ABG Interpretation AcidBase Disorders Jeremy Barnett MD Department

ABG Interpretation & Acid-Base Disorders Jeremy Barnett MD ~ Department of Critical Care Amy Gutman ~ Department of Emergency Medicine

Outline ABG analysis steps Calculating anion gap Calculating delta gap Differentials for specific acid -base disorders A-a Gradients

What Are You Ordering When Requesting An ABG? PO 2 PCO 2 p. H Electrolytes (Na+, K+, Ca++) Lactate HCO 3 Sa. O 2 Other calculated results
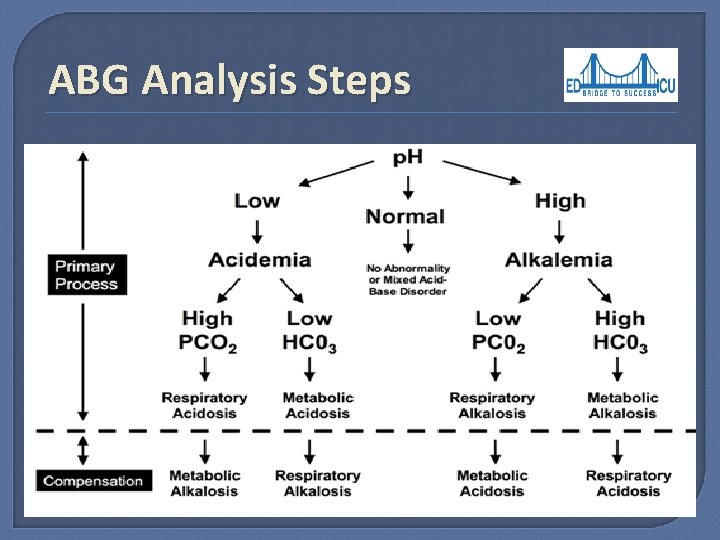
ABG Analysis Steps 1. 2. 3. 4. 5. 6. 7. Is the p. H acidemic or alkalemic? Is the primary disorder respiratory or metabolic? Is the compensation appropriate? Is the compensation acute or chronic? Is there an anion gap? If there is a anion gap, does the delta-delta indicate an additional non-anion gap? What is the differential diagnosis for the clinical process(es)?
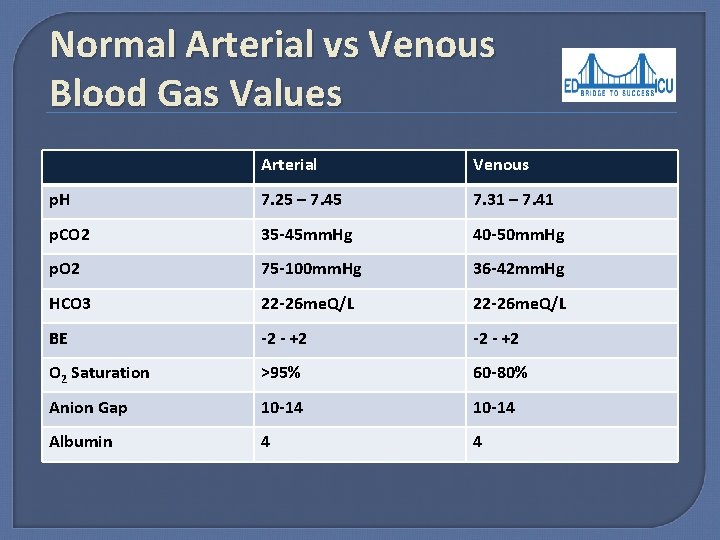
Normal Arterial vs Venous Blood Gas Values Arterial Venous p. H 7. 25 – 7. 45 7. 31 – 7. 41 p. CO 2 35 -45 mm. Hg 40 -50 mm. Hg p. O 2 75 -100 mm. Hg 36 -42 mm. Hg HCO 3 22 -26 me. Q/L BE -2 - +2 O 2 Saturation >95% 60 -80% Anion Gap 10 -14 Albumin 4 4

ABG Interpretation Steps Step 1 • Hypoxia? Step 2 • Acedemia or Alkalemia? Step 3 • Primary Disorder (Respiratory or Metabolic)? Step 4 • Compensation Appropriate, Acute & / or Chronic? Step 5 • Anion Gap? Step 6 • Delta Gap? Step 7 • Differential Diagnosis?

