18 6 BaseCatalyzed Enolization Enolate Anions Acidity of

18. 6 Base-Catalyzed Enolization: Enolate Anions
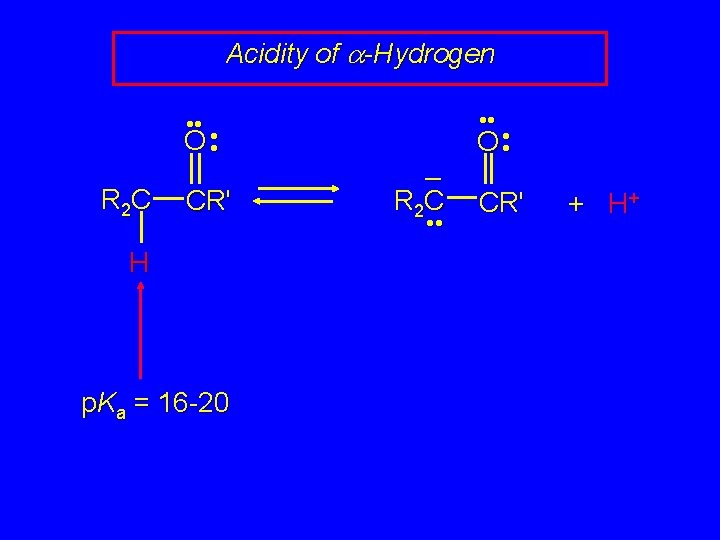
Acidity of a-Hydrogen • • O • • R 2 C CR' H p. Ka = 16 -20 • • – R 2 C • • O • • CR' + H+
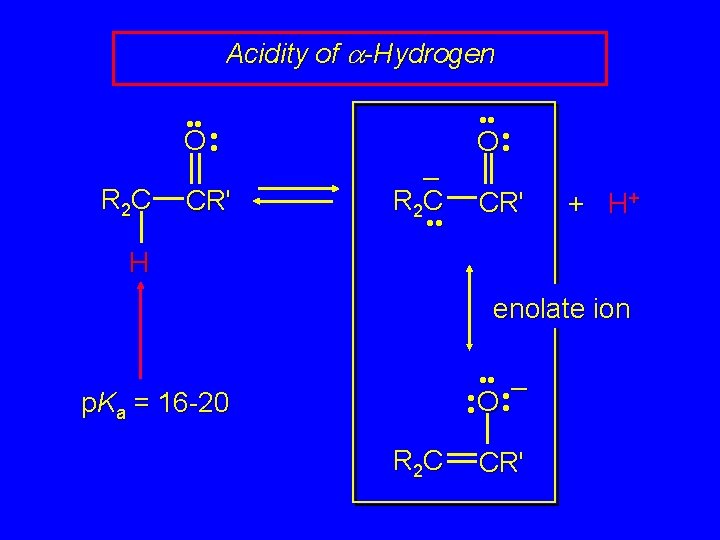
Acidity of a-Hydrogen • • O • • R 2 C CR' • • – R 2 C • • O • • CR' + H+ H enolate ion • • – • • O • • p. Ka = 16 -20 R 2 C CR'
