Blood Gas Analysis and its Clinical Interpretation Dr

Blood Gas Analysis and it’s Clinical Interpretation Dr R. S. Gangwar MD, PDCC, FIPM Assistant Professor Geriatric ICU, DGMH

Outline 1. 2. 3. 4. 5. 6. Common Errors During ABG Sampling Components of ABG Discuss simple steps in analyzing ABGs Calculate the anion gap Calculate the delta gap Differentials for specific acid-base disorders

Delayed Analysis Consumptiom of O 2 & Production of CO 2 continues after blood drawn ØIced Sample maintains values for 1 -2 hours ØUniced sample quickly becomes invalid within 15 -20 minutes Pa. CO 2 3 -10 mm. Hg/hour p. H d/t lactic acidosis generated by glycolysis in R. B. C. Pa. O 2
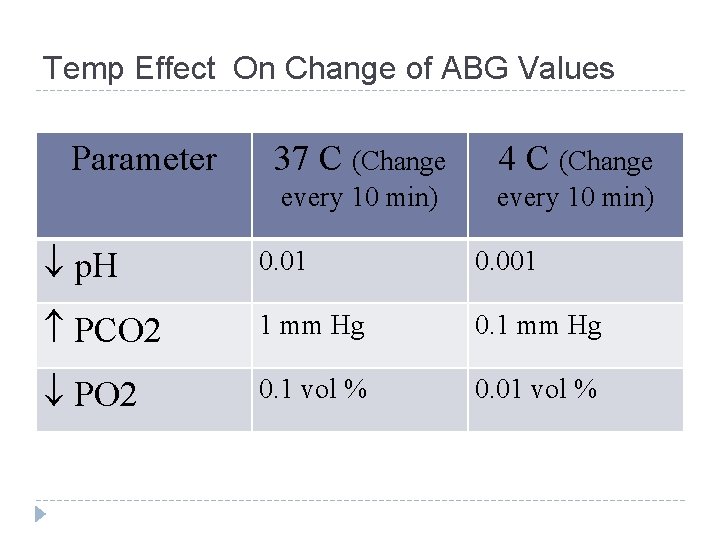
Temp Effect On Change of ABG Values Parameter 37 C (Change 4 C (Change every 10 min) p. H 0. 01 0. 001 PCO 2 1 mm Hg 0. 1 mm Hg PO 2 0. 1 vol % 0. 01 vol %

FEVER OR HYPOTHERMIA 1. 2. 3. q Most ABG analyzers report data at N body temp If severe hyper/hypothermia, values of p. H & PCO 2 at 37 C can be significantly diff from pt’s actual values Changes in PO 2 values with temp also predictable If Pt. ’s temp < 37 C Substract 5 mm. Hg Po 2, 2 mm. Hg Pco 2 and Add 0. 012 p. H per 1 C decrease of temperature Hansen JE, Clinics in Chest Med 10(2), 1989 227 -237

AIR BUBBLES : 1. 2. PO 2 150 mm. Hg & PCO 2 0 mm Hg in air bubble(R. A. ) Mixing with sample, lead to Pa. O 2 & Pa. CO 2 q. To avoid air bubble, sample drawn very slowly and preferabily in glass syringe Steady State: ØSampling should done during steady state after change in oxygen therepy or ventilator parameter ØSteady state is achieved usually within 3 -10 minutes

Leucocytosis : Ø p. H and Po 2 ; and Pco 2 Ø 0. 1 ml of O 2 consumed/d. L of blood in 10 min in pts with N TLC Ø Marked increase in pts with very high TLC/plt counts – hence imm chilling/analysis essential EXCESSIVE HEPARIN effect on results HCO 3 - & Pa. CO 2 ØOnly. 05 ml heperin required for 1 ml blood. ØDilutional q. So syringe be emptied of heparin after flushing or only dead space volume is sufficient or dry heperin should be used

q TYPE OF SYRINGE 1. p. H & PCO 2 values unaffected PO 2 values drop more rapidly in plastic syringes (ONLY if PO 2 > 400 mm Hg) Differences usually not of clinical significance so plastic syringes can be and continue to be used 2. q q. Risk of alteration of results with: 1. size of syringe/needle 2. vol of sample q HYPERVENTILATION OR BREATH HOLDING May lead to erroneous lab results

