Angela Chen Sweeteners from Starch Sweeteners from Starch














































































































- Slides: 150

Angela Chen

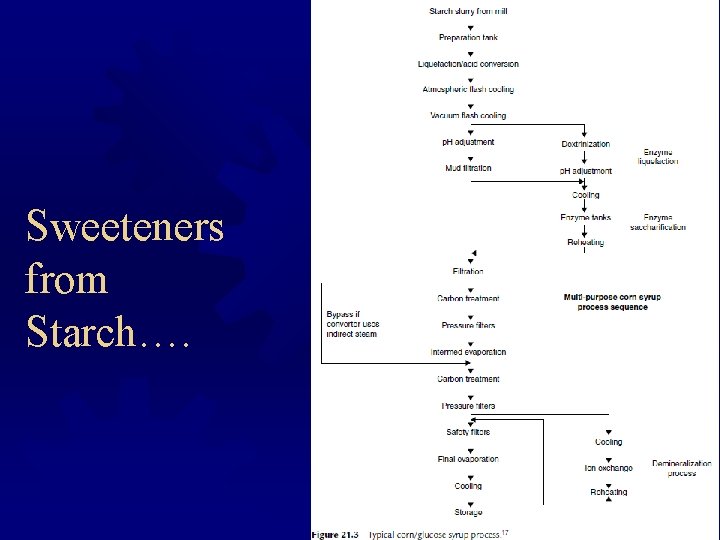
Sweeteners from Starch….

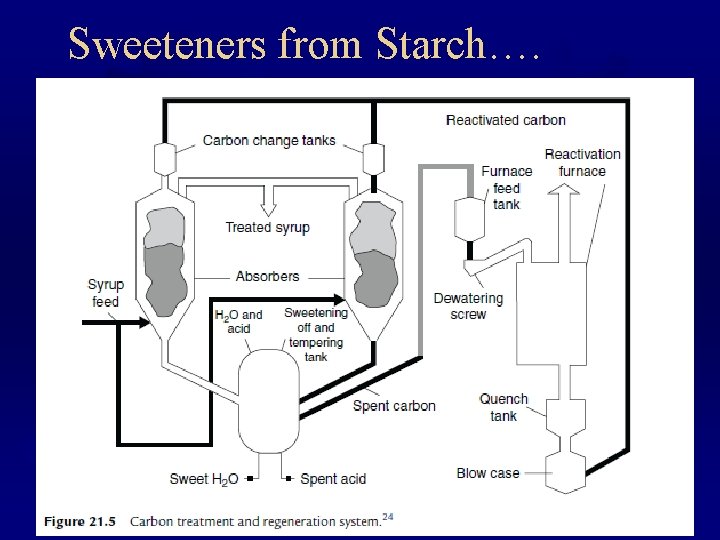
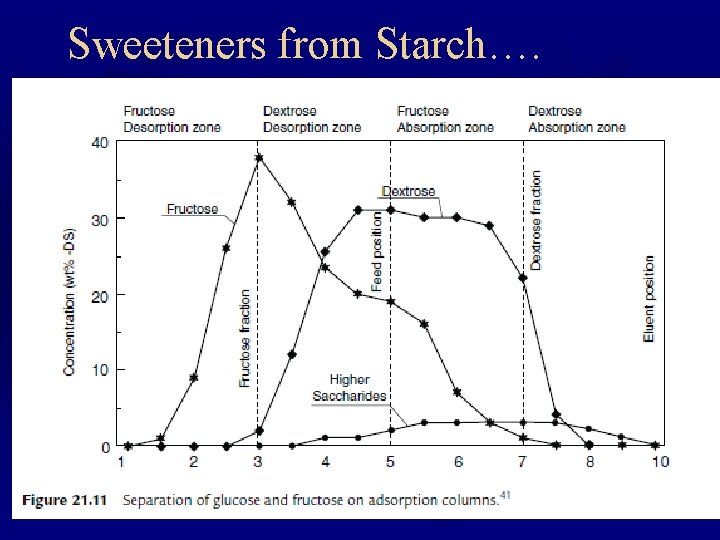
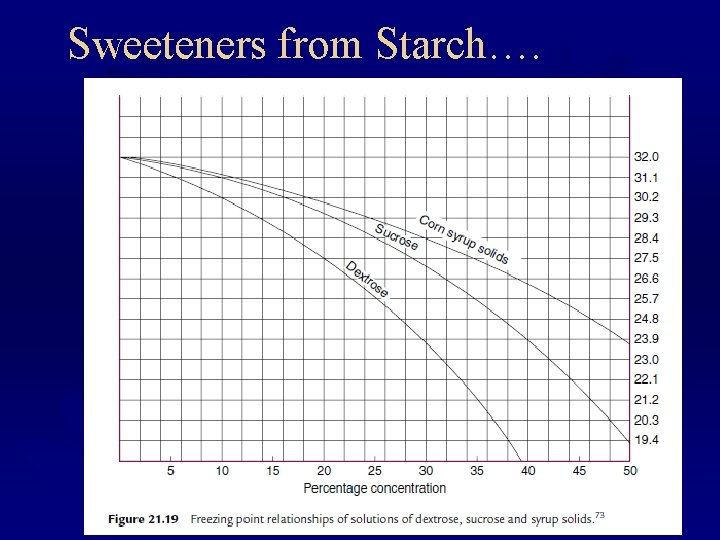
Sweeteners from Starch….

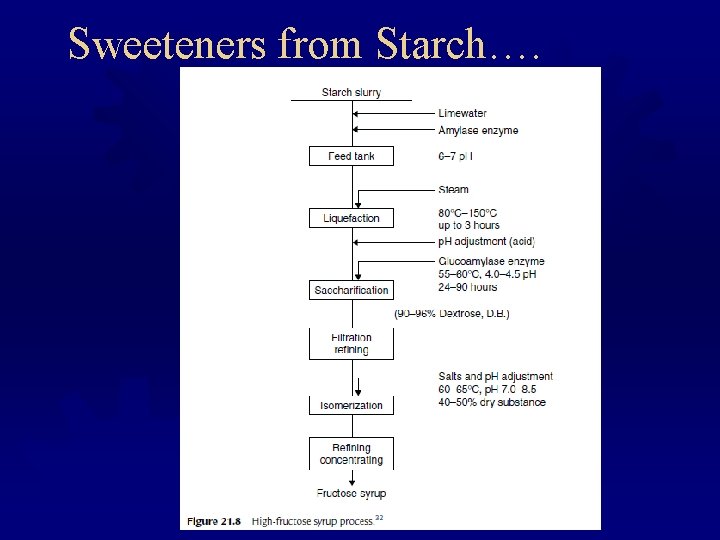
Sweeteners from Starch….

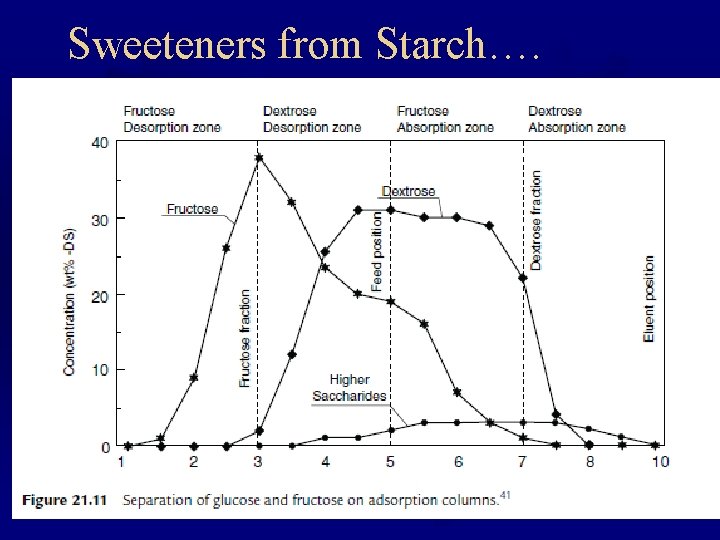
Sweeteners from Starch….

Sweeteners from Starch….

Sweeteners from Starch….