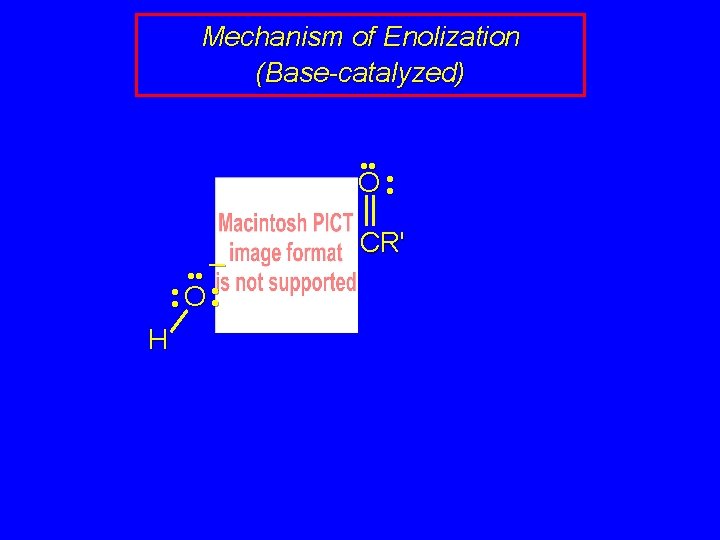
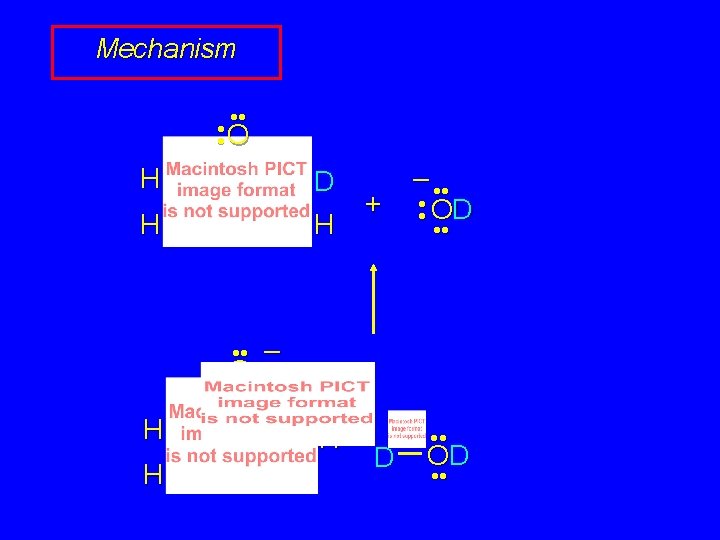
Mechanism of Enolization (Base-catalyzed) • • O • • – • • O • • H R 2 C H CR'
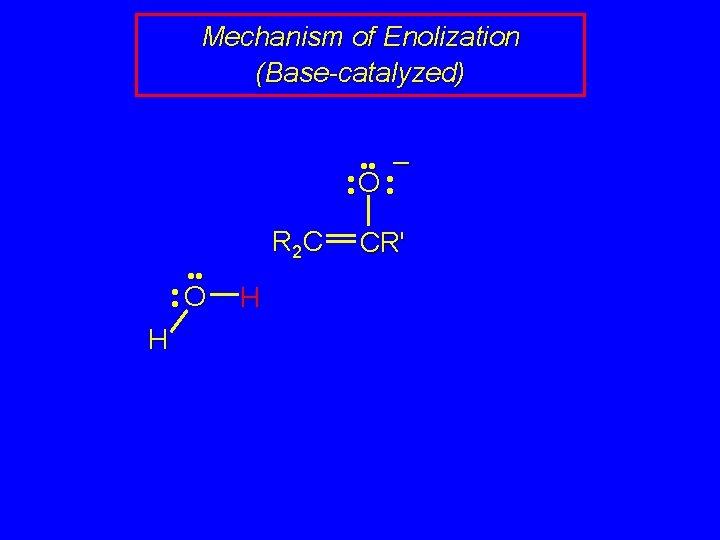
Mechanism of Enolization (Base-catalyzed) • • – • • O • • O H R 2 C H CR'
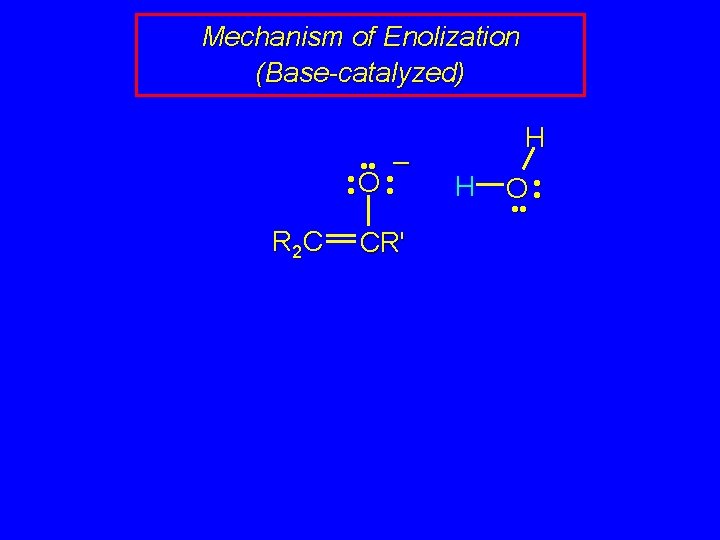
Mechanism of Enolization (Base-catalyzed) • • – • • O • • R 2 C CR' H H O • •
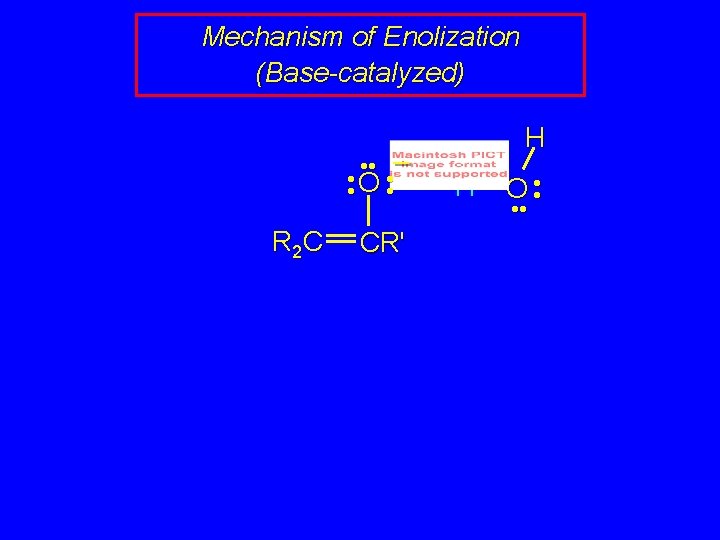