COMPONENTS OF THE ABG p. H: Measurement of acidity or alkalinity, based on the hydrogen (H+). 7. 35 – 7. 45 Pao 2 : The partial pressure oxygen that is dissolved in arterial blood. 80 -100 mm Hg. q q PCO 2: The amount of carbon dioxide dissolved in arterial blood. 35– 45 mm. Hg HCO 3 : The calculated value of the amount of bicarbonate in the blood. 22 – 26 mmol/L Sa. O 2: The arterial oxygen saturation. >95% p. H, Pa. O 2 , Pa. CO 2 , Lactate and electrolytes are measured Variables HCO 3 (Measured or calculated)

Contd… Buffer Base: It is total quantity of buffers in blood including both volatile(Hco 3) and nonvolatile (as Hgb, albumin, Po 4) Base Excess/Base Deficit: Amount of strong acid or base needed to restore plasma p. H to 7. 40 at a Pa. CO 2 of 40 mm Hg, at 37*C. Calculated from p. H, Pa. CO 2 and HCT Negative BE also referred to as Base Deficit True reflection of non respiratory (metabolic) acid base status Normal value: -2 to +2 m. Eq/L
![CENTRAL EQUATION OF ACID-BASE PHYSIOLOGY Ø Henderson Hasselbach Equation: q [H+] in n. Eq/L CENTRAL EQUATION OF ACID-BASE PHYSIOLOGY Ø Henderson Hasselbach Equation: q [H+] in n. Eq/L](http://slidetodoc.com/presentation_image_h/e72d14f4a99a1908310decc2b35714b8/image-11.jpg)
CENTRAL EQUATION OF ACID-BASE PHYSIOLOGY Ø Henderson Hasselbach Equation: q [H+] in n. Eq/L = 24 x (PCO 2 / [HCO 3 -] ) Ø where [ H+] is related to p. H by q [ H+] in n. Eq/L = 10 (9 -p. H) To maintain a constant p. H, PCO 2/HCO 3 - ratio should be constant When one component of the PCO 2/[HCO 3 - ]ratio is altered, the compensatory response alters the other component in the same direction to keep the PCO 2/[HCO 3 - ] ratio constant

Compensatory response or regulation of p. H By 3 mechanisms: Chemical buffers: Ø Ø React instantly to compensate for the addition or subtraction of H+ ions CO 2 elimination: Ø Ø Ø Controlled by the respiratory system Change in p. H result in change in PCO 2 within minutes HCO 3 - elimination: Ø Ø Controlled by the kidneys Change in p. H result in change in HCO 3 It takes hours to days and full compensation occurs in 2 -5 days
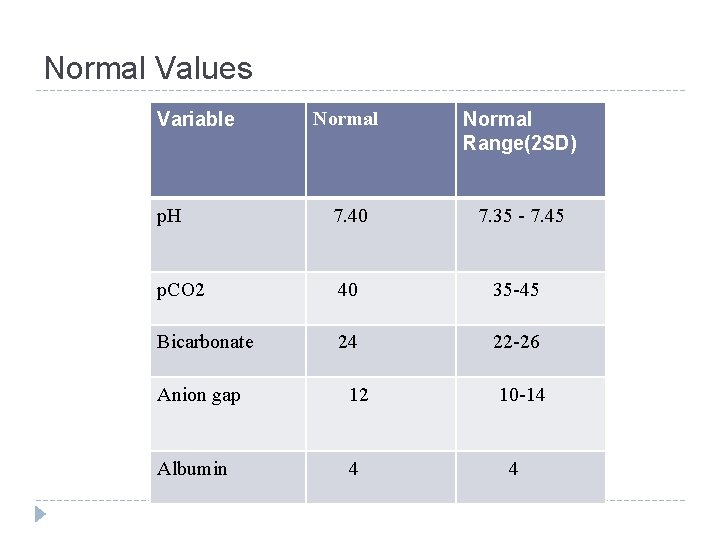
Normal Values Variable Normal Range(2 SD) p. H 7. 40 7. 35 - 7. 45 p. CO 2 40 35 -45 Bicarbonate 24 22 -26 Anion gap 12 Albumin 4 10 -14 4

Steps for ABG analysis 1. 2. 3. 4. 5. 6. 7. What is the p. H? Acidemia or Alkalemia? What is the primary disorder present? Is there appropriate compensation? Is the compensation acute or chronic? Is there an anion gap? If there is a AG check the delta gap? What is the differential for the clinical processes?