Hydrocolloids Binding water with carbohydrates

Starches- Our #1 Hydrocolloid? ® Hydrocolloids are substances that will form a gel or add viscosity on addition of water. ® Most are polysaccharides and all form significant H- bonding with water with processing. ® Size, structure, and charge are the most important factors relating to texture and physical features of foods

Small versus Large ® Small molecule sugars would create a high osmotic pressure if stored in sufficient quantities to be useful. ® Polymerized sugars reduce the number of molecules present and hence the osmotic effects. ® Free polymers are too thick to allow cell to function ® Thus, plants store energy into starch granules

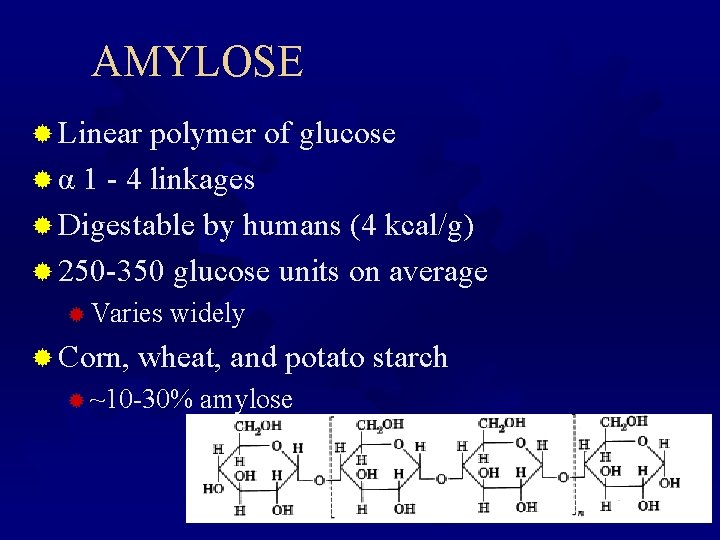
AMYLOSE ® Linear polymer of glucose ® α 1 - 4 linkages ® Digestable by humans (4 kcal/g) ® 250 -350 glucose units on average ® Varies widely ® Corn, wheat, and potato starch ® ~10 -30% amylose

AMYLOPECTIN ® Branched chain polymer of glucose ® α 1 - 4 and α 1 - 6 glycosidic linkages ® Mostly digestible by humans ® 1, 000 glucose units is common ® Branch points every ~15 -25 units

Starch ® Amylose may have a few branched chains ® Helical structure with a hydrophobic core ® Core may contain lipids, metals, etc. ® Amylose to Amylopectin ratios ~ 1: 4 ® Varies with the plant source ® Waxy starches are ~100% amylopectin ® Sugary “mutant” starches have more amylose

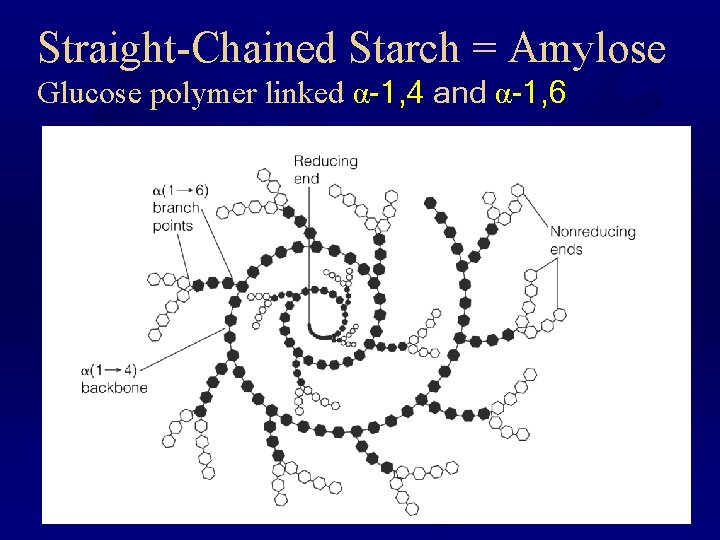
Straight-Chained Starch = Amylose Glucose polymer linked α-1, 4 and α-1, 6

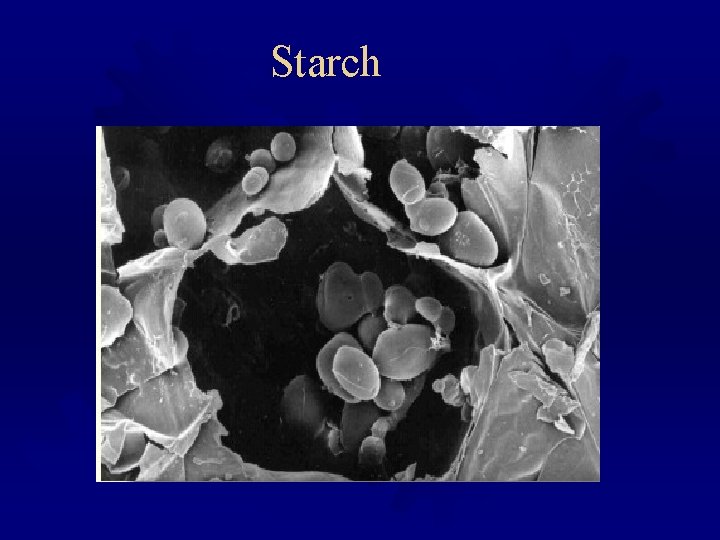
Starch

® Birefringence When starch granules are viewed under the microscope by polarized light they exhibit a phenomenon known as birefringency - the refraction of polarized light by the intact crystalline regions to give a characteristic "Maltese cross" pattern on each granule. The cross disappears upon heating and gelatinization.

Modified Starches ® Gelatinization is the easiest modification ® Heated in water then dried. ® Acid and/heat will form “dextrins” ® α-Amylase hydrolyzes α (1 -4) linkage ® random attack to make shorter chains ® ® β-Amylase Also attacks α (1 - 4) linkages ® Starts at the non-reducing end of the starch chain ® Gives short dextrins and maltose ® ® Both enzymes have trouble with α (1 - 6) linkages

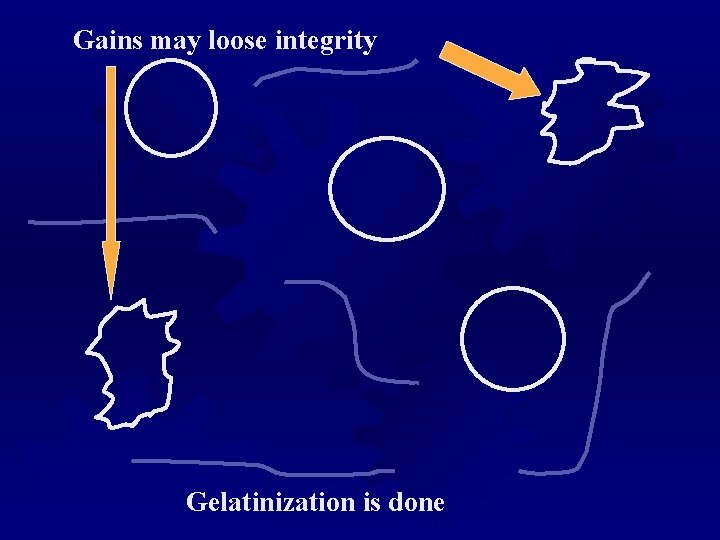
Gelatinization of Starch ® Native starch granules are insoluble in cold water, despite some “swelling” ® Heated water increases kinetic energy, breaking some intermolecular bonds, and allows water to penetrate ® The gelatinization point is where crystallinity is lost ® GTR is the temperature range over which gelatinization occurs. ® As water is bound, the viscosity increases. ® GTR is different from different starch types ® There must be enough water to break open and bind to the starch hydrogen binding sites.