Mechanism of Enolization (Base-catalyzed) • • – • • O • • R 2 C CR' H H O • •
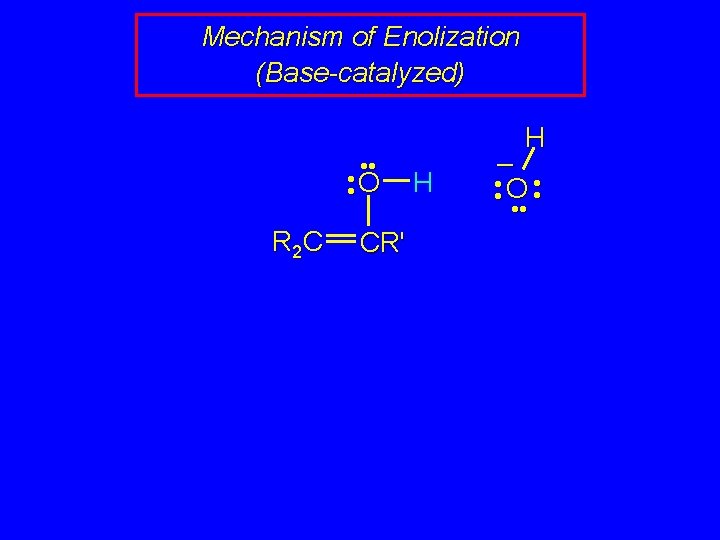
Mechanism of Enolization (Base-catalyzed) • • O R 2 C CR' H H – • • O • •
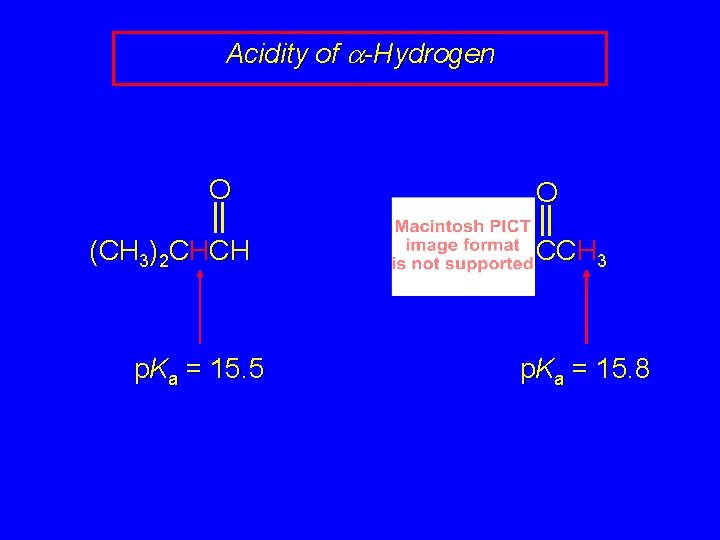
Acidity of a-Hydrogen O (CH 3)2 CHCH p. Ka = 15. 5 O CCH 3 p. Ka = 15. 8
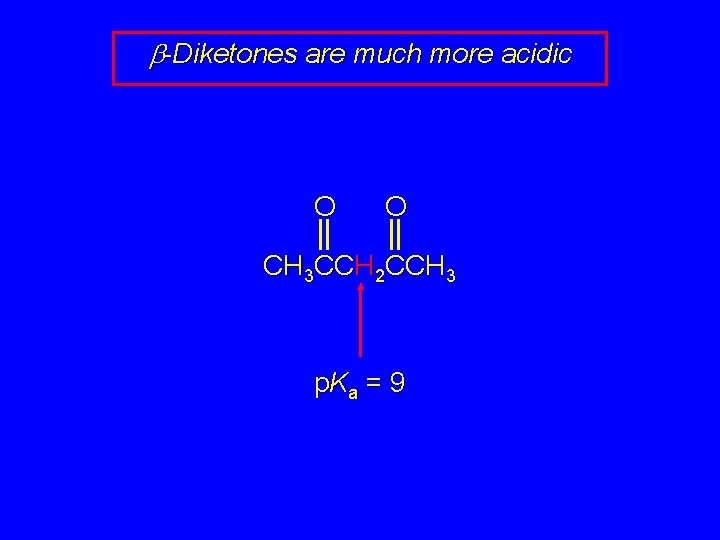
b-Diketones are much more acidic O O CH 3 CCH 2 CCH 3 p. K a = 9