Step 1: Look at the p. H: is the blood acidemic or alkalemic? EXAMPLE : 65 yo M with CKD presenting with nausea, diarrhea and acute respiratory distress v v ABG : ABG 7. 23/17/235 on 50% VM BMP Na 123/ Cl 97/ HCO 3 7/BUN 119/ Cr 5. 1 ACIDMEIA OR ALKALEMIA ? ?

EXAMPLE ONE ABG 7. 23/17/235 on 50% VM v BMP Na 123/ Cl 97/ HCO 3 7/BUN 119/ Cr 5. 1 Answer PH = 7. 23 , HCO 3 7 Acidemia v
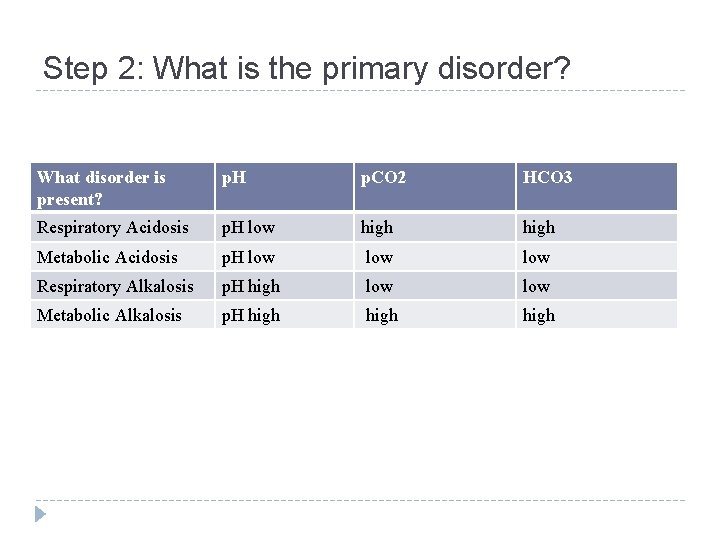
Step 2: What is the primary disorder? What disorder is present? p. H p. CO 2 HCO 3 Respiratory Acidosis p. H low high Metabolic Acidosis p. H low low Respiratory Alkalosis p. H high low Metabolic Alkalosis p. H high

Contd…. ØMetabolic Conditions are suggested if p. H changes in the same direction as p. CO 2 or p. H is abnormal but p. CO 2 remains unchanged ØRespiratory p. H Conditions are suggested if: changes in the opp direction as p. CO 2 or p. H is abnormal but HCO 3 remains unchanged

EXAMPLE v v ABG 7. 23/17/235 on 50% VM BMP Na 123/ Cl 97/ HCO 3 7/BUN 119/ Cr 5. PH is low , CO 2 is Low PH and PCO 2 are going in same directions then its most likely primary metabolic

EXPECTED CHANGES IN ACID-BASE DISORDERS Primary Disorder Metabolic acidosis Metabolic alkalosis Acute respiratory acidosis Chronic respiratory acidosis Acute respiratory alkalosis Chronic respiratory alkalosis Expected Changes PCO 2 = 1. 5 × HCO 3 + (8 ± 2) PCO 2 = 0. 7 × HCO 3 + (21 ± 2) delta p. H = 0. 008 × (PCO 2 - 40) delta p. H = 0. 003 × (PCO 2 - 40) delta p. H = 0. 008 × (40 - PCO 2) delta p. H = 0. 003 × (40 - PCO 2) From: THE ICU BOOK - 2 nd Ed. (1998) [Corrected]