Gelatinization Starch grains swell when heated in water

H-bonds break, amylose can spill from the grain

Gains may loose integrity Gelatinization is done

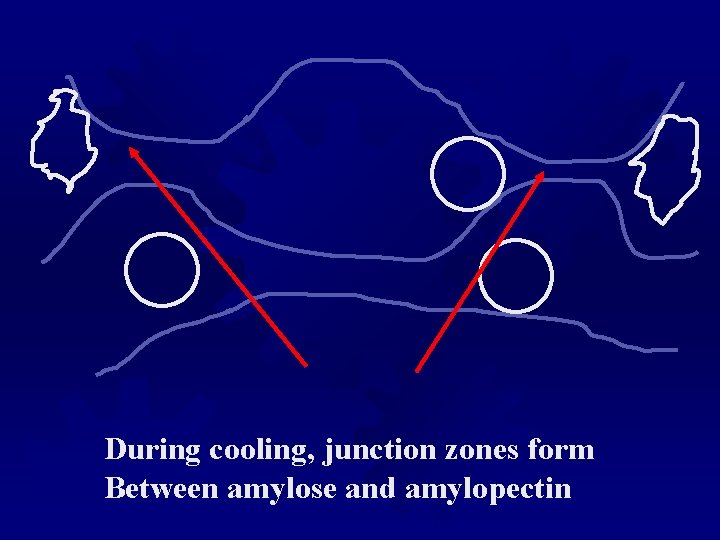
During cooling, junction zones form Between amylose and amylopectin

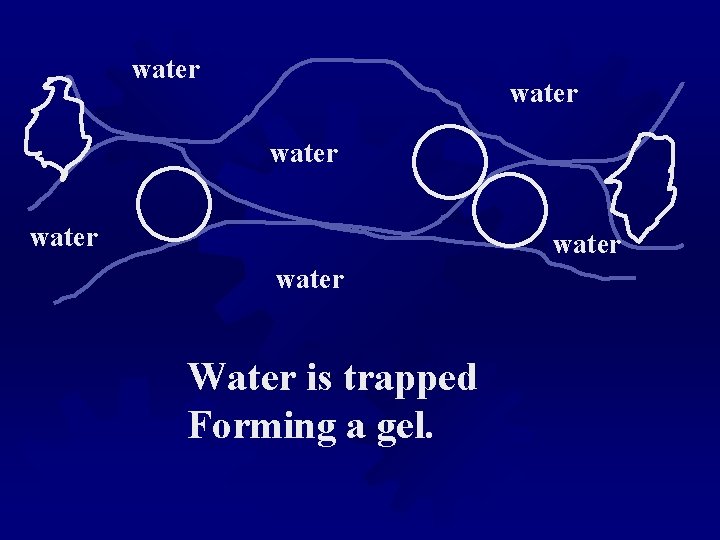
water water Water is trapped Forming a gel.

WATER As the gel dehydrates and/or junction zones Tighten, water is “squeezed” from the gel, in a syneresis process.

Starch Modifications ® Cross-linking (common modification) ® Alkali treatment (p. H 7. 5 -12) with salt Phosphorus oxychloride ® Sodium trimetaphosphate ® Adipic and acetic anhydride ® Starch phosphates formed after neutralization ®

Cross-Linking ® Resists viscosity breakdown ® Resists prolonged heating effects ® Resists high shear rates ® Resists high acid environments ® Increased viscosity ® Increased texture

Starch Modifications ® Starch Substitutions ® Adding monofunctional groups “Blocking Groups” added to the starch ® Acetyl (2. 5% max starch acetates) ® Hydroxypropyl, phosphates, ethers ® ® Slows retrogradation (re-association of amylose) ® Lowers GTR, stabilizes the starch Acetate + Starch

Starch Modifications ® Oxidation and Bleaching Hydrogen peroxide ® Ammonium persulfate ® Na/Ca hypochlorite ® l 0. 0082 lbs chlorine/pound of starch K-permanganate ® Na-chlorite ® ® Whitens the starch ® Removes carotenes and other natural pigments ® ~25% of oxidizers break C-C linages ® ~75% of oxidizers will oxidize the hydroxyl groups ® Lowers viscosity, improves clarity of gels

Polysaccharide Breakdown Products

Hydrolytic Products ® ® ® Maltose Maltitol Maltodextrins Dextrans ® ® ® Maltose = glucose disaccharide Maltitol = example of a “polyol” Maltodextrins = enzyme converted starch fragments ® Dextrins = starch fragments (α-1 -4) linkages produced by ns hydrolysis of amylose ® Dextrans = polysaccharides made by bacteria and yeast ns metabolism, fragments with mostly α (1 - 6) linkages

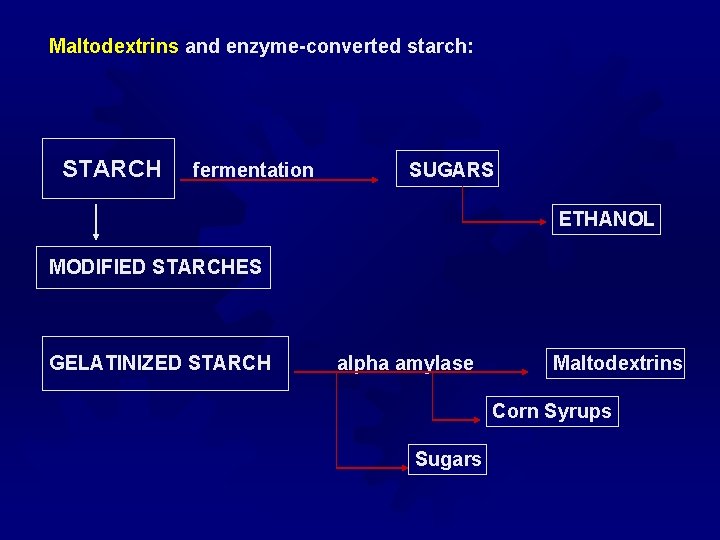
Maltodextrins and enzyme-converted starch: STARCH fermentation SUGARS ETHANOL MODIFIED STARCHES GELATINIZED STARCH alpha amylase Maltodextrins Corn Syrups Sugars

The smaller the size of the products in these reactions, the higher the dextrose equivalence (DE), and the sweeter they are Starch DE = 0 Glucose (dextrose) DE = 100 Maltodextrin (MD) DE is <20 Corn syrup solids (CS) DE is >20 Low DE syrup alpha amylase MD beta amylase High DE Syrup

Dextrinization ® A non-enzymatic method to product low- molecular weight fragments ® High heat treatment of acidified starch ® “Pyro-conversion” of starch to dextrins ® Both breaks and re-forms bonds ® Wide-range of products formed Vary in viscosity ® Solubility ® Color (white, yellow) ® Reducing capacity ® Stability ®

Hydrocolloids Binding water with carbohydrates “Gums”

“Vegetable gum” polysaccharides are substances derived from plants, including seaweed and various shrubs or trees, have the ability to hold water, and often act as thickeners, stabilizers, or gelling agents in various food products. Plant gums - exudates, seeds (guar, xanthan, locust bean, etc) Marine hydrocolloids - extracts from seaweeds (Carageenan, agar, alginates) Microbiological polysaccharides - exocellular polysaccharides Modified, natural polysaccharides

FUNCTIONS IN FOOD ® Gelation ® Viscosity ® Suspension ® Emulsification and stability ® Whipping ® Freeze thaw protection ® Fiber (dietary fiber) Gut health ® Binds cholesterol ®

STRUCTURAL CONSIDERATIONS ® Electrical charge, p. H sensitive ® Interactions with Oppositely charged molecules ® Salts ® Acids ® ® Chain length ® Longer chains are more viscous ® Linear vs Branched chains ® Inter-entangled, enter-woven molecules

“Structural” Polysaccharides Cellulose ® Polymer of glucose linked ß-1, 4 Hemicellulose ® Similar to cellulose ® Consist of glucose and other monosaccharides ® Arabinose, xylose, other 5 -carbon sugars Pectin ® Polymer of galacturonic acid

MODIFIED CELLULOSES ® Chemically modified cellulose ® Do not occur naturally in plants ® Similar to starch, but β-(1, 4) glycosidic bonds ® Carboxymethyl cellulose (CMC) most common ® Acid treatment to add a methyl group ® Increases water solubility, thickening agent ® Sensitive to salts and low p. H ® Fruit fillings, custards, processed cheeses, high fiber filler

PECTINS ® Linear polymers of galacturonic acid Gels form with degree of methylation of its carboxylic acid groups ® Many natural sources ® ® Susceptible to degrading enzymes Polygalacturonase (depolymerize) ® Pectin esterases (remove methyl groups) ® ® Longer polymers, higher viscosity ® Lower methylation, lower viscosity ® Increase electrolytes (ie. metal cations), higher viscosity ® p. H and soluble solids impact viscosity

