Salt formation Fe Br 3 Iron III bromide
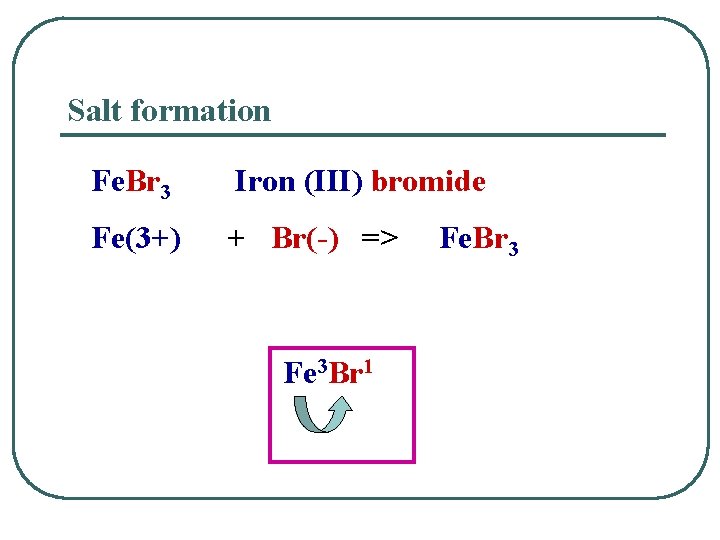
Salt formation Fe. Br 3 Iron (III) bromide Fe(3+) + Br(-) => Fe 3 Br 1 Fe. Br 3

Some acids Formula HF HCl HBr HI HNO 3 HNO 2 H 2 SO 4 H 2 SO 3 Name hydrofluoric acid hydrochloric acid hydrobromic acid hydroiodic acid nitrous acid sulfuric acid Sulfurous acid
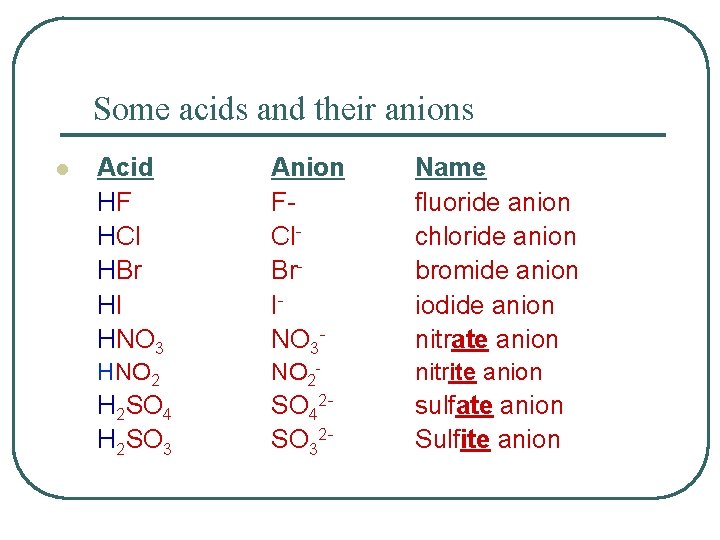
Some acids and their anions l Acid HF HCl HBr HI HNO 3 Anion FCl. Br. INO 3 - Name fluoride anion chloride anion bromide anion iodide anion nitrate anion HNO 2 - nitrite anion H 2 SO 4 H 2 SO 3 SO 42 SO 32 - sulfate anion Sulfite anion

Naming salts of acids l Higher oxidation state is named (stem)ic acid. l Lower oxidation state is named (stem)ous acid l Salts are named based on the acids. • • Anions of -ic acids make “ate” salts. Anions of -ous acids make “ite” salts. acid ic salt ate ous ite
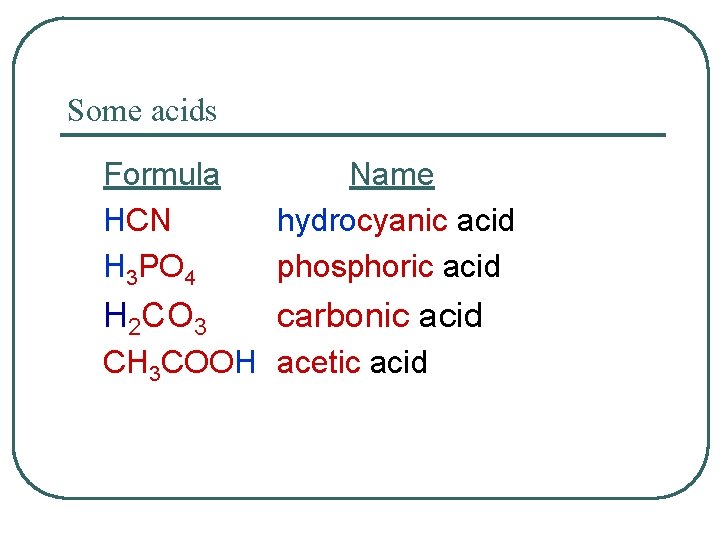
Some acids Formula HCN H 3 PO 4 Name hydrocyanic acid phosphoric acid H 2 CO 3 carbonic acid CH 3 COOH acetic acid

Some acids and their anions l Acid HCN H 3 PO 4 H 2 CO 3 CH 3 COOH Anion CNPO 43 CO 32 CH 3 COO- Name cyanide anion phosphate anion carbonate anion acetate anion

Common bases Formula Li. OH Na. OH KOH Rb. OH Cs. OH Ca(OH)2 Sr(OH)2 Ba(OH)2 Name lithium hydroxide sodium hydroxide potassium hydroxide rubidium hydroxide cesium hydroxide calcium hydroxide strontium hydroxide barium hydroxide

More bases l Formula Cu(OH)2 Fe(OH)3 Zn(OH)2 Mg(OH)2 Name copper (II) hydroxide iron (III) hydroxide zinc (II) hydroxide magnesium (II) hydroxide

Naming some oxides l l l Formula Name CO CO 2 SO 3 carbon monoxide carbon dioxide sulfur trioxide

Naming some inorganic compounds Cation named first Anion named second l l Li. Br Mg. Cl 2 Li 2 S Al 2 O 3 lithium bromide magnesium (II) chloride lithium sulfide Aluminum (III) oxide

Naming some inorganic compounds l l l l KOH Ba(OH)2 Al(OH)3 Fe(OH)2 Fe(OH)3 Ba(CN)2 (NH 4)2 S NH 4 CN potassium hydroxide barium (II) hydroxide aluminum (III) hydroxide iron (III) hydroxide barium (II) cyanide ammonium sulfide ammonium cyanide

Naming some salts l Acid HNO 2 nitrous acid l HNO 3 nitric acid l H 2 SO 3 sulfurous acid Salt Na. NO 2 sodium nitrite Na. NO 3 sodium nitrate Na 2 SO 3 sodium sulfite

Classify each of the following ions as a monoatomic or a polyatomic l l l Mg 2+ SO 32 Cu+ NH 4+ Br- monatomic cation polyatomic anion monatomic cation polyatomic cation monatomic anion I will do it!

Name the following compounds l l l l Cu. CO 3 (NH 4)2 CO 3 Zn. O Fe(CH 3 COO)3 Al 2(CO 3)3 Mg. Cl 2 Zn(OH)2 (NH 4)2 S You do it!

Write the correct formula for the following You do it! compounds l l l Potassium iodide Copper(II) nitrate Silver(I) sulfite Magnesium (II) carbonate Zinc (II) carbonate PI Cu. NO 3 Ag. SO 4 Mg 3 CO 3 Zn(CO 3)2
- Slides: 15