Step 3 -4: Is there appropriate compensation? Is it chronic or acute? Ø Respiratory Acidosis Acute (Uncompensated): for every 10 increase in p. CO 2 -> HCO 3 increases by 1 and there is a decrease of 0. 08 in p. H Chronic (Compensated): for every 10 increase in p. CO 2 -> HCO 3 increases by 4 and there is a decrease of 0. 03 in p. H Ø Respiratory Alkalosis Ø Ø Acute (Uncompensated): for every 10 decrease in p. CO 2 -> HCO 3 decreases by 2 and there is a increase of 0. 08 in PH Chronic (Compensated): for every 10 decrease in p. CO 2 -> HCO 3 decreases by 5 and there is a increase of 0. 03 in PH q Partial Compensated: Change in p. H will be between 0. 03 to 0. 08 for every 10 mm. Hg change in PCO 2 1 4 10 2 5

Step 3 -4: Is there appropriate compensation? Ø Ø Metabolic Acidosis Ø Winter’s formula: Expected p. CO 2 = 1. 5[HCO 3] + 8 ± 2 OR p. CO 2 = 1. 2 ( HCO 3) Ø If serum p. CO 2 > expected p. CO 2 -> additional respiratory acidosis and vice versa Metabolic Alkalosis Expected PCO 2 = 0. 7 × HCO 3 + (21 ± 2) OR p. CO 2 = 0. 7 ( HCO 3) If serum p. CO 2 < expected p. CO 2 - additional respiratory alkalosis and vice versa

EXAMPLE v v ABG 7. 23/17/235 on 50% VM BMP Na 123/ Cl 97/ HCO 3 7/BUN 119/ Cr 5. Winter’s formula : 17= 1. 5 (7) +8 ± 2 = 18. 5(16. 5 – 20. 5) So correct compensation so there is only one disorder Primary metabolic

Step 5: Calculate the anion gap AG used to assess acid-base status esp in D/D of met acidosis AG & HCO 3 - used to assess mixed acid-base disorders q AG based on principle of electroneutrality: Total Serum Cations = Total Serum Anions Na + (K + Ca + Mg) = HCO 3 + Cl + (PO 4 + SO 4 + Protein + Organic Acids) Na + UC = HCO 3 + Cl + UA Na – (HCO 3 + Cl) = UA – UC Na – (HCO 3 + Cl) = AG Normal =12 ± 2
![Contd… AG corrected = AG + 2. 5[4 – albumin] If there is an Contd… AG corrected = AG + 2. 5[4 – albumin] If there is an](http://slidetodoc.com/presentation_image_h/e72d14f4a99a1908310decc2b35714b8/image-25.jpg)
Contd… AG corrected = AG + 2. 5[4 – albumin] If there is an anion Gap then calculate the Delta/delta gap (step 6) to determine additional hidden nongap metabolic acidosis or metabolic alkalosis If there is no anion gap then start analyzing for nonanion gap acidosis

EXAMPLE Calculate Anion gap v v ABG 7. 23/17/235 on 50% VM BMP Na 123/ Cl 97/ HCO 3 7/BUN 119/ Cr 5/ Albumin 2. AG = Na – Cl – HCO 3 (normal 12 ± 2) 123 – 97 – 7 = 19 AG corrected = AG + 2. 5[4 – albumin] = 19 + 2. 5 [4 – 2] = 19 + 5 = 24

Step 6: Calculate Delta Gap Delta gap = (actual AG – 12) + HCO 3 Adjusted HCO 3 should be 24 (+_ 6) {18 -30} If delta gap > 30 -> additional metabolic alkalosis If delta gap < 18 -> additional non-gap metabolic acidosis If delta gap 18 – 30 -> no additional metabolic disorders

EXAMPLE : Delta Gap v v ABG 7. 23/17/235 on 50% VM BMP Na 123/ Cl 97/ HCO 3 7/BUN 119/ Cr 5/ Albumin 4. Delta gap = (actual AG – 12) + HCO 3 (19 -12) +7 = 14 Delta gap < 18 -> additional non-gap metabolic acidosis So Metabolic acidosis anion and non anion gap
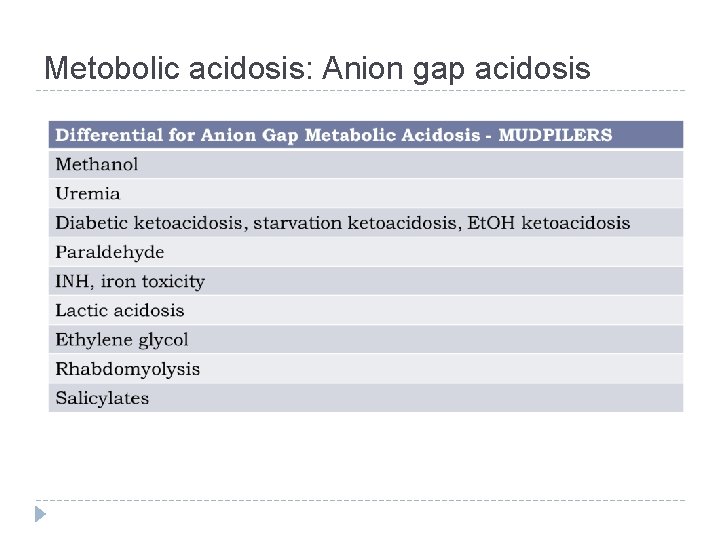
Metobolic acidosis: Anion gap acidosis