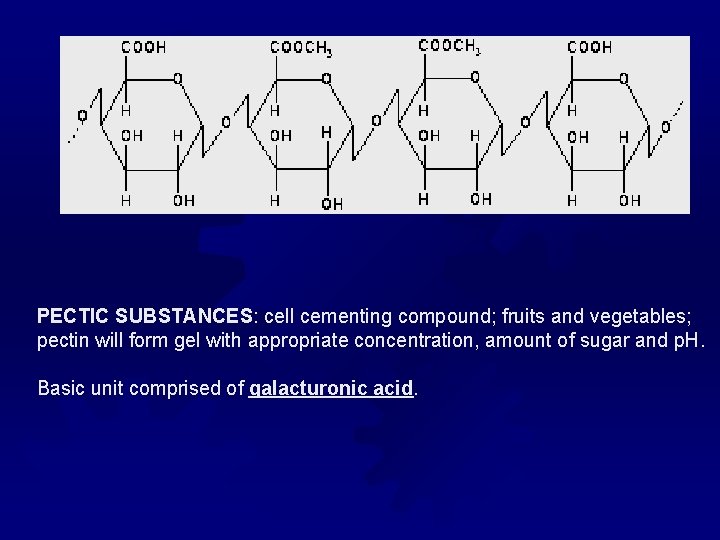
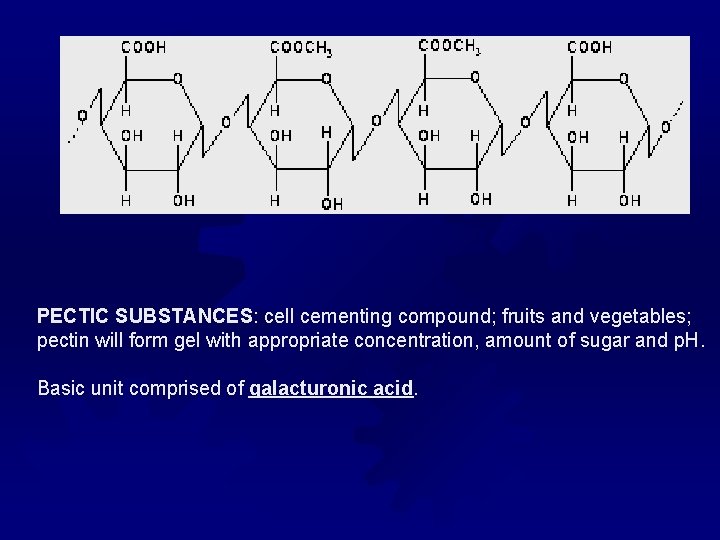
PECTIC SUBSTANCES: cell cementing compound; fruits and vegetables; pectin will form gel with appropriate concentration, amount of sugar and p. H. Basic unit comprised of galacturonic acid

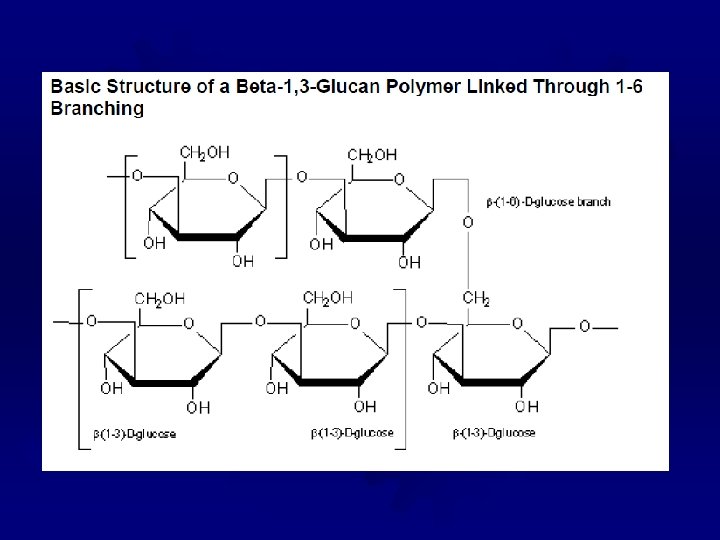
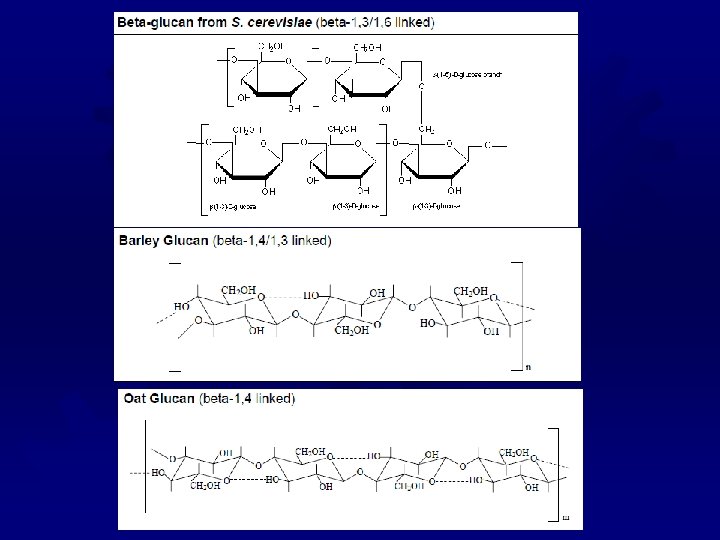
BETA-GLUCANS ® Extracts from the bran of barley and oats ® Long glucose chains with mixed ß-linkages ® Very large (~250, 000 glucose units) ® Water soluble, but have a low viscosity ® Can be used as a fat replacer ® Responsible for the health claims (cholesterol) for whole oat products ® Formulated to reduce the glycemic index of a food

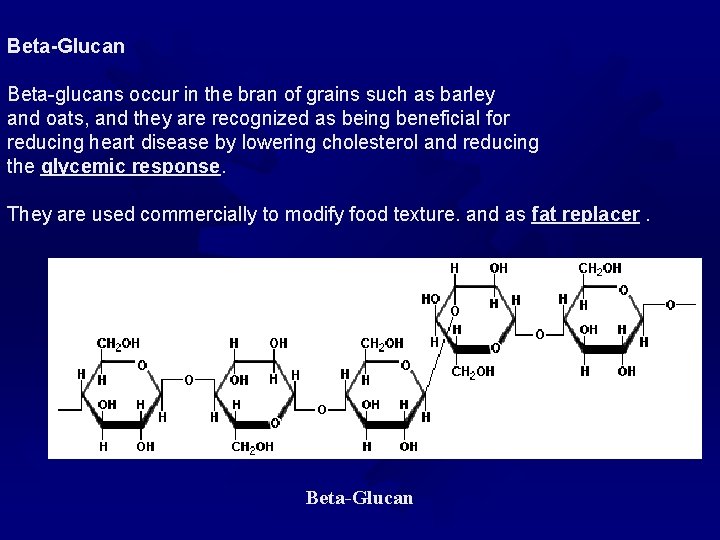
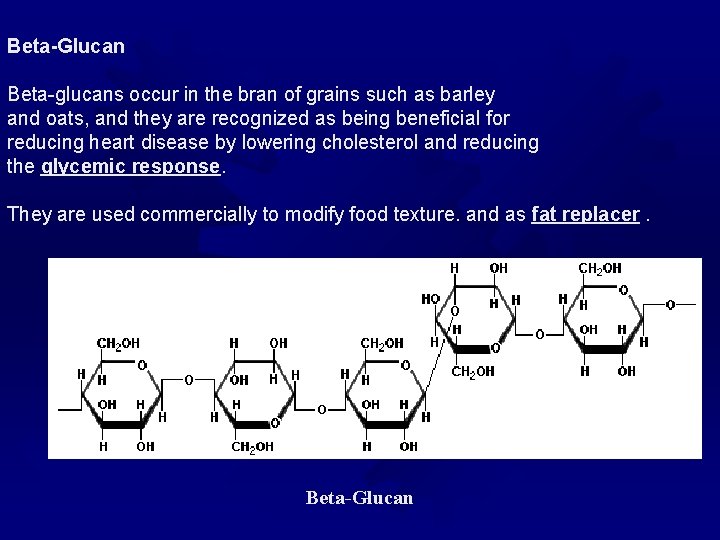
Beta-Glucan Beta-glucans occur in the bran of grains such as barley and oats, and they are recognized as being beneficial for reducing heart disease by lowering cholesterol and reducing the glycemic response They are used commercially to modify food texture. and as fat replacer Beta-Glucan

Others CHITIN ® Polymer of N-Acetyl-D-glucosamine ® Found in the exoskeleton of insects and shellfish ® Many uses in industry, food and non-food. INULIN ® Chains of fructose that end in a glucose molecule Generally a sweet taste ® Isolated from Jerusalem artichokes and chicory ® Act as a dietary fiber ® Potentially a pre-biotic compound ®

Paper Review Producing fructo-oligosaccharides: For Tuesday

Starch ® Starch must be cooked to act as a thickening agent ® Pre-gelatinized starch is made by quickly cooking a starch and drying the product. ® Pre-gelatinized starch rapidly re-hydrates without further cooking ® Useful thickening agent ® Can be used in dried sauces and salad dressings ® Used in products that do not require more cooking

Starch ® Starch suspensions are not stable to heating ® Swollen starch granules break down in hot, stirred or acidic conditions ® Combinations (ie. heat and acid) will depolymerize ® Cross-linking can help stabilize and slow or maybe prevent breakdown

Starch ® Starch gels change their properties during storage ® Slow retrogradation of amylopectin is common ® The texture of a starch gel will change and show some syneresis. ® Again, modified starch will resist changes during storage ® Starch acetates or phosphates are common modifications, altering the helical arrangements, and slow or inhibit retrogradation. ® All stabilized starches must also be labeled as “modified starch” on an ingredient list.

