A very very bad joke A cation and

A very, very bad joke… A cation and an anion were walking down the street… …and the cation says to the anion, “Whoa, I think I lost an electron!” The anion replies, “Are you sure? ” …to which the cation replies, “Yes…I’m POSITIVE!” <FORCED LAUGHTER> www. bitterfilms. com

Cations and Anions and Electrons, Oh My! Al 0 Al 3+ + 3 e 13 protons ____ 13 protons 13 electrons 10 electrons ____ Al 0 has lost 3 electrons. Al 0 has been oxidized: Its oxidation state has increased from 0 3. Al 3+ + 3 e. Al 0 13 protons ____ 13 protons 10 electrons 13 electrons ____ Al 3+ has gained 3 electrons. Al 3+ has been reduced: Its oxidation state has been reduced from 3 0.

Redox Reactions: A New Type of Reaction All chemical reactions can be categorized, roughly into: • Acid-base reactions – depending on your definition § Displacement reactions – exchange of ions Na. Br (aq) + Ag. NO 3 (aq) § Na. NO 3 (aq) + Ag. Br (s) Acid-base reactions – exchange of H+ or OH- HBr (aq) + Li. OH (aq) Li. Br (aq) + H 2 O (l) • Oxidation-Reduction (redox) reactions – exchange of electrons Al 0 I 20 + 2 e- Al 3+ + 3 e 2 I-

Redox Rules! Calculating Oxidation States 1. Oxidation states for elements are always zero. e. g. N 2(g), Co(s), H 2(g), C(gr), P 4(s), Na(s), Ne(g) O H H 2. Oxidation states for monatomic ions are the same as their charges. e. g. Al 3+, Cd 2+, Zn 2+, Ag+, and Group IA(1) and IIA(2) ions 3. The oxidation state of oxygen is usually 2 -, except for peroxides (-1). e. g. Na 2 O, Al 2 O 3; compare with H 2 O 2, Na 2 O 2 – remember, O 224. The oxidation state of hydrogen is usually 1+, except for hydrides (-1). e. g. H 2 O, OH-; compare with Na. H, Li. Al. H 4 – look for H bonded to metal 5. The oxidation state of halides are 1 -, except when bonded to oxygen. e. g. HF, Na. Cl, Mg. Br 2 (F is always 1 -); compare with Cl. O 3 - 6. The oxidation numbers MUST add up to the molecule’s total charge.

More Practice with Oxidation States Cr 2 O 72 - Cr = ? O = -2 2(? ) + 7(-2) = -2 Ti. Cl 4 4 Cl = 4(-1) Ti = +4 Cr = +6 dichromate titanium(IV) chloride H 2 SO 3 S = +4 Ba. O 2 2 O = 2(-1) barium peroxide P 2 O 5 2 P = 2(+5) diphosphorus pentoxide sulfurous acid
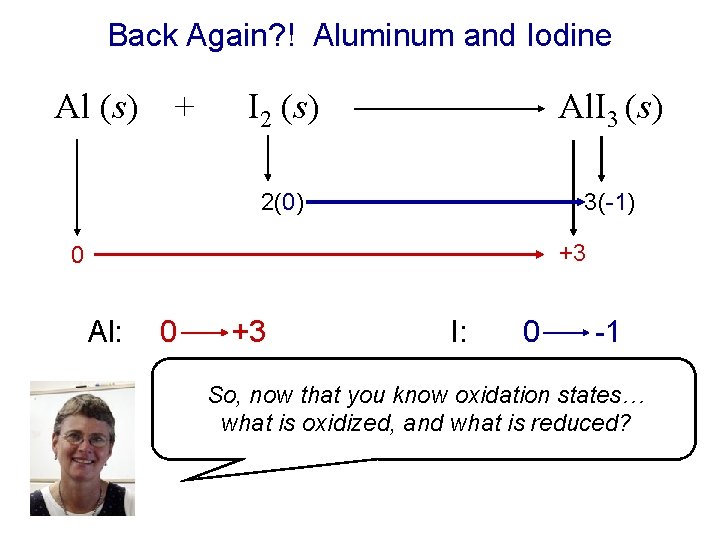
Back Again? ! Aluminum and Iodine Al (s) + I 2 (s) Al. I 3 (s) 2(0) 3(-1) +3 0 Al: 0 +3 I: 0 -1 So, now that you know oxidation states… what is oxidized, and what is reduced?