EXAMPLE: WHY ANION GAP? 65 yo M with CKD presenting with nausea, diarrhea and acute respiratory distress v v ABG : ABG 7. 23/17/235 on 50% VM BMP Na 123/ Cl 97/ HCO 3 7/BUN 119/ Cr 5. 1 So for our patient for anion gap portion its due to BUN of 119 UREMIA But would still check lactic acid

Nongap metabolic acidosis For non-gap metabolic acidosis, calculate the urine anion gap URINARY AG Total Urine Cations = Total Urine Anions Na + K + (NH 4 and other UC) = Cl + UA (Na + K) + UC = Cl + UA (Na + K) – Cl = UA – UC (Na + K) – Cl = AG Distinguish GI from q. Urenal AG = causes UNA + of UKloss – Uof CL HCO 3 by estimating Urinary NH 4+. Hence a -ve UAG (av -20 meq/L) seen in GI, while +ve value (av +23 meq/L) seen in renal problem. Kaehny WD. Manual of Nephrology 2000; 48 -62

EXAMPLE : NON ANION GAP ACIDOSIS 65 yo M with CKD presenting with nausea, diarrhea and acute respiratory distress v v v ABG : ABG 7. 23/17/235 on 50% VM BMP Na 123/ Cl 97/ HCO 3 14 AG = 123 – 97 -14 = 12 Most likely due to the diarrhea
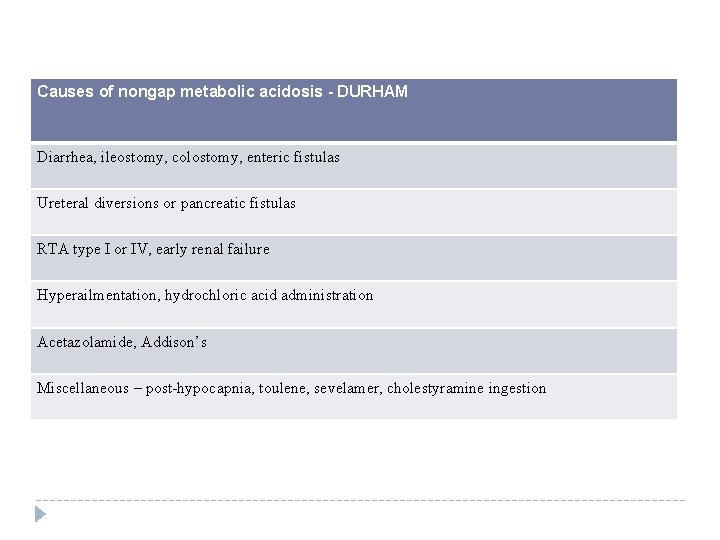
Causes of nongap metabolic acidosis - DURHAM Diarrhea, ileostomy, colostomy, enteric fistulas Ureteral diversions or pancreatic fistulas RTA type I or IV, early renal failure Hyperailmentation, hydrochloric acid administration Acetazolamide, Addison’s Miscellaneous – post-hypocapnia, toulene, sevelamer, cholestyramine ingestion