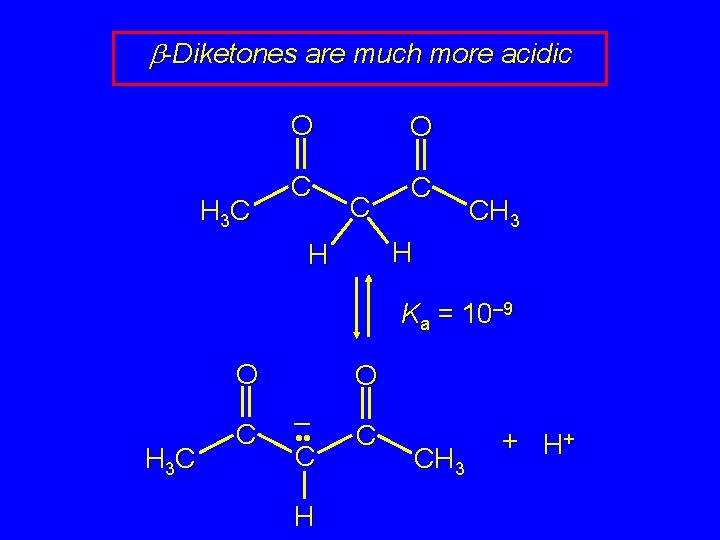
b-Diketones are much more acidic H 3 C O O C CH 3 H H Ka = 10– 9 O H 3 C C O – • • C H C CH 3 + H+
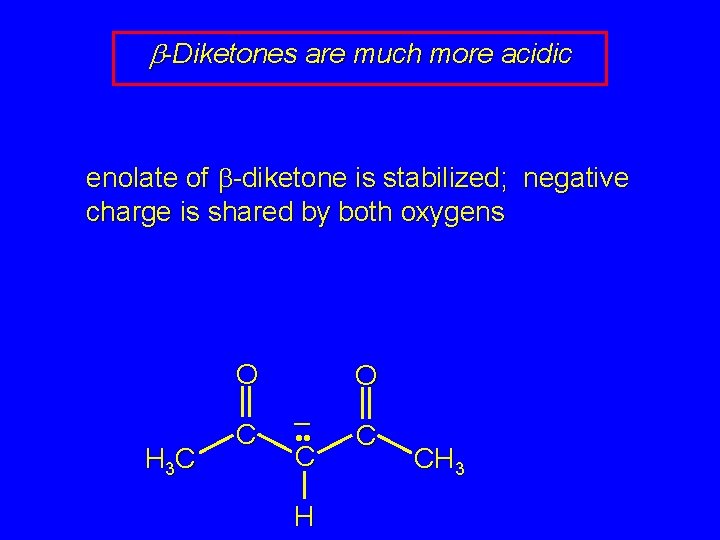
b-Diketones are much more acidic enolate of b-diketone is stabilized; negative charge is shared by both oxygens O H 3 C C O – • • C H C CH 3
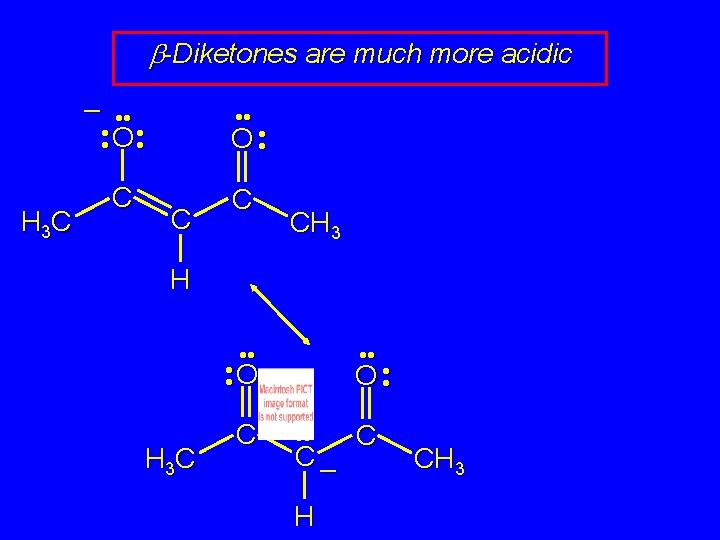
b-Diketones are much more acidic H 3 C – • • O • • C CH 3 H • • O H 3 C C O • • C– H C CH 3
