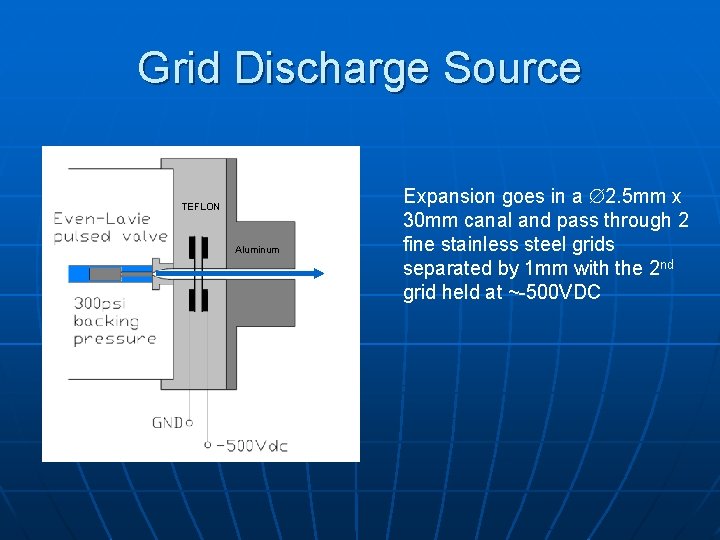
Grid Discharge Source TEFLON Aluminum Expansion goes in a 2. 5 mm x 30 mm canal and pass through 2 fine stainless steel grids separated by 1 mm with the 2 nd grid held at ~-500 VDC

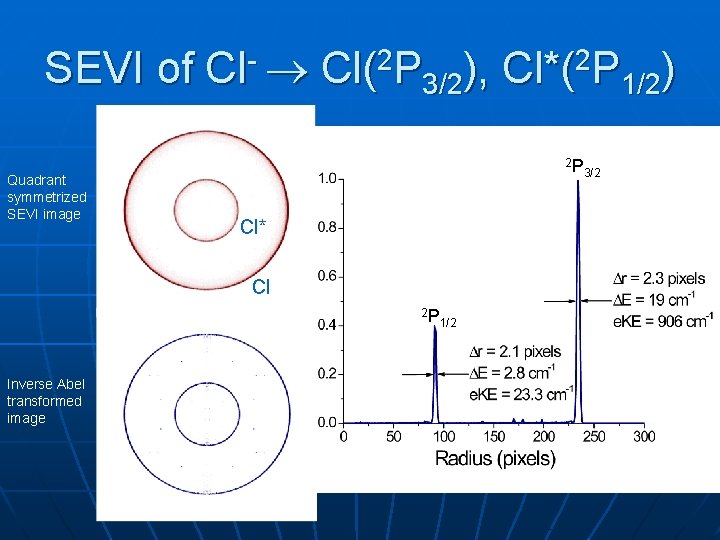
SEVI of Cl- Cl(2 P 3/2), Cl*(2 P 1/2) Quadrant symmetrized SEVI image 2 P Cl* Cl 2 P Inverse Abel transformed image 1/2 3/2

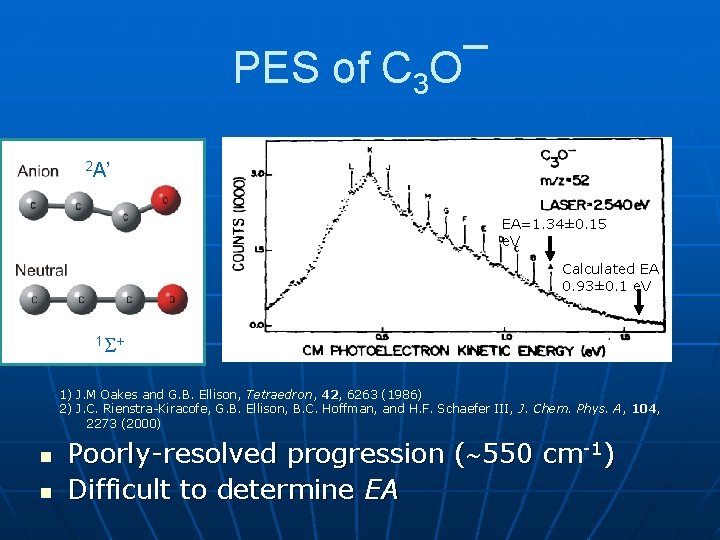
PES of C 3 O¯ 2 A’ EA=1. 34± 0. 15 e. V EA=1. 34 Calculated EA 0. 93± 0. 1 e. V 1 + 1) J. M Oakes and G. B. Ellison, Tetraedron, 42, 6263 (1986) 2) J. C. Rienstra-Kiracofe, G. B. Ellison, B. C. Hoffman, and H. F. Schaefer III, J. Chem. Phys. A, 104, 2273 (2000) n n Poorly-resolved progression ( 550 cm-1) Difficult to determine EA

SEVI of C 3 O¯ and C 3 S¯ Garand, submitted closely spaced doublets photoelectron angular distributions are different