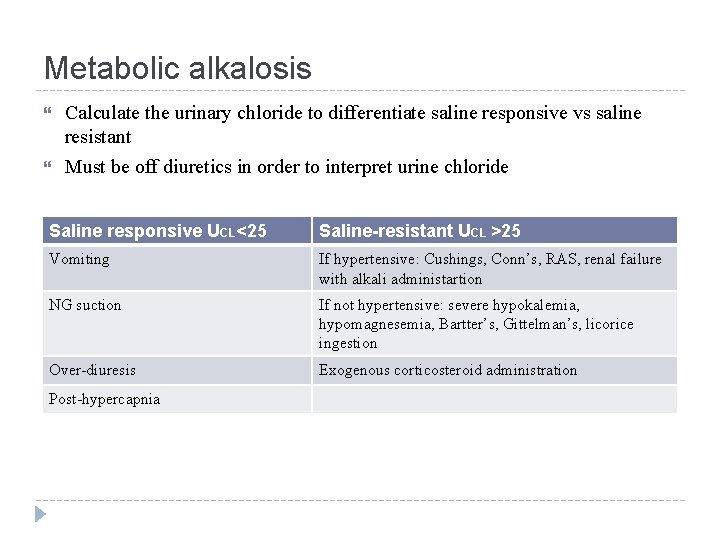
Metabolic alkalosis Calculate the urinary chloride to differentiate saline responsive vs saline resistant Must be off diuretics in order to interpret urine chloride Saline responsive UCL<25 Saline-resistant UCL >25 Vomiting If hypertensive: Cushings, Conn’s, RAS, renal failure with alkali administartion NG suction If not hypertensive: severe hypokalemia, hypomagnesemia, Bartter’s, Gittelman’s, licorice ingestion Over-diuresis Exogenous corticosteroid administration Post-hypercapnia
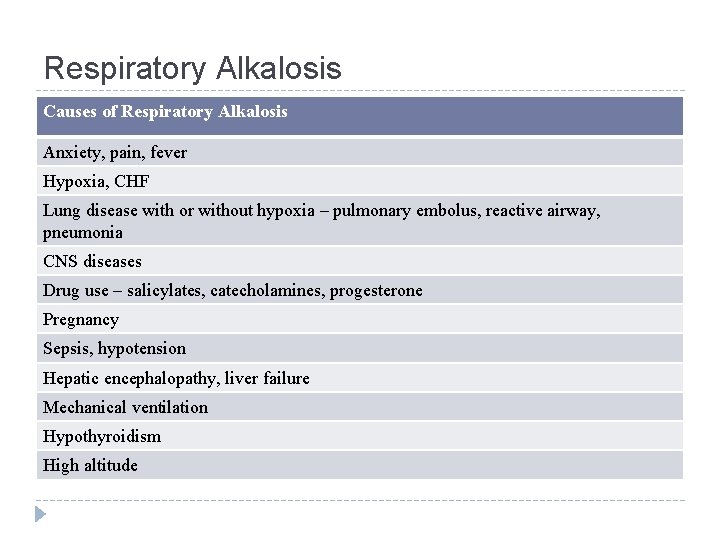
Respiratory Alkalosis Causes of Respiratory Alkalosis Anxiety, pain, fever Hypoxia, CHF Lung disease with or without hypoxia – pulmonary embolus, reactive airway, pneumonia CNS diseases Drug use – salicylates, catecholamines, progesterone Pregnancy Sepsis, hypotension Hepatic encephalopathy, liver failure Mechanical ventilation Hypothyroidism High altitude

Case 1. 7. 27/58/60 on 5 L, HCO 3 - 26, anion gap is 12, albumin is 4. 0 1. p. H= Acidemia (p. H < 7. 4) 2. CO 2= Acid (CO 2>40) Opposite direction so Primary disturbance = Respiratory Acidosis 3 &4: Compensation : Acute or chronic? ACUTE CO 2 has increased by (58 -40)=18 If chronic the p. H will decrease 0. 05 (0. 003 x 18 = 0. 054) p. H would be 7. 35 If acute the p. H will decrease 0. 14 (0. 008 x 18 = 0. 144) p. H would be 7. 26.