Mnemonic Devices for Remembering Redox Rules LEO the lion goes GER When an atom/molecule Loses Electrons, it is Oxidized. -its oxidation state becomes more positive -it is the reducing agent, or reductant When an atom/molecule Gains Electrons, it is Reduced. -its oxidation state becomes more negative -it is the oxidizing agent, or oxidant http: //www. franklinhoward. com/ LEO Oxidation O Reduction R Is I Loss L Gain G

Back For Good!! Aluminum and Iodine Al (s) + I 2 (s) Al. I 3 (s) 3(-1) 2(0) +3 0 Al: 0 +3 I: oxidation state increases loses electrons Al is oxidized Al is the reducing agent 2 Al (s) + 3 I 2 (s) 0 -1 oxidation state decreases gains electrons I 2 is reduced I 2 is the oxidizing agent 2 Al. I 3 (s)

Identifying Oxidizing/Reducing Agents Oxidizing/reducing agents are ALWAYS REACTANTS! Oxidizing agents (oxidants) are reactants that are REDUCED. Reducing agents (reductants) are reactants that are OXIDIZED! Zn (s) + 2 H+ (aq) 0 2(+1) Zn loses/gains electrons Zn is oxidized/reduced Zn is the reductant/oxidant H 2 (g) + Zn 2+ (aq) 2(0) +2 H+ loses/gains electrons H+ is oxidized/reduced H+ is the reductant/oxidant

Identifying Oxidizing/Reducing Agents Oxidizing/reducing agents are ALWAYS REACTANTS! Oxidizing agents (oxidants) are reactants that are REDUCED. Reducing agents (reductants) are reactants that are OXIDIZED! 2 H 2 O (l) + Al (s) + Mn. O 4 - (aq) 0 Al(OH)4 - (aq) + Mn. O 2 (s) +4 +3 +7 What is oxidized? What is reduced? Al (s) Mn. O 4 - (aq) What is the oxidizing agent? What is the reducing agent? Mn. O 4 - (aq) Al (s) How many electrons are transferred? 3 e-