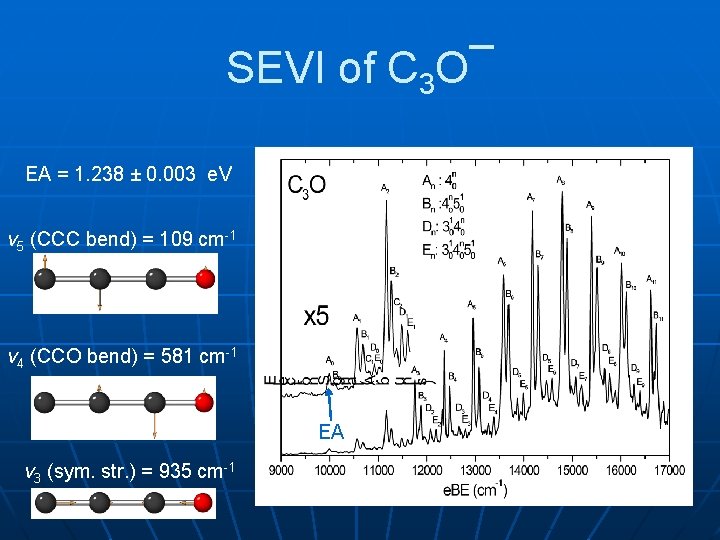
SEVI of C 3 O¯ EA = 1. 238 ± 0. 003 e. V v 5 (CCC bend) = 109 cm-1 v 4 (CCO bend) = 581 cm-1 EA v 3 (sym. str. ) = 935 cm-1

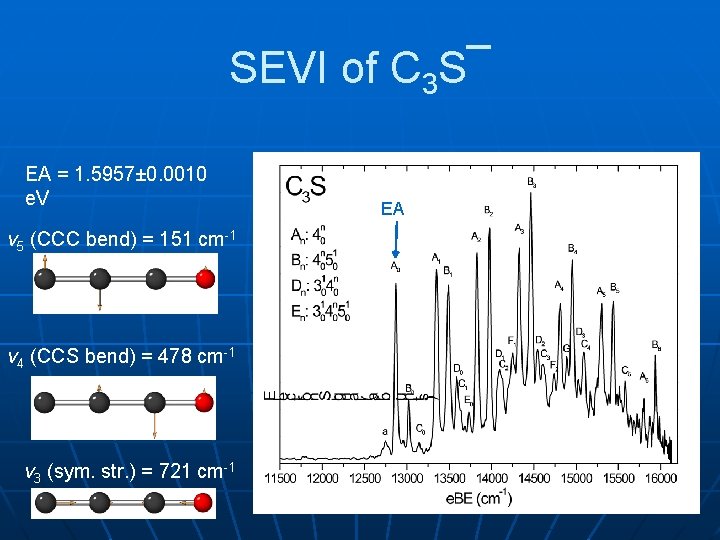
SEVI of C 3 S¯ EA = 1. 5957± 0. 0010 e. V v 5 (CCC bend) = 151 cm-1 v 4 (CCS bend) = 478 cm-1 v 3 (sym. str. ) = 721 cm-1 EA

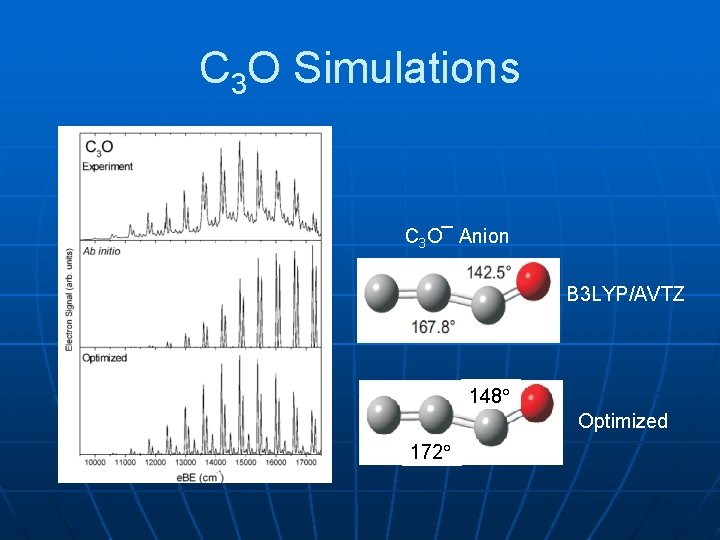
C 3 O Simulations C 3 O¯ Anion B 3 LYP/AVTZ 148 Optimized 172

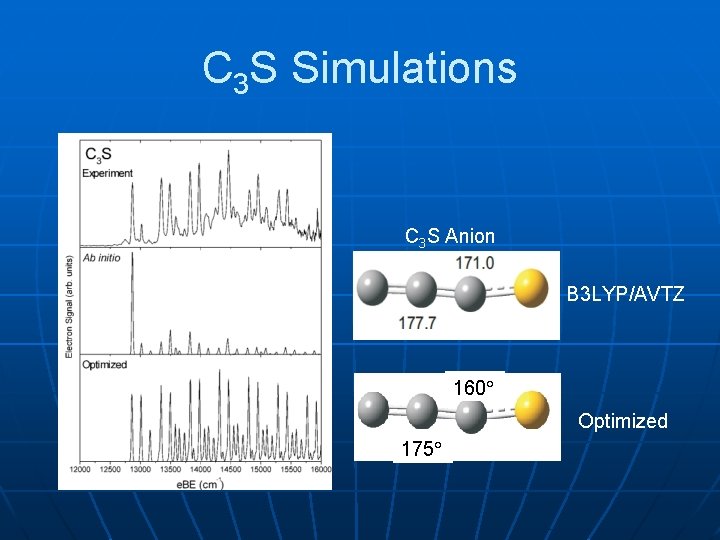
C 3 S Simulations C 3 S Anion B 3 LYP/AVTZ 160 Optimized 175