Contd. 5: Anion gap –N/A 6: There is an acute respiratory acidosis, is there a metabolic problem too? ΔHCO 3 - = 1 m. Eq/L↑/10 mm. Hg↑p. CO 2 The p. CO 2 is up by 18 so it is expected that the HCO 3 - will go up by 1. 8. Expected HCO 3 - is 25. 8, compared to the actual HCO 3 - of 26, so there is no additional metabolic disturbance. Dx-ACUTE RESPIRATORY ACIDOSIS

Case. 2 7. 54/24/99 on room air, HCO 3 - 20, anion gap is 12, albumin is 4. 0. 1: p. H= Alkalemia (p. H > 7. 4) 2. CO 2= Base (CO 2<40) p. H & p. CO 2 change in opposite Direction So Primary disturbance = Respiratory Alkalosis 3 &4: Compensation ? acute or chronic? ACUTE ΔCO 2 =40 -24=16 If chronic the p. H will increase 0. 05 (0. 003 x 16 = 0. 048) p. H would be 7. 45 If acute the p. H will increase 0. 13(0. 008 x 16 = 0. 128) p. H would be 7. 53

Contd… 5: Anion gap – N/A 6: There is an acute respiratory alkalosis, is there a metabolic problem too? ΔHCO 3 - = 2 m. Eq/L↓/10 mm. Hg↓p. CO 2 The p. CO 2 is down by 16 so it is expected that the HCO 3 - will go down by 3. 2. Expected HCO 3 - is 20. 8, compared to the actual HCO 3 of 20, so there is no additional metabolic disturbance. Dx-ACUTE RESPIRATORY ALKALOSIS

Case-3 7. 58/55/80 on room air, HCO 3 - 46, anion gap is 12, albumin is 4. 0. Ucl -20 1: p. H= Alkalemia(p. H > 7. 4) 2: CO 2= Acid (CO 2>40) Same direction so Primary disturbance = Metabolic Alkalosis 3&4: Compensation: ∆ p. CO 2=0. 7 x ∆ HCO 3 The HCO 3 - is up by 22. CO 2 will increase by 0. 7 x 22 = 15. 4. Expected CO 2 is 55. 4, compared to the actual CO 2 of 55, therefore there is no additional respiratory disturbance.

contd 5: No anion gap is present; and no adjustment needs to be made for albumin. Metabolic Alkalosis Urinary chloride is 20 meq/l (< 25 meq/l)so chloride responsive, have to treat with Normal saline. Dx-METABOLIC ALKALOSIS

Case-4 7. 46/20/80 on room air, HCO 3 - 16, anion gap = 12, albumin = 4. 0 1: p. H = Alkalemia (p. H > 7. 4) 2: CO 2 = Base (CO 2<40) So Primary disturbance = Respiratory Alkalosis 3 &4: Compensation? acute or chronic? Chronic ΔCO 2 =40 -20= 20. If chronic the p. H will increase 0. 06 (0. 003 x 20 = 0. 06) p. H would be 7. 46. If acute the p. H will increase 0. 16 (0. 008 x 20 = 0. 16) p. H would be 7. 56.

Contd…. 5: Anion gap – N/A 6: There is a chronic respiratory alkalosis, is there a metabolic problem also? Chronic: ΔHCO 3 - = 4 m. Eq/L↓/10 mm. Hg↓p. CO 2 The p. CO 2 is down by 20 so it is expected that the HCO 3 - will go down by 8. Expected HCO 3 - is 16, therefore there is no additional metabolic disorder. Dx-CHRONIC RESPIRATORY ALKALOSIS

Case-5 7. 19/35/60 on 7 L, HCO 3 - 9, anion gap = 18, albumin = 4. 0 1: p. H = Acidemia (p. H < 7. 4) 2: CO 2= Base (CO 2<40) So Primary disturbance: Metabolic Acidosis 3&4: Compensation ? 5: ∆ p. CO 2=1. 2 x ∆ HCO 3 CO 2 will decrease by 1. 2 (∆HCO 3 -) 1. 2 (24 -9) 18. 40 – 18= 22 Actual CO 2 is higher than expected Respiratory Acidosis Anion Gap = 18 (alb normal so no correction necessary)

Contd…. . 6: Delta Gap: Delta gap = (actual AG – 12) + HCO 3 = (18 -12) + 9 = 6 + 9 = 15 which is<18 Non-AG Met Acidosis Dx-ANION GAP METABOLIC ACIDOSIS with NON-ANION GAP METABOLIC ACIDOSIS with RESPIRATORY ACIDOSIS