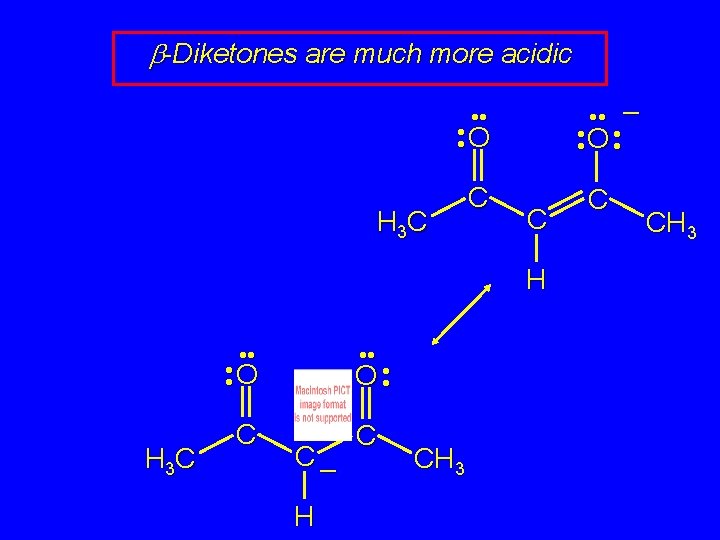
b-Diketones are much more acidic • • – • • O • • O H 3 C C C H • • O H 3 C C O • • C– H C CH 3

18. 7 The Haloform Reaction

The Haloform Reaction Under basic conditions, halogenation of a methyl ketone often leads to carbon-carbon bond cleavage. Such cleavage is called the haloform reaction because chloroform, bromoform, or iodoform is one of the products.