“Vegetable gum” polysaccharides are substances derived from plants, including seaweed and various shrubs or trees, have the ability to hold water, and often act as thickeners, stabilizers, or gelling agents in various food products. Plant gums - exudates, seeds (guar, xanthan, locust bean, etc) Marine hydrocolloids - extracts from seaweeds (Carageenan, agar, alginates) Microbiological polysaccharides - exocellular polysaccharides Modified, natural polysaccharides

FUNCTIONS IN FOOD ® Gelation ® Viscosity ® Suspension ® Emulsification and stability ® Whipping ® Freeze thaw protection ® Fiber (dietary fiber) Gut health ® Binds cholesterol ®

STRUCTURAL CONSIDERATIONS ® Electrical charge, p. H sensitive ® Interactions with Oppositely charged molecules ® Salts ® Acids ® ® Chain length ® Longer chains are more viscous ® Linear vs Branched chains ® Inter-entangled, enter-woven molecules

“Structural” Polysaccharides Cellulose ® Polymer of glucose linked ß-1, 4 Hemicellulose ® Similar to cellulose ® Consist of glucose and other monosaccharides ® Arabinose, xylose, other 5 -carbon sugars Pectin ® Polymer of galacturonic acid

MODIFIED CELLULOSES ® Chemically modified cellulose ® Do not occur naturally in plants ® Similar to starch, but β-(1, 4) glycosidic bonds ® Carboxymethyl cellulose (CMC) most common ® Acid treatment to add a methyl group ® Increases water solubility, thickening agent ® Sensitive to salts and low p. H ® Fruit fillings, custards, processed cheeses, high fiber filler

PECTINS ® Linear polymers of galacturonic acid Gels form with degree of methylation of its carboxylic acid groups ® Many natural sources ® ® Susceptible to degrading enzymes Polygalacturonase (depolymerize) ® Pectin esterases (remove methyl groups) ® ® Longer polymers, higher viscosity ® Lower methylation, lower viscosity ® Increase electrolytes (ie. metal cations), higher viscosity ® p. H and soluble solids impact viscosity

PECTIC SUBSTANCES: cell cementing compound; fruits and vegetables; pectin will form gel with appropriate concentration, amount of sugar and p. H. Basic unit comprised of galacturonic acid

BETA-GLUCANS ® Extracts from the bran of barley and oats ® Long glucose chains with mixed ß-linkages ® Very large (~250, 000 glucose units) ® Water soluble, but have a low viscosity ® Can be used as a fat replacer ® Responsible for the health claims (cholesterol) for whole oat products ® Formulated to reduce the glycemic index of a food

Beta-Glucan Beta-glucans occur in the bran of grains such as barley and oats, and they are recognized as being beneficial for reducing heart disease by lowering cholesterol and reducing the glycemic response They are used commercially to modify food texture. and as fat replacer Beta-Glucan



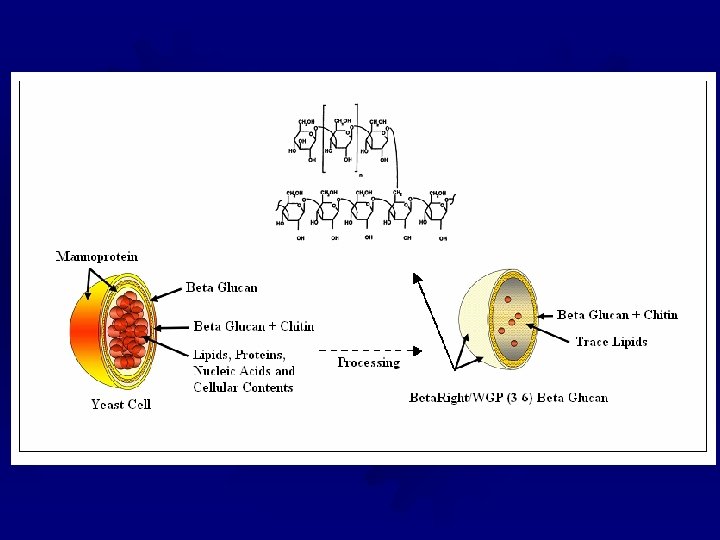
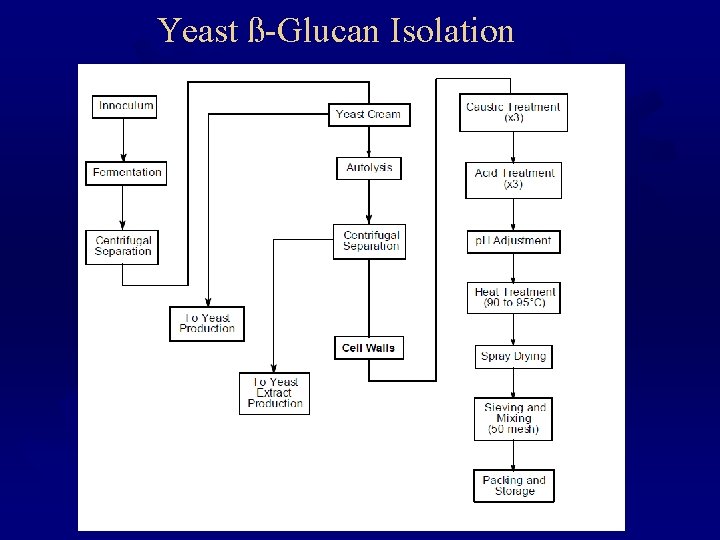
Yeast ß-Glucan Isolation


Sugar Reactions (Gluconic acid) (Glucuronic acid)

Properties of Glucose ® C 1 of glucose is the carbonyl carbon ® Glucose has 4 chiral centers ® Non-super-imposable on its mirror image ® Carbons 2, 3, 4, 5 are chiral carbons ® The carbonyl carbon (C 1) is also the site of many reactions involving glucose ® They have two enantiomeric forms, D and L, depending on the location of the hydroxyl group at the chiral carbons.

Sugars ® They have two enantiomeric forms, D and L, depending on the location of the hydroxyl group at the chiral carbons. ® An enantiomer is one of two stereoisomers that are mirror images of each other, non-superposable. ® Isomerism in which two isomers are mirror images of each other. (D vs L). ® Vary in their 3 -D space

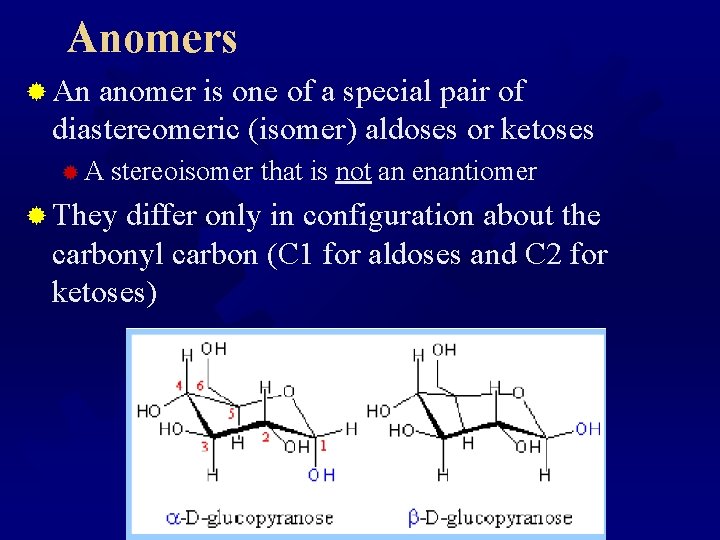
Anomers ® An anomer is one of a special pair of diastereomeric (isomer) aldoses or ketoses ® A stereoisomer that is not an enantiomer ® They differ only in configuration about the carbonyl carbon (C 1 for aldoses and C 2 for ketoses)