Step 1: Determine Adequacy of Oxygenation (Pa. O 2) Is Patient Hypoxic (Normal: 80 -100 mm. Hg) ? NO YES ? A-a Gradient NORMAL No Defect ? A-a Gradient HIGH Compensated Defect NORMAL Hypoventilation HIGH Other Defect

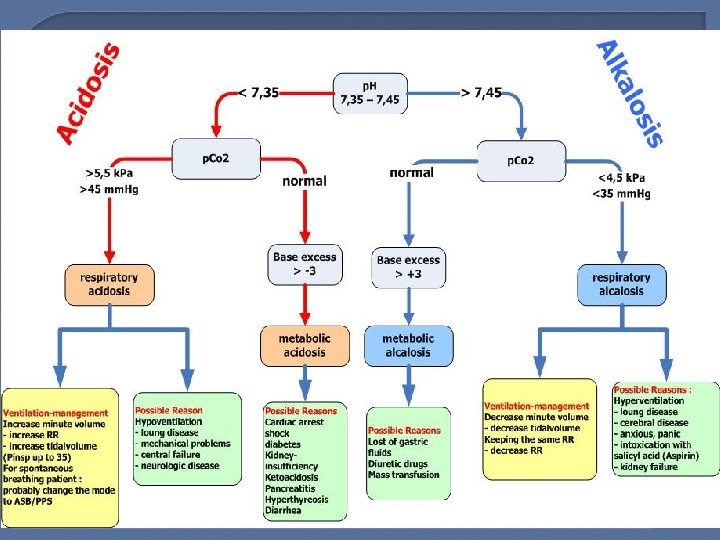
Step 2: Determine p. H (Acidemic vs Alkalemic) Normal p. H range: 7. 35– 7. 45 • p. H <7. 4 • p. H >7. 4 = Acidosis / increased serum hydrogen ion concentration = Alkalosis / decreased serum hydrogen ion concentration Both metabolic & respiratory abnormalities can alter p. H Respiratory & renal function strive to keep p. H = 7. 4 • Minute ventilation responds quickly to metabolic acid-base problems • Renal bicarbonate excretion or retention takes days to compensate for respiratory acid-base problems For our purposes, 3 questions: • Is abnormality respiratory or metabolic? • If respiratory, is it acute or chronic? • If metabolic, is respiratory system responding appropriately?
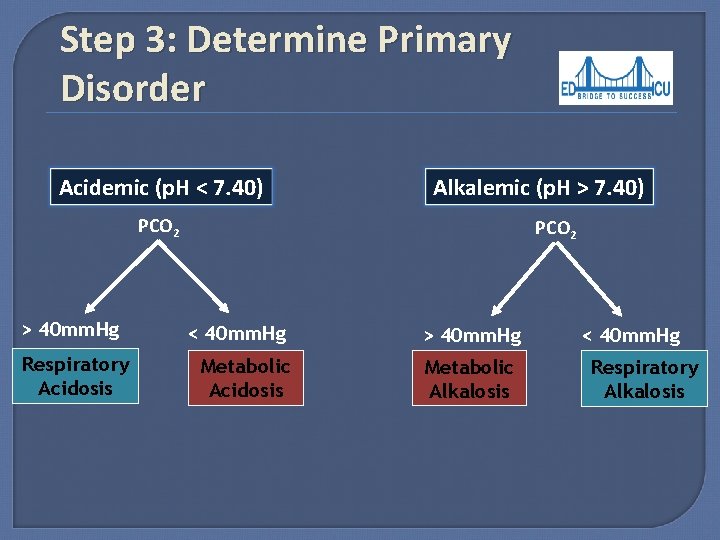
Step 3: Determine Primary Disorder Acidemic (p. H < 7. 40) Alkalemic (p. H > 7. 40) PCO 2 > 40 mm. Hg Respiratory Acidosis PCO 2 < 40 mm. Hg Metabolic Acidosis > 40 mm. Hg Metabolic Alkalosis < 40 mm. Hg Respiratory Alkalosis