Case-6 7. 54/80/65 on 2 L, HCO 3 - 54, anion gap 12, albumin = 4. 0 , Ucl 40 meq/l 1: p. H = Alkalemia (p. H > 7. 4) 2: CO 2= Acid (CO 2>40) So Primary disturbance: Metabolic Alkalosis 3&4: Compensation? ∆ p. CO 2=0. 7 x ∆ HCO 3 CO 2 will increase by 0. 7 (∆HCO 3 -) 0. 7 (54 -24) 21 40 + 21 = 61 Actual CO 2 is higher than expected Respiratory Acidosis

Contd…. 5: Anion Gap = 12 (alb normal so no correction necessary) Urinary chloride is 40 meq/l (> 25 meq/l)so chloride resistant. So treatment would be disease specific and repletion of potassium Dx-METABOLIC ALKALOSIS with RESPIRATORY ACIDOSIS

Case-7 7. 6/30/83 on room air, HCO 3 - 28, anion gap = 12, albumin = 4. 0 1: p. H = Alkalemia (p. H > 7. 4) 2: CO 2= Base (CO 2<40) So. Primary Disturbance: Metabolic Alkalosis 3&4: Compensation ? ∆ p. CO 2=0. 7 x ∆ HCO 3 CO 2 will increase by 0. 7 (∆HCO 3 -) 0. 7 (28 -24) 2. 8 40 + 2. 8 = 42. 8 Actual CO 2 is lower than expected Respiratory Alkalosis Anion Gap = 12 (alb normal so no correction necessary) See urinary chloride for further Dx. Dx-METABOLIC ALKALOSIS with RESPIRATORY ALKALOSIS

Case-8 A 50 yo male present with sudden onset of SOB with following ABG 7. 25/46/78 on 2 L, HCO 3 - 20, anion gap = 10, albumin = 4. 0 1: p. H = Acidemia (p. H < 7. 4) 2: CO 2= Acid (CO 2>40) So Primary disturbance: Respiratory Acidosis 3 &4: If respiratory disturbance is it acute or chronic? ACUTE ∆ CO 2 = 46 -40= 6 If chronic the p. H will decrease 0. 02 (0. 003 x 6 = 0. 018) p. H would be 7. 38 If acute the p. H will decrease 0. 05 (0. 008 x 6 = 0. 048) p. H would be 7. 35.

Contd… Anion Gap = 10 (alb normal so no correction necessary) 6: There is an acute respiratory acidosis, is there a metabolic problem too? ∆ HCO 3 - = 1 m. Eq/L↑/10 mm. Hg↑p. CO 2 The HCO 3 - will go up 1 m. Eq/L for every 10 mm. Hg the p. CO 2 goes up above 40 The p. CO 2 is up by 6 so it is expected that the HCO 3 - will go up by 0. 6. Expected HCO 3 - is 24. 6, compared to the actual HCO 3 - of 20. Since the HCO 3 - is lower than expected Non-Anion Gap Metabolic Acidosis (which we suspected). Dx-RESPIRATORY ACIDOSIS with NON-ANION GAP METABOLIC ACIDOSIS

Case-9 7. 15/22/75 on room air, HCO 3 - 9, anion gap = 10, albumin = 2. 0 1: p. H = Acidemia (p. H < 7. 4) 2: CO 2= Base (CO 2<40) So Primary disturbance: Metabolic Acidosis 3&4: ∆ Compensation ? p. CO 2=1. 2 x ∆ HCO 3 Expected p. CO 2 = 1. 2 x ∆ HCO 3 - 1. 2 (24 -9) 1. 2 (15) 18. The expected p. CO 2 is 22 mm. Hg. The actual p. CO 2 is 22, which is expected, so there is no concomitant disorder.

Contd…. 5: Anion Gap = 10 AGc = 10 + 2. 5(4 -2) = 15 Anion Gap Metabolic Acidosis 6: Delta Gap: Delta gap = (actual AG – 12) + HCO 3 = (15 -12) + 9 = 3+ 9 = 12 which is<18 Non-AG Met Acidosis Dx-ANION GAP METABOLIC ACIDOSIS with NON-ANION GAP METABOLIC ACIDOSIS

- Slides: 53