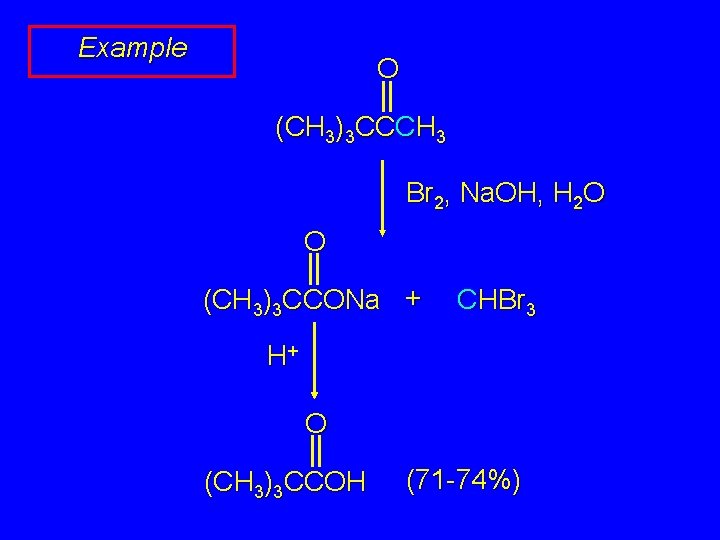
Example O (CH 3)3 CCCH 3 Br 2, Na. OH, H 2 O O (CH 3)3 CCONa + CHBr 3 H+ O (CH 3)3 CCOH (71 -74%)
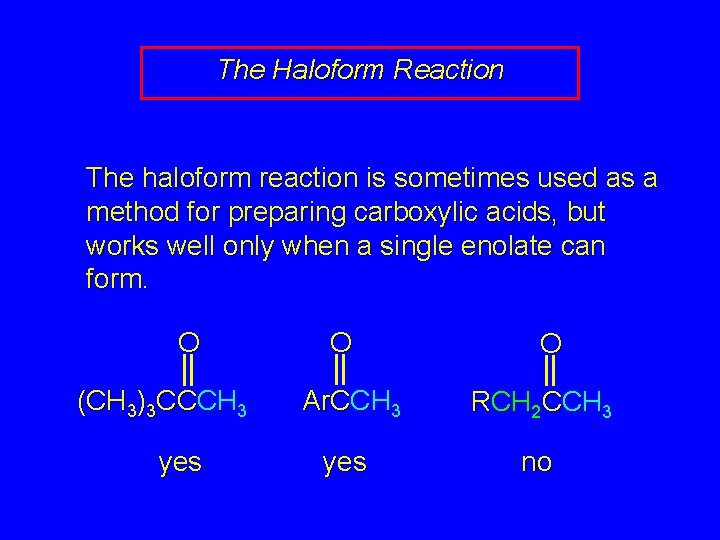
The Haloform Reaction The haloform reaction is sometimes used as a method for preparing carboxylic acids, but works well only when a single enolate can form. O (CH 3)3 CCCH 3 yes O O Ar. CCH 3 RCH 2 CCH 3 yes no
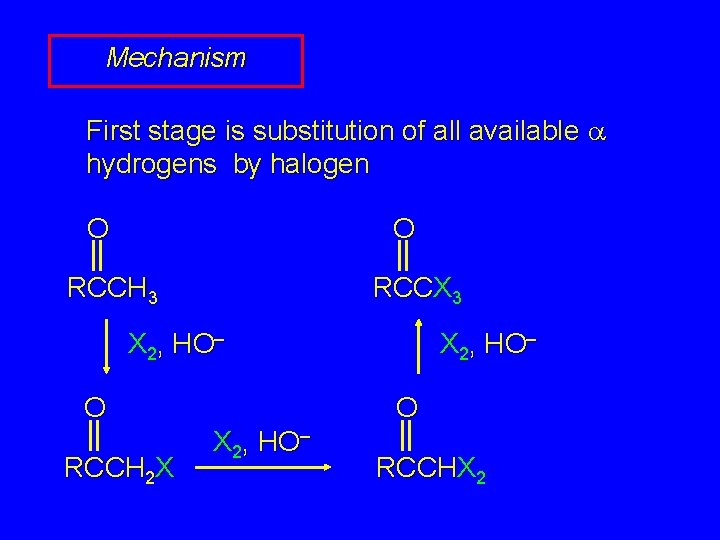
Mechanism First stage is substitution of all available a hydrogens by halogen O O RCCH 3 RCCX 3 X 2, HO– O RCCH 2 X X 2, HO– O X 2, HO– RCCHX 2