Carbonyl Carbons ® Carbonyl carbons are subject to nucleophilic attack, since it is electron deficient. ® Electrons are drawn to this site ® -OH groups on the sugar act as the nucleophile, and add to the carbonyl carbon to recreate the ring form

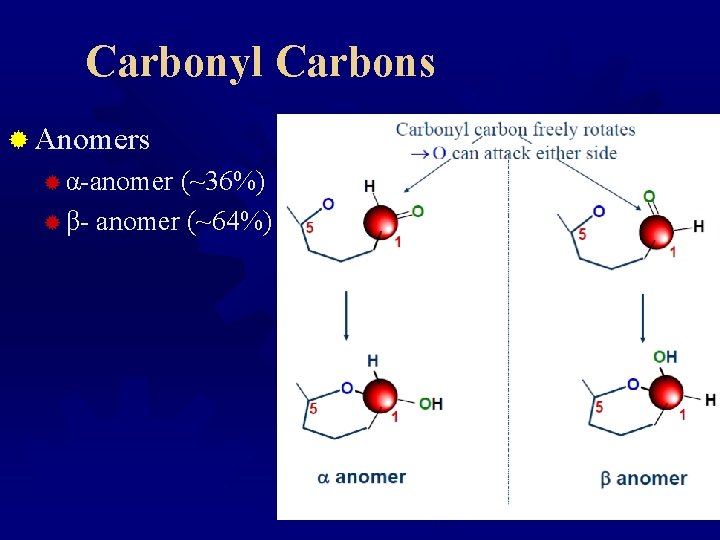
Carbonyl Carbons ® Anomers ® α-anomer (~36%) ® β- anomer (~64%)

Sugar Anomers => Mutarotation ® Interconversion of α- and β- anomers ® The α- and β- anomers of carbohydrates are typically stable. ® In solution, a single molecule can interchange between straight and ring form ® different ring sizes ® α and β anomeric isomers ® ® The process is ® dynamic equilibrium ® due to reversibility of reaction ® All isomers can potentially exist in solution ® energy/stability of different forms vary

Mutarotation ® α- and β- anomers

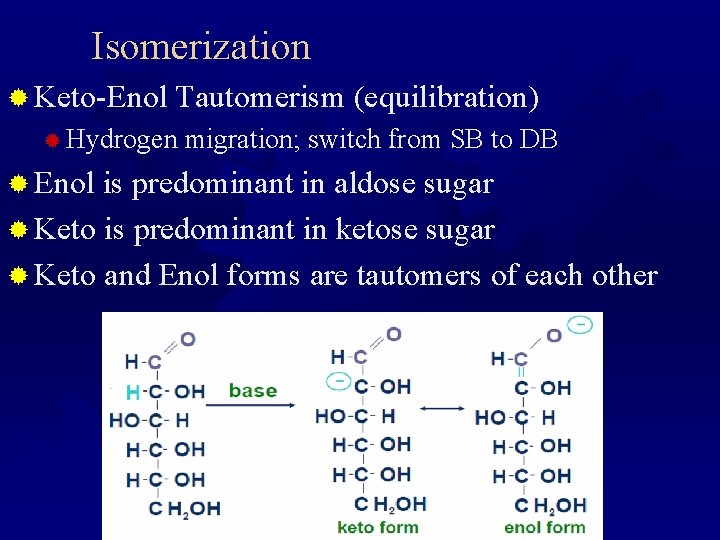
Isomerization ® Keto-Enol Tautomerism (equilibration) ® Hydrogen migration; switch from SB to DB ® Enol is predominant in aldose sugar ® Keto is predominant in ketose sugar ® Keto and Enol forms are tautomers of each other

Isomerization ® Glucose and mannose are enantiomers, but with dramatically different properties ® Glucose and fructose are isomers

Pectins in Foods

Plant Cell Water-Filled

PECTINS ® Linear polymers of galacturonic acid Gels form with degree of methylation of its carboxylic acid groups ® Many natural sources ® ® Susceptible to degrading enzymes Polygalacturonase (depolymerize); PG ® Pectin esterases (remove methyl groups), PME ® ® Longer polymers, higher viscosity ® Lower methylation, lower viscosity ® Increase electrolytes (ie. metal cations), higher viscosity ® p. H and soluble solids impact viscosity

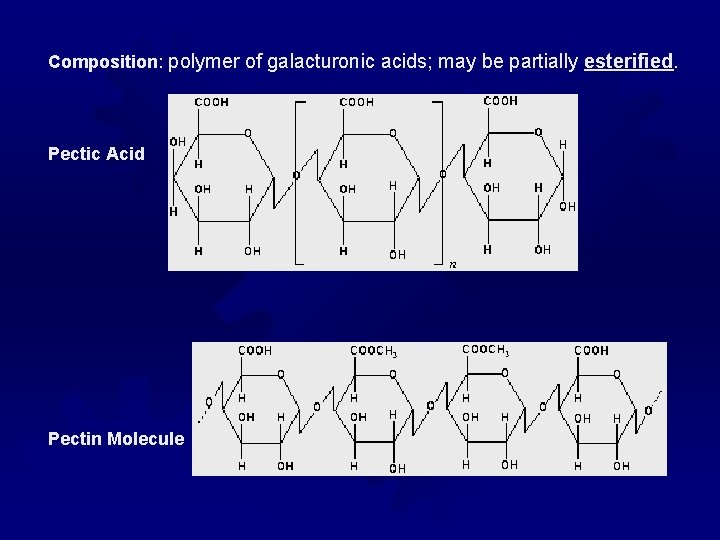
Composition: polymer of galacturonic acids; may be partially esterified Pectic Acid Pectin Molecule

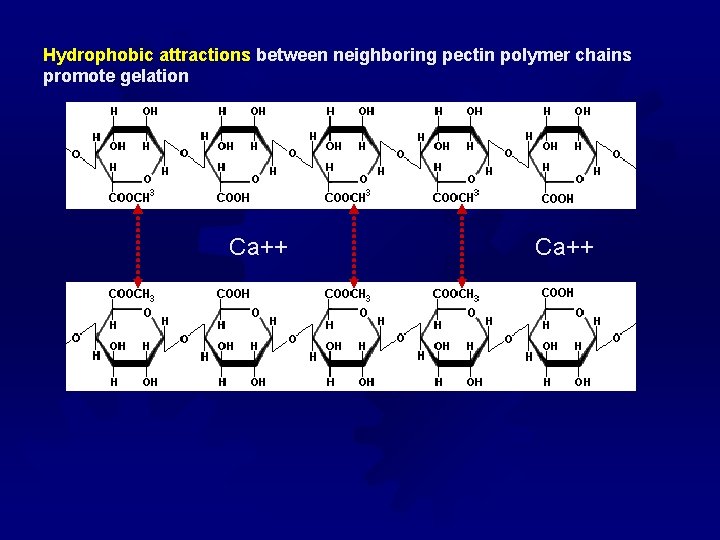
Pectins ® Pectins are important because they form gels ® Mechanism of gel formation differs by the degree of esterification (DE) of the pectin molecules ® DE refers to that percentage of pectin units with a methyl group attached ® Free COOH groups can crosslink with divalent cations ® Sugar and acid under certain conditions can contribute to gel structure and formation ® LM pectin “low methoxyl pectin” has DE < 50% ; gelatin is pectin” controlled by adding cations (like Ca++ and controlling the p. H) ® HM pectin “high methoxyl pectin” has DE >50% and forms a gel pectin under acidic conditions by hydrophobic interactions and Hbonding with dissolved solids (i. e. sugar)

Hydrophobic attractions between neighboring pectin polymer chains promote gelation Ca++

Proteins ® Many important functions ® Functional ® Nutritional ® Biological ® Enzymes ® Structurally complex and large compounds ® Major source of nitrogen in the diet ® By weight, proteins are about 16% nitrogen