C 3 O, C 3 S summary n n n C 3 O¯ SEVI spectrum represents dramatic improvement over previous PES Accurate EA’s for C 3 O, C 3 S, several vibrational frequencies determined for first time Analysis still needs work • large-amplitude bending motion in anions, R-T effects

SEVI of Cn. H¯ anions n n n anions and neutrals seen in interstellar medium even n: closely spaced 2 +, 2 states in neutral odd n: evidence for linear and cyclic isomers in anion, neutral Taylor, 1998

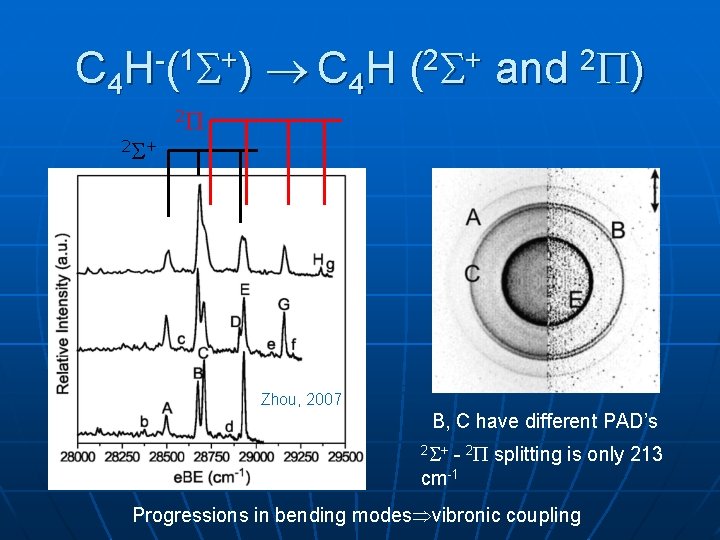
C 4 H-(1 +) C 4 H (2 + and 2 ) 2 2 + Zhou, 2007 B, C have different PAD’s 2 + - 2 splitting is only 213 cm-1 Progressions in bending modes vibronic coupling

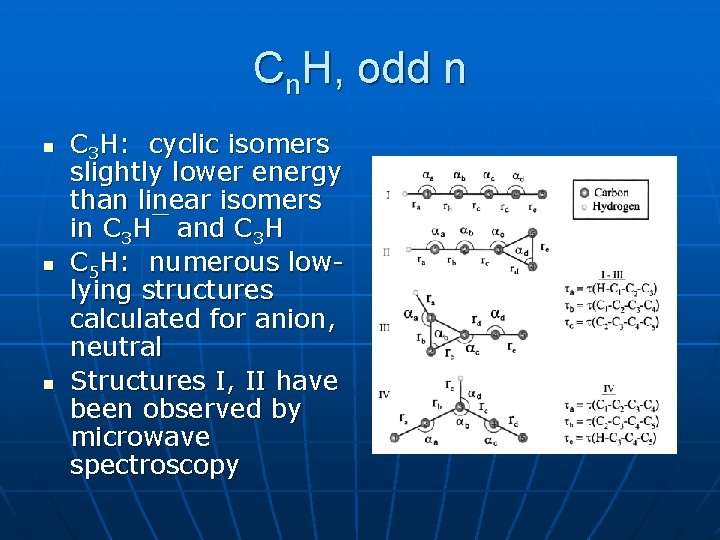
Cn. H, odd n n C 3 H: cyclic isomers slightly lower energy than linear isomers in C 3 H¯ and C 3 H C 5 H: numerous lowlying structures calculated for anion, neutral Structures I, II have been observed by microwave spectroscopy

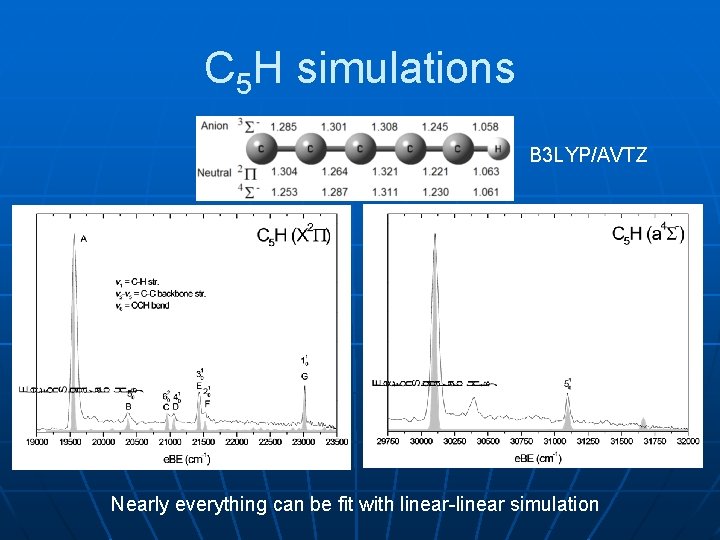
PES/SEVI of C 5 H¯ PES X 0: “linear” linear A 0, B 0: cyclic (Sheehan, 2008) SEVI