Primary Disorder? p. H? p. CO 2 or HCO 3? Respiratory Acidosis p. H low p. CO 2 high Metabolic Acidosis p. H low HCO 3 low Respiratory Alkalosis p. H high p. CO 2 low Metabolic Alkalosis p. H high HCO 3 high • If p. H & PCO 2 going in opposite direction = Respiratory • If p. H & PCO 2 going in same direction = Metabolic • May not work with mixed disorders
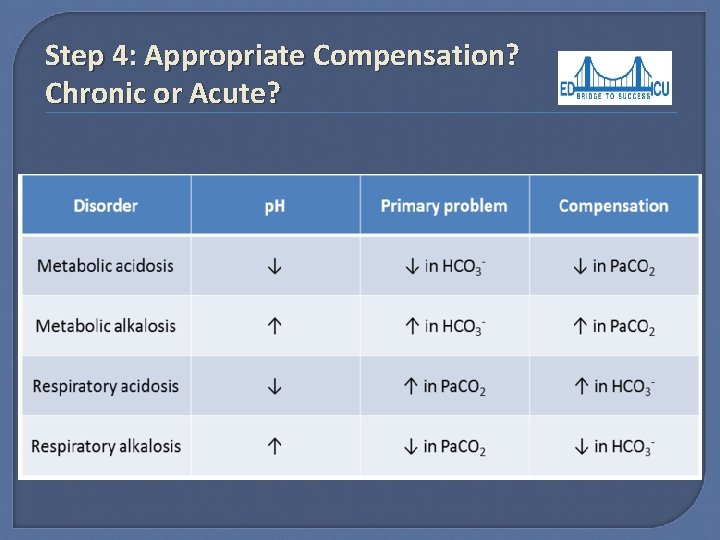
Step 4: Appropriate Compensation? Chronic or Acute? Ø Acute Respiratory Acidosis Ø Ø For every 10 increase in p. CO 2, HCO 3 increases by 1, p. H decreases 0. 08 Ø Acute Respiratory Alkalosis Ø For every 10 decrease in p. CO 2, HCO 3 decreases by 2, p. H increases 0. 08 Chronic Respiratory Acidosis increases by 4, p. H decreases 0. 03 Ø Chronic Respiratory Alkalosis Ø For every 10 decrease in p. CO 2, HCO 3 decreases by 5, p. H increases 0. 03 Ø Partial Compensation: Change in p. H between 0. 03 - 0. 08 for every 10 mm. Hg change in PCO 2

Respiratory Component (Pa. CO 2)? • Normal range: Pa. CO 2 35– 45 m. Hg • Primary respiratory acidosis if p. H <7. 35 & HCO 3– normal • Pa. CO 2 >45 mm. Hg: Respiratory compensation for metabolic alkalosis if p. H >7. 45 & HCO 3– increased • Pa. CO 2 <35 mm. Hg: Primary respiratory alkalosis if p. H >7. 45 & HCO 3– normal • Respiratory compensation for metabolic acidosis if p. H <7. 35 & HCO 3– decreased

Metabolic Component (HCO 3)? Normal HCO 3– range 22– 26 mmol/L HCO 3 <22 mmol/L: Primary metabolic acidosis if p. H <7. 35 • Renal compensation for respiratory alkalosis if p. H >7. 45 HCO 3 >26 mmol/L: Primary metabolic alkalosis if p. H >7. 45 • Renal compensation for respiratory acidosis if p. H <7. 35

Base Excess ~ Degree of Metabolic Disturbance Bicarbonate poor indicator of acid-base disturbance as affected by respiratory & metabolic components Base excess (-2 to +2 m. Eq/L) • Dose of acid or alkali to return blood to normal p. H Examples: • Abnormal base excess with normal AG Normal AG metabolic acidosis (hypercholoremia, GI HCO 3 loss, RTA) • Normal base excess with abnormal AG Lactic acidosis, HAGMA with pre-existing metabolic alkalosis • HAGMA masked by hypoalbuminemia (uncorrected AG) ASA toxicity: respiratory alkalosis + increased AG metabolic acidosis

Appropriateness of Respiratory Response to Metabolic Acidosis Predicted Change in PCO 2 = (1. 5 x HCO 3) + 8 If patient’s PCO 2 is roughly this value, the response is appropriate If patient’s PCO 2 higher than this value, they are failing to compensate