Practical Applications: Electroplating deposits coats of metal onto objects Bronze baby shoes Cu+ (aq) + e- Cu (s) http: //www. castinggifts. com/shoebronzing Recoating Tarnished objects + (aq) + NO - (aq) Ag. NO 3 (s) Ag 3 H 2 O Ag+ (aq) + e. Ag (s) http: //www. collectorssecret. com/images/sprbeforeafter. jpg
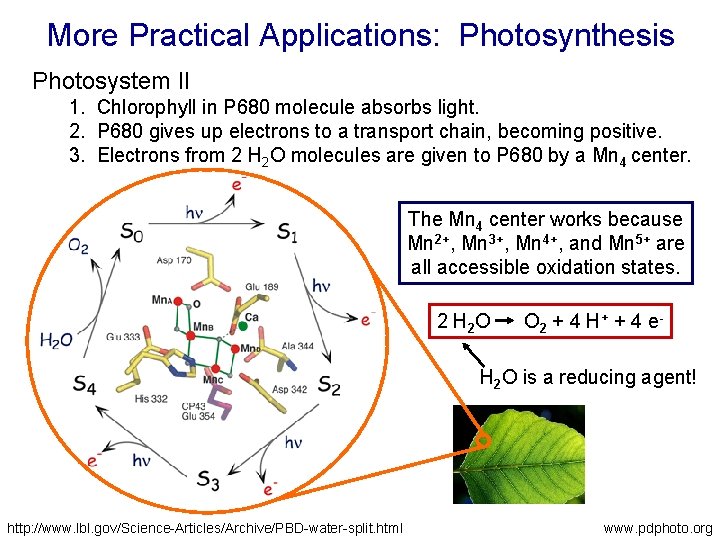
More Practical Applications: Photosynthesis Photosystem II 1. Chlorophyll in P 680 molecule absorbs light. 2. P 680 gives up electrons to a transport chain, becoming positive. 3. Electrons from 2 H 2 O molecules are given to P 680 by a Mn 4 center. The Mn 4 center works because Mn 2+, Mn 3+, Mn 4+, and Mn 5+ are all accessible oxidation states. 2 H 2 O O 2 + 4 H+ + 4 e- H 2 O is a reducing agent! http: //www. lbl. gov/Science-Articles/Archive/PBD-water-split. html www. pdphoto. org
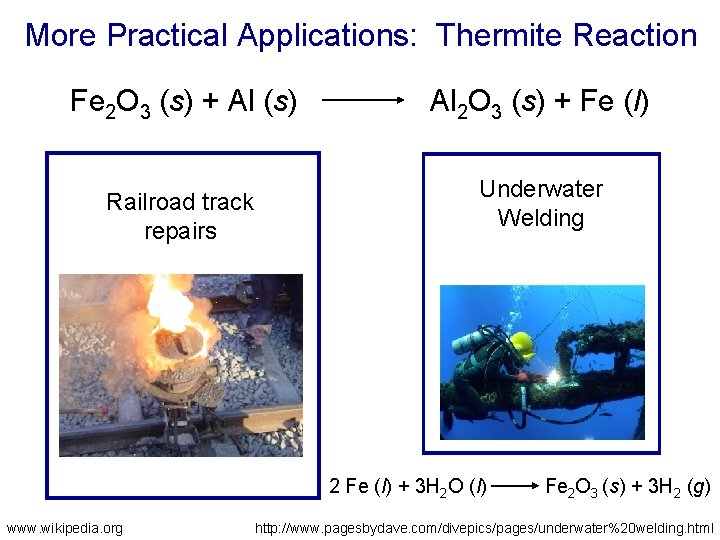
More Practical Applications: Thermite Reaction Fe 2 O 3 (s) + Al (s) Railroad track repairs Al 2 O 3 (s) + Fe (l) Underwater Welding 2 Fe (l) + 3 H 2 O (l) www. wikipedia. org Fe 2 O 3 (s) + 3 H 2 (g) http: //www. pagesbydave. com/divepics/pages/underwater%20 welding. html
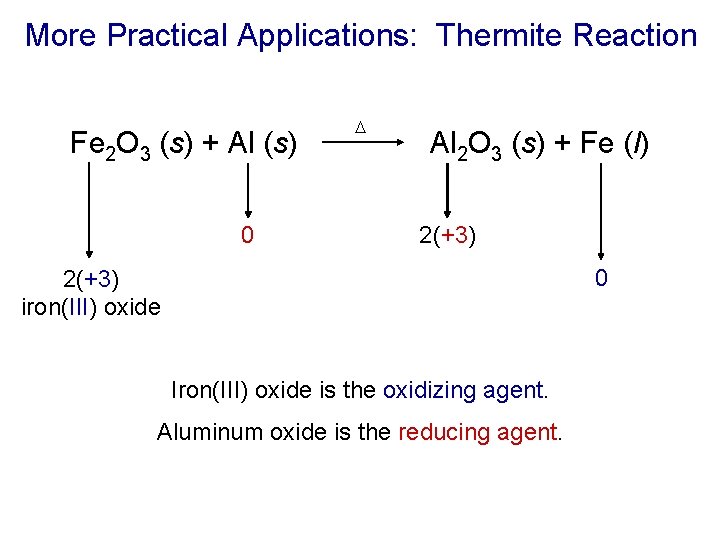
More Practical Applications: Thermite Reaction Fe 2 O 3 (s) + Al (s) 0 D Al 2 O 3 (s) + Fe (l) 2(+3) 0 2(+3) iron(III) oxide Iron(III) oxide is the oxidizing agent. Aluminum oxide is the reducing agent.

Limits to Redox Reactions What is the oxidation number of S in the following compounds? H 2 S S 8 -2 0 Which cannot be an oxidizing agent? (reduced) H 2 S – cannot gain more electrons SCl 2 +2 Na 2 SO 3 +4 SO 42 - +6 Which cannot be a reducing agent? (oxidized) SO 42 - – cannot lose more electrons

Disproportionation and Conproportionation These are special types of redox reactions: Disproportionation – One compound decomposes into two oxidation states H 2 O 2 (aq) catalyst 2(-1) H 2 O (l) + O 2 (g) -2 2(0) H 2 O 2 is BOTH oxidized and reduced! Conproportionation – Two oxidation states become one. Cr 1+ (aq) + Cr 3+ (aq) 2 Cr 2+ (aq)
- Slides: 16