C 5 H simulations B 3 LYP/AVTZ Nearly everything can be fit with linear-linear simulation

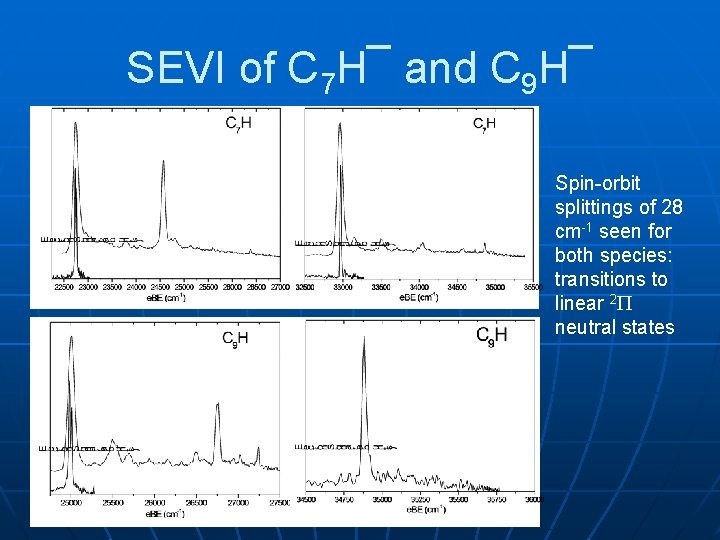
SEVI of C 7 H¯ and C 9 H¯ Spin-orbit splittings of 28 cm-1 seen for both species: transitions to linear 2 neutral states

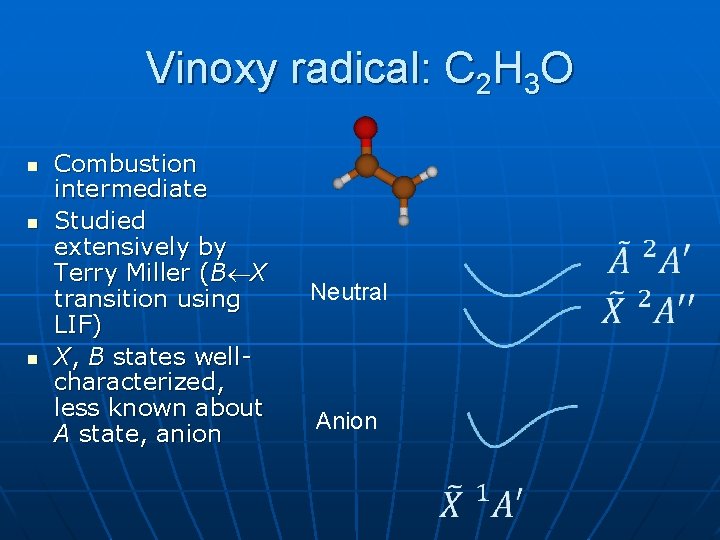
Vinoxy radical: C 2 H 3 O n n n Combustion intermediate Studied extensively by Terry Miller (B X transition using LIF) X, B states wellcharacterized, less known about A state, anion Neutral Anion

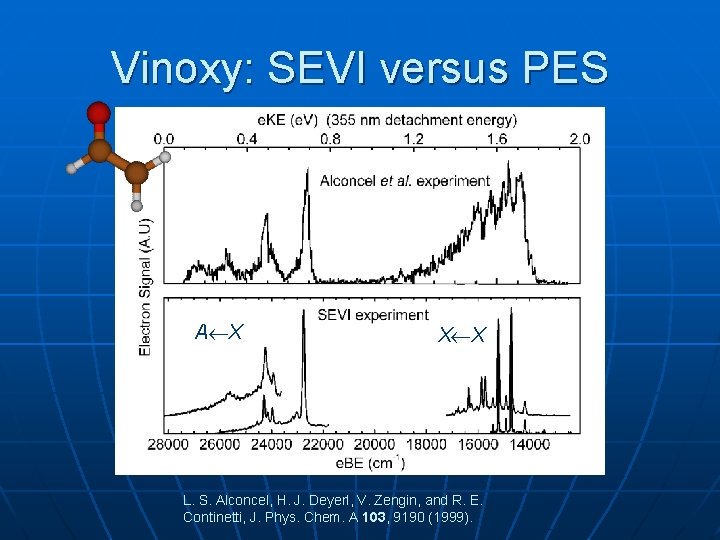
Vinoxy: SEVI versus PES A X X X L. S. Alconcel, H. J. Deyerl, V. Zengin, and R. E. Continetti, J. Phys. Chem. A 103, 9190 (1999).