Winter’s Formula ~ Predicting p. CO 2 in Metabolic Acidosis In metabolic acidosis the expected p. CO 2 estimated from HCO 3 Expected p. CO 2 = (1. 5 x HCO 3) + 8 + 2 p. CO 2 (actual) > p. CO 2 (predicted) = additional respiratory acidosis (“mixed acidosis”) p. CO 2 (actual) < p. CO 2 (predicted) = respiratory alkalosis

Step 5: Calculate Anion Gap AG = Na+ – (HCO 3 - + Cl-) Alternative: (Na+ + K) – (HCO 3 - + Cl-) Normal 12 ± 2 If albumin <4 add 2. 5 to AG for every decrease of 1 Corrected = AG + 2. 5 [4 – albumin] If no AG, evaluate for non-anion gap acidosis If there is an AG, calculate delta gap (“excess anion gap”) Calculate delta gap if AG to determine “hidden” metabolic disorders (i. e. non-AG metabolic acidosis, metabolic alkalosis)

Step 6: Calculate Delta Gap Compares increase in anion gap to the decrease in HCO 3 Delta gap = (Actual AG – 12) + HCO 3 Delta gap >30 = additional metabolic alkalosis Delta gap < 18 = additional non-gap metabolic acidosis Delta gap 18– 30 = no additional metabolic disorders

Example Delta Gap Calculation ABG 7. 23 / 17 / 235 on 50% ventimask BMP Na 123 / Cl 97/ HCO 3 7/ Albumin 4 • Delta gap = (actual AG – 12) + HCO 3 = (19 -12) + 7 = 14 • Delta gap <18 = Additional non-AG metabolic acidosis = Metabolic acidosis anion & non-anion gaps

Delta Ratio & Delta Gap ~ High Anion Gap Metabolic Acidosis (HAGMA) Delta Ratio = • < 0. 4 • • • 0. 4– 0. 8 1 -2 >2 Evaluate if high AG metabolic acidosis AG = (AG – 12) HCO 3 = (24 – HCO 3) Hyperchloremic Non-AG metabolic acidosis HAGMA + NAGMA Pure HAGMA Metabolic Acidosis w/ preexisting elevated HCO 3 (metabolic alkalosis or respiratory acidosis) Delta Gap = (Actual AG – 12) + HCO 3 • DG > 30 • DG < 18 • DG 18– 30 Additional metabolic alkalosis Additional non-anion gap metabolic acidosis No additional metabolic disorder

Expected Change in Acid. Base Disorders Primary Disorder Expected Change Metabolic acidosis PCO 2 = 1. 5 × HCO 3 + (8 ± 2) Metabolic alkalosis PCO 2 = 0. 7 × HCO 3 + (21 ± 2) Acute respiratory acidosis Delta p. H = 0. 008 × (PCO 2 - 40) Chronic respiratory acidosis Delta p. H = 0. 003 × (PCO 2 - 40) Acute respiratory alkalosis Delta p. H = 0. 008 × (40 - PCO 2) Chronic respiratory alkalosis Delta p. H = 0. 003 × (40 - PCO 2)

Non-Anion Gap Metabolic Acidosis HARD-UP FUSED-CARS Hyperalimentation, Hydrochloric Acid Fistula Acetazolamide, Addison’s Ureterogastric conduits RTA I, IV, Renal disease Saline administration Diarrhea Endocrine (hyperparathyroid) Uretero-Pelvic Shunt Diarrhea Post hypocapnia Carbonic Anhydrase inhibitors RTA Spironolactone • For non-AG metabolic acidosis, calculate urine anion gap • UAG = UNa+ + UK+ – UCL • If UAG>0: renal problem • If UAG<0: non-renal problem

Anion Gap Metabolic Acidosis CAT - MUDPILES Carbon monoxide, Cyanide, Congenital heart failure Aminoglycosides Tolulene, Theophylline Methanol Uremia DKA, alcoholic ketosis, starvation ketosis Propylene glycol, Paracetamol, Paraldehyde Isoniazid, Iron, Inborn errors of metabolism Lactic acidosis Ethylene glycol, Ethanol Salicylates