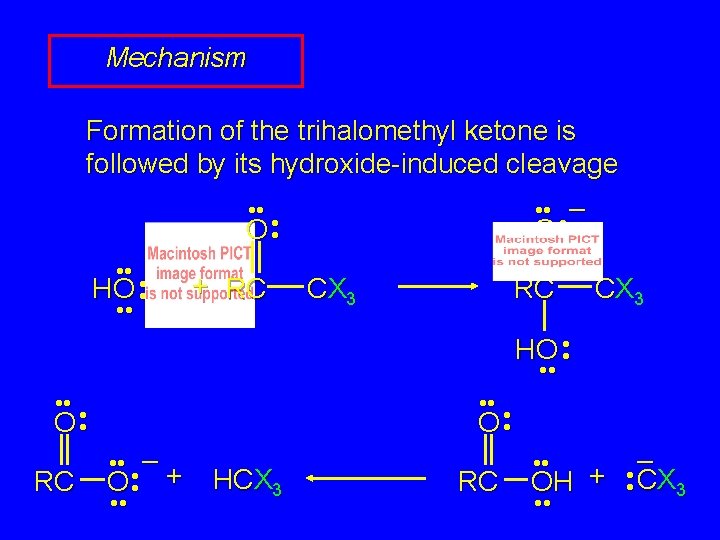
Mechanism Formation of the trihalomethyl ketone is followed by its hydroxide-induced cleavage • • – • • O • • – + RC HO • • CX 3 RC CX 3 HO • • O • • RC • • O • • – O • • + • • HCX 3 RC • • – OH + • • CX 3 • •

18. 8 Some Chemical and Stereochemical Consequences of Enolization

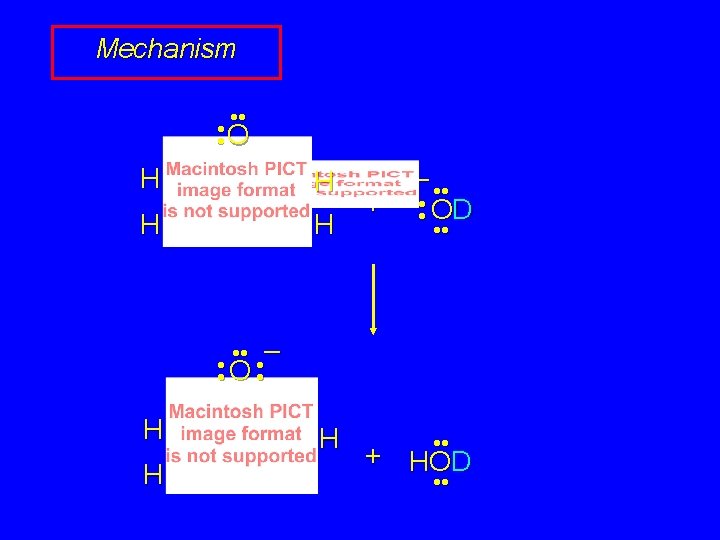
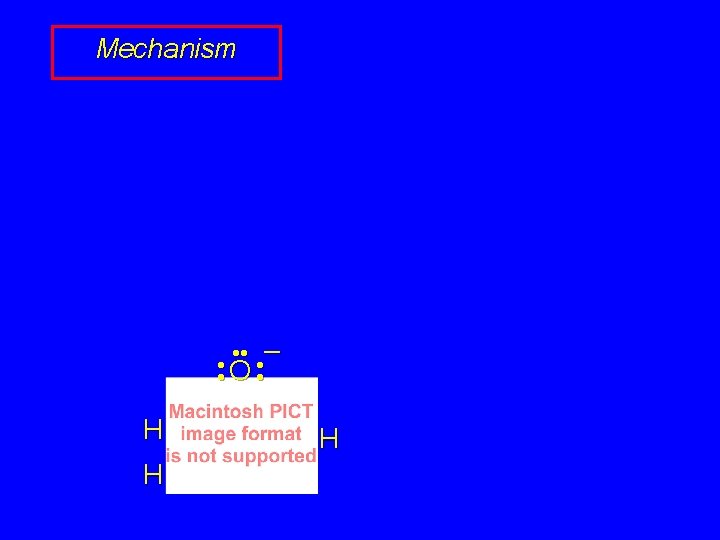
Hydrogen-Deuterium Exchange O H H + 4 D 2 O KOD, heat O D D + 4 DOH
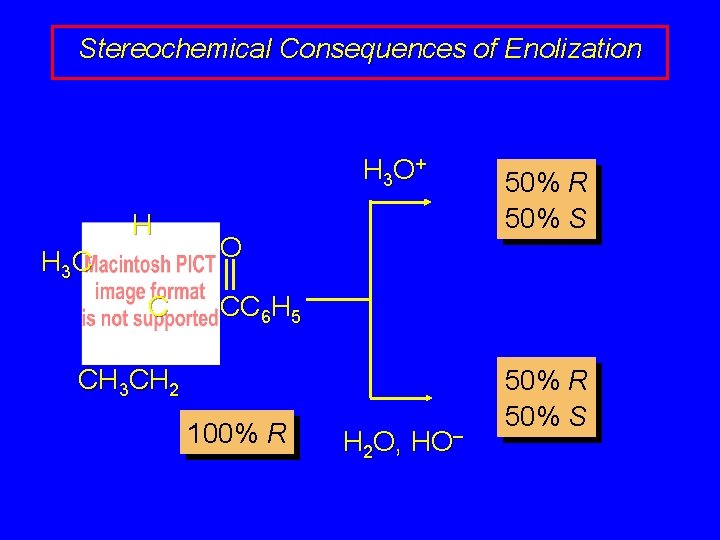