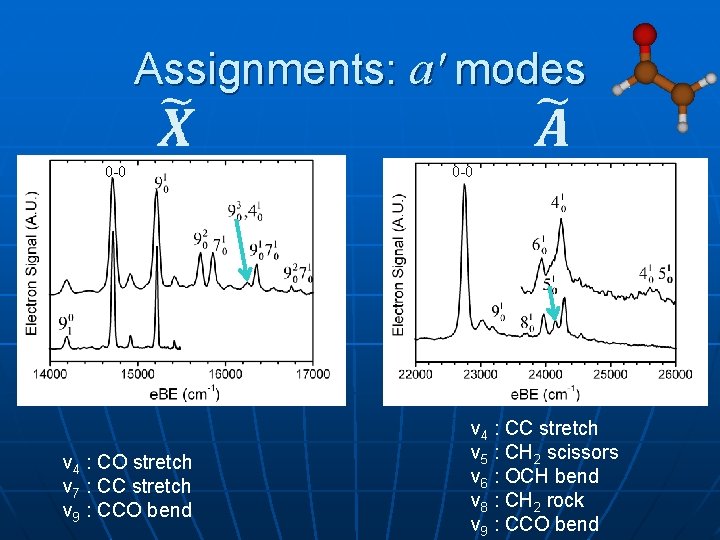
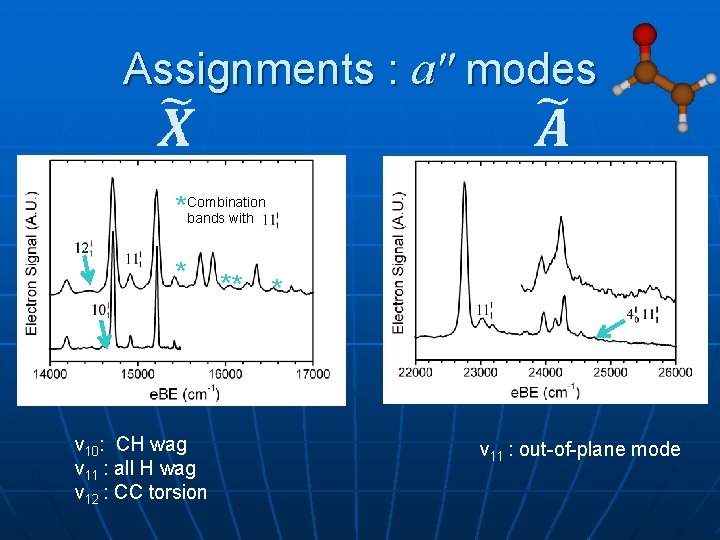
Main Vibrations a' anion radical ν 4: CO stretch exp: ---; 1528; 1533 ν 7: CC stretch ---; 1137; 1533 ν 9: CCO bend 524; 498; 423 a" anion ; radical ; ν 10: CH wag exp: 813; ---; --thy: 939; 942; n/a ν 11: all CH wag 358; ---; --469; 734; n/a ν 12: CH 2 twist 643; ---; --670; 429; n/a

Franck-Condon Simulations n Franck-Condon Factor n Duschinsky Rotation • Q’ and Q : normal coordinates • J : Duschinsky Rotation Matrix : mixing of normal modes • K : mass-weighted geometry change between normal coordinates

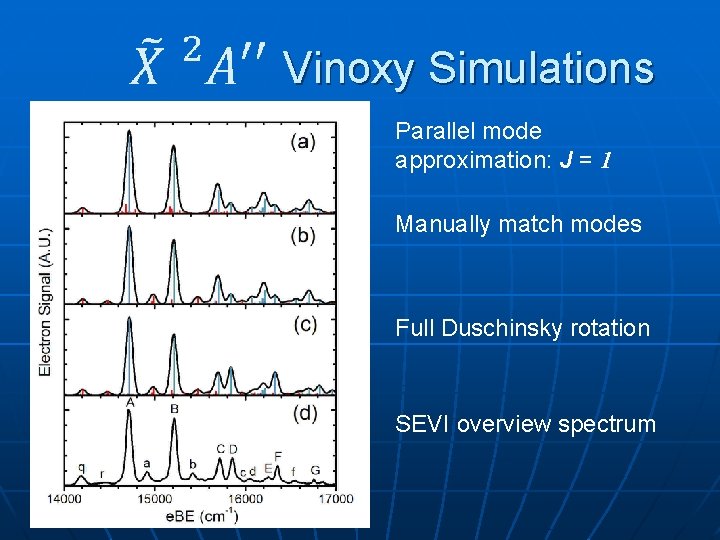
Vinoxy Simulations Parallel mode approximation: J = 1 Manually match modes Full Duschinsky rotation SEVI overview spectrum

Assignments: a' modes 0 -0 ν 4 : CO stretch ν 7 : CC stretch ν 9 : CCO bend 0 -0 ν 4 : CC stretch ν 5 : CH 2 scissors ν 6 : OCH bend ν 8 : CH 2 rock ν 9 : CCO bend

Assignments : a" modes *Combination bands with * v 10: CH wag ν 11 : all H wag ν 12 : CC torsion ** * ν 11 : out-of-plane mode

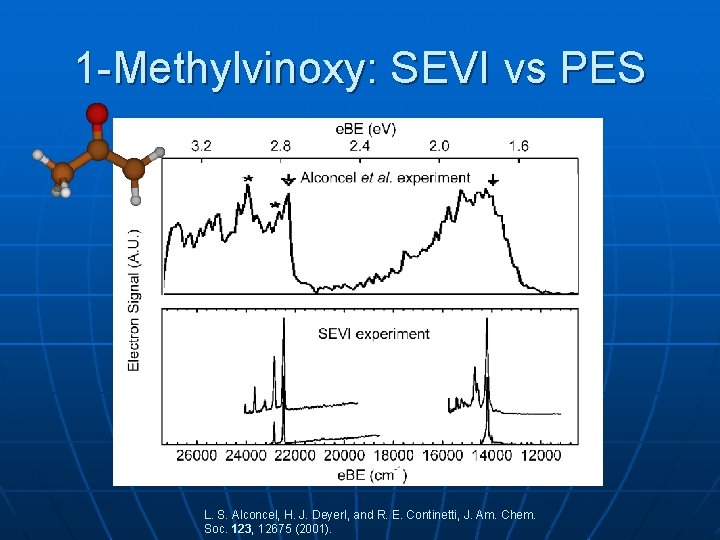
1 -Methylvinoxy: SEVI vs PES L. S. Alconcel, H. J. Deyerl, and R. E. Continetti, J. Am. Chem. Soc. 123, 12675 (2001).