Osmolar Gap Elevated osmolar gap >10 provides indirect evidence for presence of an abnormal low molecular weight solute (e. g. ethanol, methanol & ethylene glycol) Osmolar gap = Osmolality (m. Osm/kg) – Osmolarity (m. Osm/L) Osmolality is measured, Osmolarity is calculated • Osmolarity = (1. 86 x Na+) + glucose + urea + 9 (mmol/l) • Osmolarity = (1. 86 x Na+) + glucose/18 + BUN/2. 8 + 9 (mg/dl)
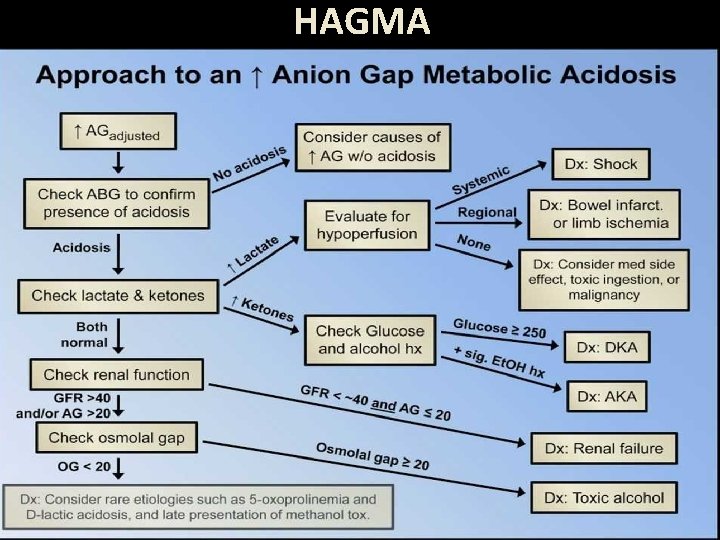
HAGMA
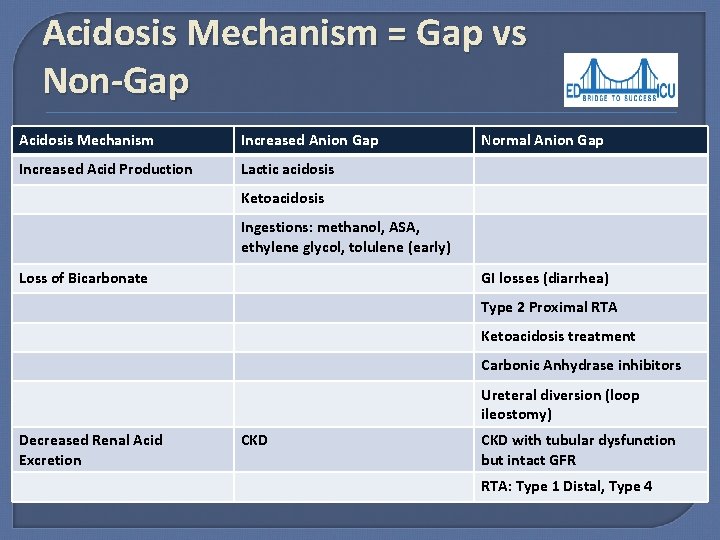
Acidosis Mechanism = Gap vs Non-Gap Acidosis Mechanism Increased Anion Gap Increased Acid Production Lactic acidosis Normal Anion Gap Ketoacidosis Ingestions: methanol, ASA, ethylene glycol, tolulene (early) Loss of Bicarbonate GI losses (diarrhea) Type 2 Proximal RTA Ketoacidosis treatment Carbonic Anhydrase inhibitors Ureteral diversion (loop ileostomy) Decreased Renal Acid Excretion CKD with tubular dysfunction but intact GFR RTA: Type 1 Distal, Type 4