Stereochemical Consequences of Enolization H 3 O + H H 3 C C O 50% R 50% S CC 6 H 5 CH 3 CH 2 100% R H 2 O, HO– 50% R 50% S
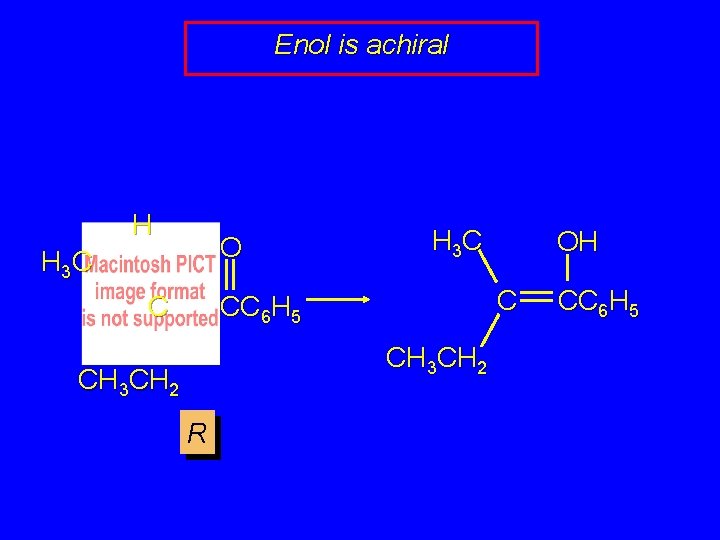
Enol is achiral H O H 3 C C CC 6 H 5 C CH 3 CH 2 R OH CC 6 H 5
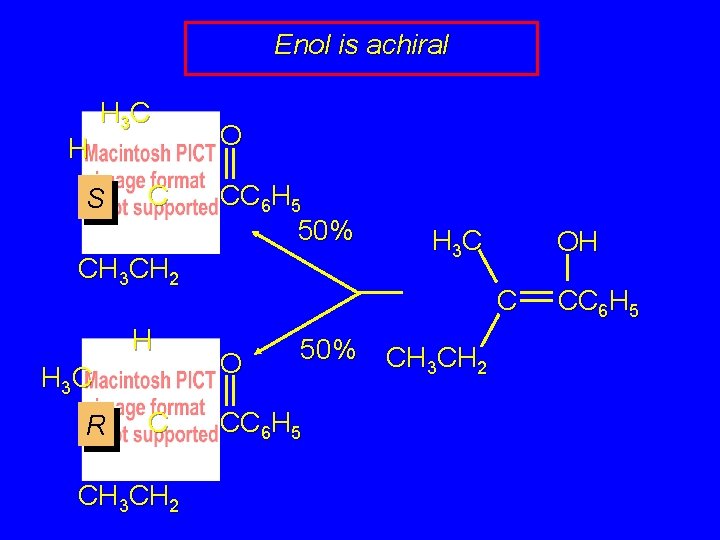
Enol is achiral H H 3 C S C O CC 6 H 5 50% CH 3 CH 2 H H 3 C R C CH 3 CH 2 H 3 C OH C O 50% CC 6 H 5 CH 3 CH 2 CC 6 H 5

Results of Rate Studies H H 3 C C CH 3 CH 2 O CC 6 H 5 Equal rates for: racemization H-D exchange bromination iodination Enol is intermediate and its formation is rate-determining
- Slides: 30