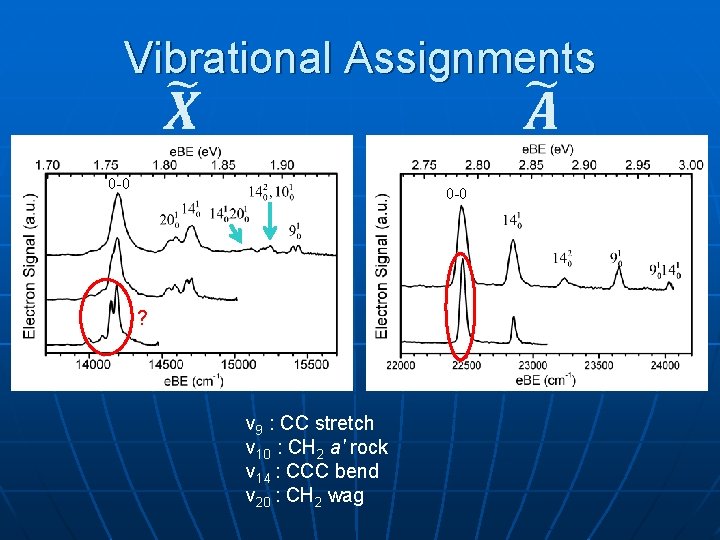
Vibrational Assignments 0 -0 ? ν 9 : CC stretch ν 10 : CH 2 a' rock ν 14 : CCC bend ν 20 : CH 2 wag

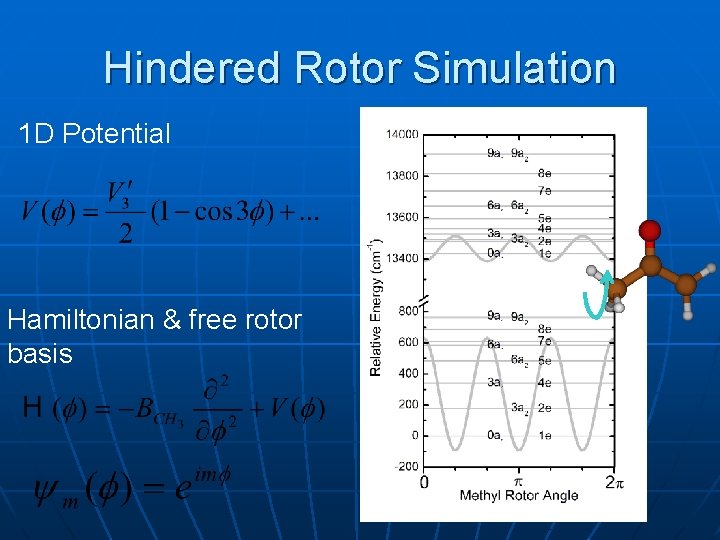
Hindered Rotor Simulation 1 D Potential Hamiltonian & free rotor basis

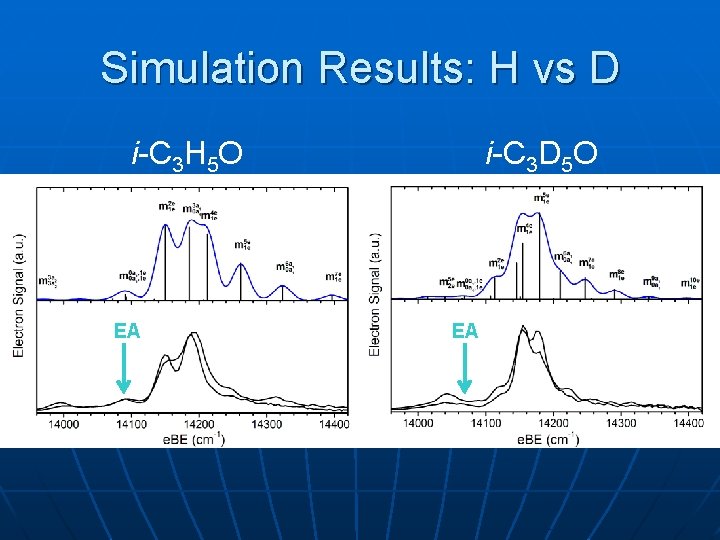
Simulation Results: H vs D i-C 3 H 5 O EA EA i-C 3 D 5 O EA EA?