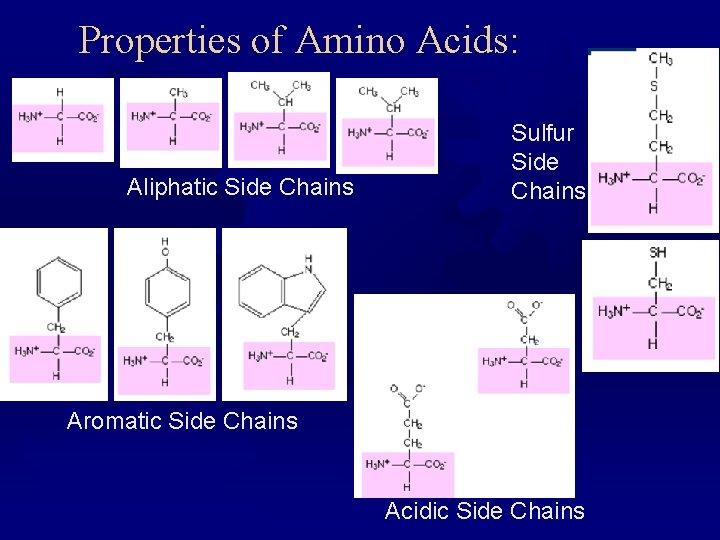
Properties of Amino Acids ® Aliphatic chains: Gly, Ala, Val, Leucine, Ile ® Hydroxy or sulfur side chains: Ser, Thr, Cys, Met ® Aromatic: Phe, Trp, Try ® Basic: His, Lys, Arg ® Acidic and their amides: Asp, Asn, Glu, Gln

Properties of Amino Acids: Aliphatic Side Chains Sulfur Side Chains Aromatic Side Chains Acidic Side Chains

Properties of Amino Acids: ® Zwitterions are electrically neutral, but carry a “formal” positive or negative charge.

The Zwitterion Nature ® Zwitterions make amino acids good acid-base buffers. ® Accepting H+ is acidic environments; donating H+ in basic environments ® For proteins and amino acids, the p. H at which they have no net charge in solution is called the Isoelectric Point of p. I (i. e. IEP). ® The solubility of a protein depends on the p. H of the solution. ® Similar to amino acids, proteins can be either positively or negatively charged due to the terminal amine -NH 2 and carboxyl (-COOH) groups. ® ® Proteins are positively charged at low p. H and negatively charged at high p. H. When the net charge is zero, we are at the IEP. ® A charged protein helps interactions with water and increases its solubility. ® As a result, protein is the least soluble when the p. H of the solution is at its isoelectric point.

Physical Nature of Proteins

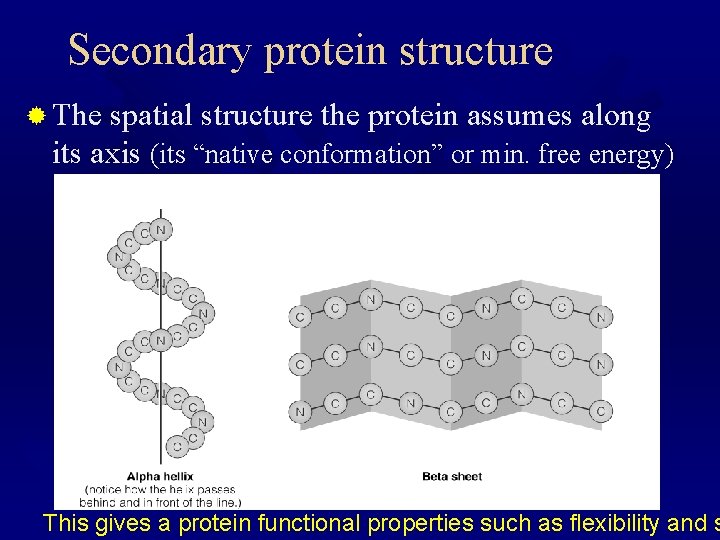
Secondary protein structure ® The spatial structure the protein assumes along its axis (its “native conformation” or min. free energy) This gives a protein functional properties such as flexibility and s

Tertiary Structure of Proteins ® 3 -D organization of a polypeptide chain ® Compacts proteins ® Interior is mostly devoid of water or charge groups 3 -D folding of chain

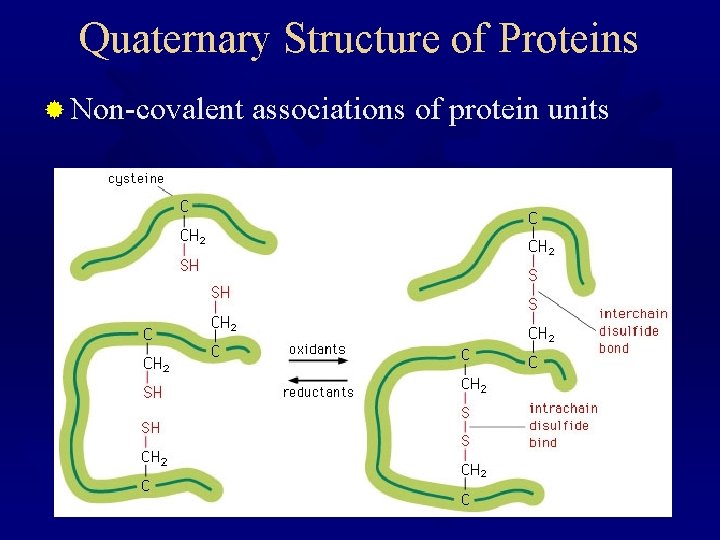
Quaternary Structure of Proteins ® Non-covalent associations of protein units

Proteins Changes in structure ® Denaturation ® Breaking of any structure except primary ® Examples: ® Heat ® Salt/Ions ® Alcohol ® p. H extremes ® Shear ® Enzymes

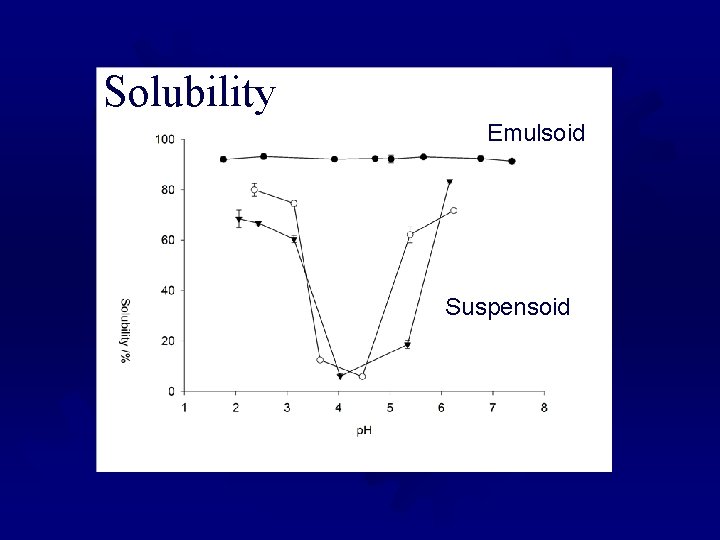
Emulsoids and Suspensiods ® Proteins should be thought of as solids ® Not in true solution, but bond to a lot of water ® Can be described in 2 ways: ® Emulsoids- have close to the same surface charge, with many shells of bound water ® Suspensoids- colloidal particles that are suspended by charge alone

Functional Properties of Proteins 3 major categories ® Hydration properties ® ® Structure formation ® ® Protein to water interactions Dispersibility, solubility, adhesion, Water holding capacity, viscosity Protein to protein interactions Gel formation, precipitation, Aggregation Surface properties ® ® Protein to interface interactions Foaming, emulsification

1. Hydration Properties (hydration) ® Proteins are important hydrocolloids ® As ingredients, many are sold as dry powders ® Hydrating and processing w/o denaturation ® Solubility- Mostly, denatured proteins are less soluble than native proteins ® Many (but not all) proteins (particularly suspensoids) aggregate or precipitate at their isoelectric point (IEP) ® Protein viscosity is influenced by amount, size, shape, p. H, water content, and solubility of the proteins

2. Structure Formation (protein interactions) ® Gels – a 3 -D network of protein and water. ® Attractive and repulsive forces between adjacent polypeptides ® Gelation- when denatured proteins aggregate and form an ordered protein matrix ® Water absorption and thickening ® Formation of viscous, solid, or visco-elastic gels ® For many proteins, heated followed by cooling forms the gel ® Texturization – Proteins are responsible for the structure and texture of many foods ® Meat, bread dough, gelatin ® Texturized proteins are modified with salts, acid/alkali, oxidants/reductants ® ® “Pink Slime” Can also be processed to mimic other proteins (i. e. surimi)