Metabolic Alkalosis Determine urinary Cl- to differentiate saline responsive vs saline resistant Must be off diuretics in order to interpret urine chloride Saline Responsive UCl-<10 Saline-Resistant UCl- >10 Vomiting If HTN: Cushings, Conn’s, RAS, renal failure with alkali administration NG suction If not HTN: hypomagnesemia, hypokalemia, Bartter’s, Gittelman’s, licorice ingestion Over-diuresis Exogenous corticosteroids Post-hypercapnia
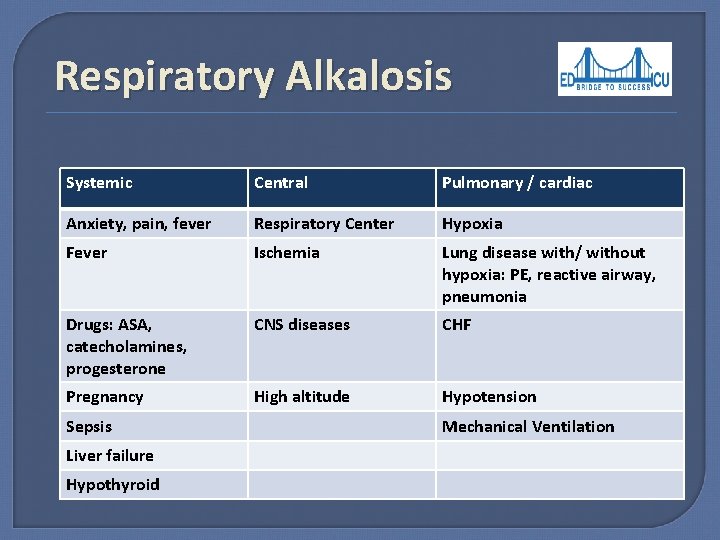
Respiratory Alkalosis Systemic Central Pulmonary / cardiac Anxiety, pain, fever Respiratory Center Hypoxia Fever Ischemia Lung disease with/ without hypoxia: PE, reactive airway, pneumonia Drugs: ASA, catecholamines, progesterone CNS diseases CHF Pregnancy High altitude Hypotension Sepsis Liver failure Hypothyroid Mechanical Ventilation

Respiratory Acidosis CNS depression: sedatives, narcotics, CVA Neuromuscular disorders: acute or chronic Acute airway obstruction Severe pneumonia, pulmonary edema, pleural effusion Chest cavity problems: hemothorax, pneumothorax, flail chest Chronic lung disease: obstructive or restrictive Central hypoventilation, OSA

Alveolar–Arterial (A-a) Gradient Measure of difference between alveolar & arterial O 2 concentration • Normal = 5 -10 mm. Hg, plus age-related changes A-a Gradient = PAO 2 – Pa. O 2 Normal < [Age/ 4] + 4 Hypoxemia DDX by assessing integrity of alveolar capillary unit In high altitude, arterial O 2 low but only because alveolar O 2 also low In ventilation perfusion mismatch (PE, right-to-left shunt), O 2 not effectively transferred from alveoli to blood resulting in elevated A-a gradient
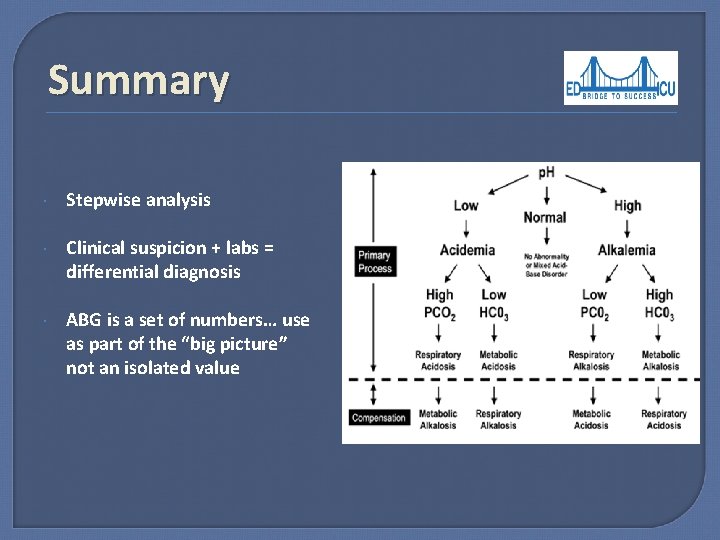
Summary Stepwise analysis Clinical suspicion + labs = differential diagnosis ABG is a set of numbers… use as part of the “big picture” not an isolated value
- Slides: 32