Vinoxy, methyl-vinoxy: n Precise EA & T 0 Vinoxy 1 -Methylvinoxy n n Electron Afinity First Term Energy 1. 8250 ± 0. 0014 e. V 0. 996 ± 0. 003 e. V 1. 7471± 0. 0018 e. V 1. 039± 0. 003 e. V Vinoxy: new frequencies for A state, Duschinsky rotation needed to simulate X state Methyl-vinoxy: resolve hindered rotor progressions in X band as well as vibrational structure

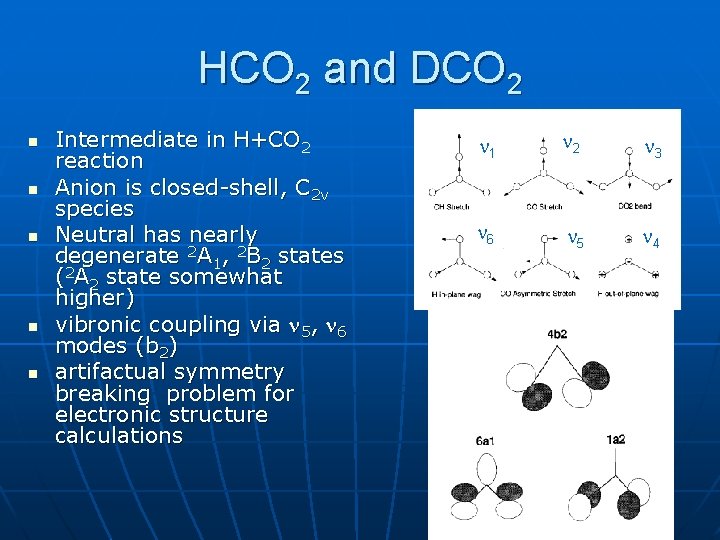
HCO 2 and DCO 2 n n n Intermediate in H+CO 2 reaction Anion is closed-shell, C 2 v species Neutral has nearly degenerate 2 A 1, 2 B 2 states (2 A 2 state somewhat higher) vibronic coupling via 5, 6 modes (b 2) artifactual symmetry breaking problem for electronic structure calculations 1 2 3 6 5 4

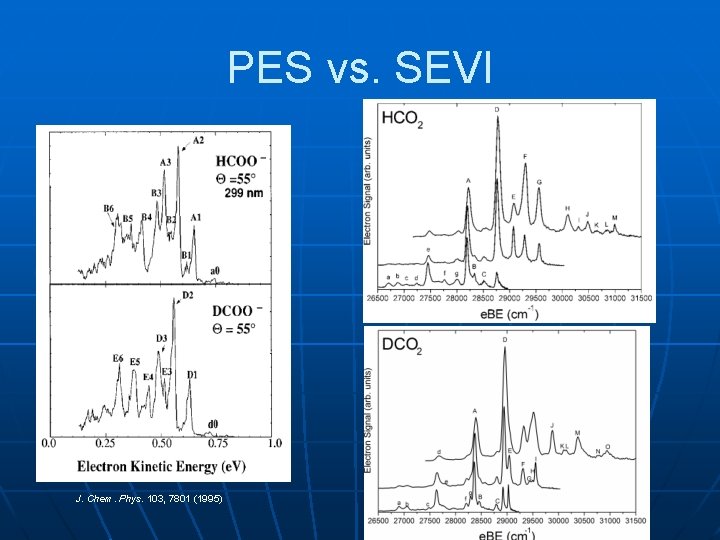
PES vs. SEVI J. Chem. Phys. 103, 7801 (1995)

Results from SEVI: n n n Many more peaks resolved, including hotbands Band A is an apparent origin Peaks exhibit two distinct PAD’s: • For HCO 2, A, D, F, H are p-like • B, C, E, G are s-like n n Implies transitions to two electronic states Sort out with simulations including vibronic coupling, Duschinsky rotation (with John Stanton)

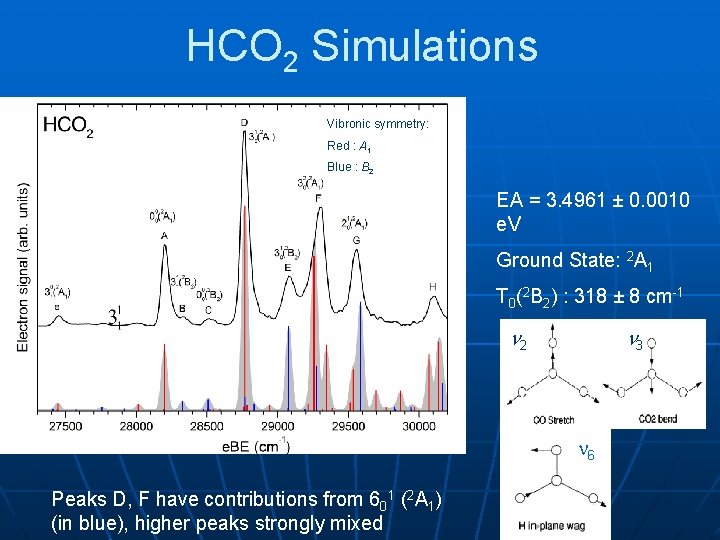
HCO 2 Simulations Vibronic symmetry: Red : A 1 Blue : B 2 EA = 3. 4961 ± 0. 0010 e. V Ground State: 2 A 1 T 0(2 B 2) : 318 ± 8 cm-1 2 3 6 Peaks D, F have contributions from 601 (2 A 1) (in blue), higher peaks strongly mixed

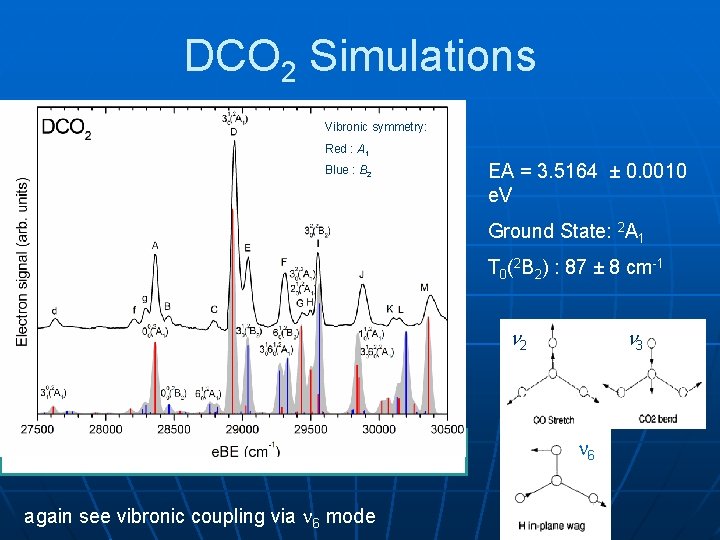
DCO 2 Simulations Vibronic symmetry: Red : A 1 Blue : B 2 EA = 3. 5164 ± 0. 0010 e. V Ground State: 2 A 1 T 0(2 B 2) : 87 ± 8 cm-1 2 3 6 again see vibronic coupling via 6 mode

Summary n SEVI offers “next generation” of anion photodetachment experments • First technique that systematically improves resolution of anion PES without sacrificing (much) generality n Where are we headed? • Bare and complexed metal/semiconductor clusters • Pre-reactive complexes and transition states (in progress) • Cold ions via trapping/cooling • Development of improved methods to simulate spectra beyond simple harmonic analysis

SEVI Group: Etienne Garand Jia Zhou John Stanton Tara Yacovitch Andreas Osterwalder Matt Nee $$$ from AFOSR

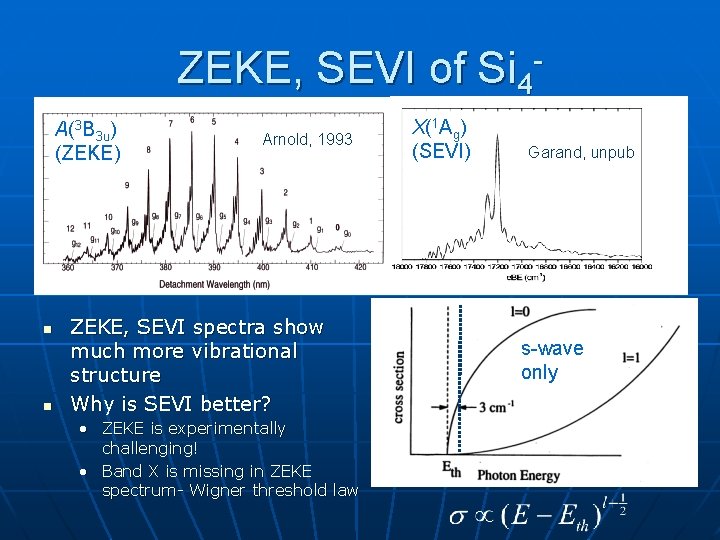
ZEKE, SEVI of Si 4 A(3 B 3 u) (ZEKE) n n Arnold, 1993 ZEKE, SEVI spectra show much more vibrational structure Why is SEVI better? • ZEKE is experimentally challenging! • Band X is missing in ZEKE spectrum- Wigner threshold law X(1 Ag) (SEVI) Garand, unpub s-wave only

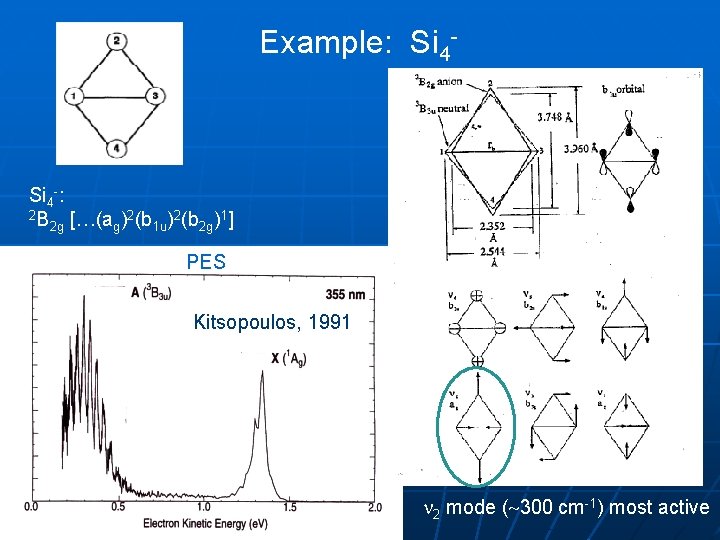
Example: Si 4 -: 2 B 2 2 1 2 g […(ag) (b 1 u) (b 2 g) ] PES Kitsopoulos, 1991 2 mode ( 300 cm-1) most active