3. Surface Properties (interfaces) ® Emulsions- Exposure of protein hydrophobic regions to lipids (ie. tertiary structures) Not all proteins make good emulsifiers ® Can strengthen a normal emulsion system ® ® Foams- trapping gas bubbles in a viscous medium ® Protein is usually soluble ® Air bubble size is critical (nebulized air) ® Duration and shear rate ® Temperature and physical kinetics ® Food ingredient interactions (i. e. salt, acid, and lipids). . bad. ® Metal ions, hydrocolloids, and sugar can increase stability

Enzymes

Enzyme Influencing Factors ® Temperature-dependence of enzymes ® Every enzyme has an optimal temperature for maximal activity ® The effectiveness of an enzyme: Enzyme activity ® For most enzymes, it is 30 -40°C ® Many enzymes denature >45°C ® Each enzyme is different, and vary by isozymes ® Often an enzyme is at is maximal activity just before it denatures at its maximum temperature

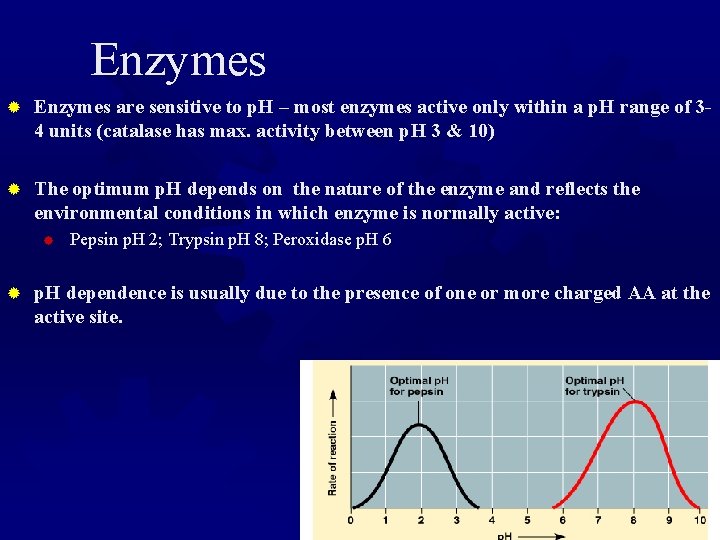
p. H ® Like temp, enzymes have an optimal p. H where they are maximally active ® Generally between 4 and 8 ® with many exceptions ® Most have a very narrow p. H range where they show activity. ® This influences their selectivity and activity.

Water Activity ® Enzymes need “free” water to operate ® Low Aw foods have slower enzyme reactions Ionic Strength ® Some ions may be needed by active sites on the protein (salting in) ® Ions may be a link between the enzyme and substrate ® Ions change the surface charge on the enzyme ® Ions may block, inhibit, or remove an inhibitor ® Others, enzyme-specific

Common Enzymes in Foods ® Polyphenol oxidase ® Plant cell wall degrading enzymes ® Proteases ® Lipases ® Peroxidase/Catalase ® Amylase ® Ascorbic acid oxidase ® Lipoxygenase

Enzyme Influencing Factors ® Enzymes are proteins that act as biological catalysts ® They are influenced in foods by: Temperature ® p. H ® Water activity ® Ionic strength (ie. Salt concentrations) ® Presence of other agents in solution ® ® Metal chelators Reducing agents Other inhibitors Also factors for Inhibition, including: Oxygen exclusion and Sulfites

The “Raw Foods” Movement ® Enzymes present in raw foods help in digesting the foods we eat ® ® Cooking destroys food enzymes forcing the body to produce more of its own digestive enzymes ® ® Eating these enzymes saves your both the work. Our body has a finite amount of enzyme producing potential ® ® ® But they have to enter the digestive system. The more enyzmes we eat, the more we preserve health and longevity Our digesting enzyme potential can be exhausted. Enzymes in raw food also carry our "life force" ® When our ability to produce digestive enzymes is exausted, we die.

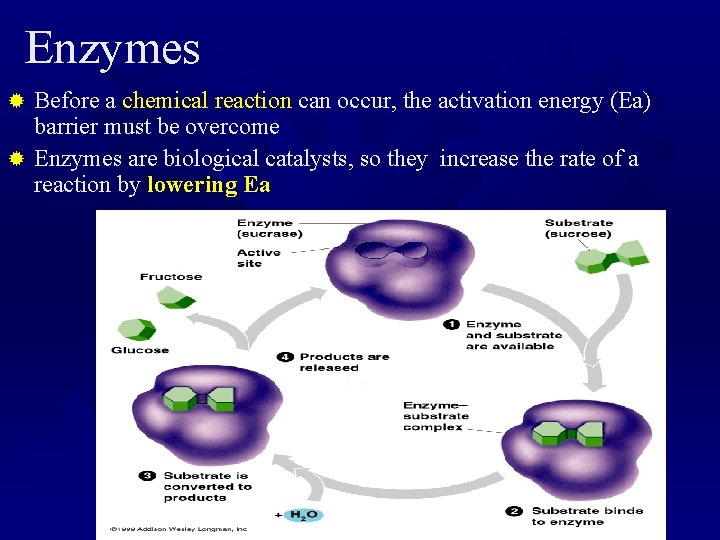
Enzymes Before a chemical reaction can occur, the activation energy (Ea) barrier must be overcome ® Enzymes are biological catalysts, so they increase the rate of a reaction by lowering Ea ®

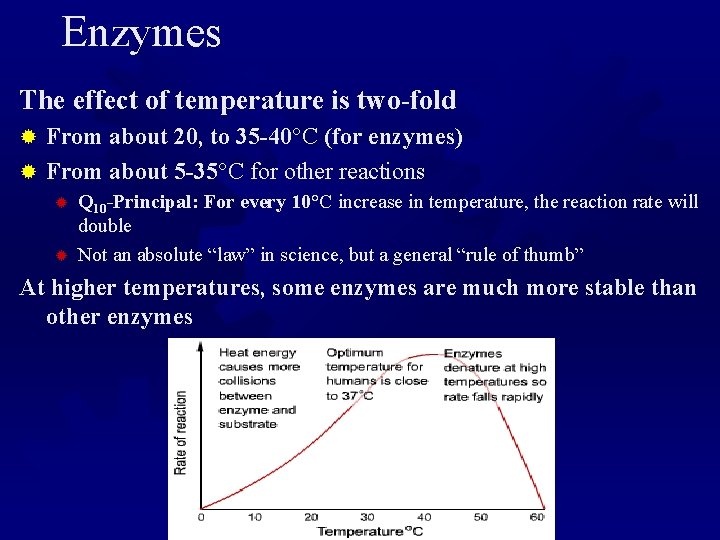
Enzymes The effect of temperature is two-fold From about 20, to 35 -40°C (for enzymes) ® From about 5 -35°C for other reactions ® ® ® Q 10 -Principal: For every 10°C increase in temperature, the reaction rate will double Not an absolute “law” in science, but a general “rule of thumb” At higher temperatures, some enzymes are much more stable than other enzymes

Enzymes ® Enzymes are sensitive to p. H – most enzymes active only within a p. H range of 34 units (catalase has max. activity between p. H 3 & 10) ® The optimum p. H depends on the nature of the enzyme and reflects the environmental conditions in which enzyme is normally active: ® ® Pepsin p. H 2; Trypsin p. H 8; Peroxidase p. H 6 p. H dependence is usually due to the presence of one or more charged AA at the active site.

Worthington Enzyme Manual http: //www. worthingtonbiochem. com/index/manual. html

Nomenclature Each enzyme can be described in 3 ways: ® Trivial name: -amylase ® Systematic name: -1, 4 -glucan-glucono-hydrolase substrate ® reaction Number of the Enzyme Commission: E. C. 3. 2. 1. 1 3 - hydrolases (class) ® 2 - glucosidase (sub-class) ® 1 - hydrolyzing O-glycosidic bond (sub-class) ® 1 - specific enzyme ®

Enzyme Class Characterizations 1. Oxidoreductase Oxidation/reduction reactions 2. Transferase Transfer of one molecule to another (i. e. functional groups) 3. Hydrolase Catalyze bond breaking using water (ie. protease, lipase) 4. Lyase Catalyze the formation of double bonds, often in dehydration reations 5. Isomerase Catalyze intramolecular rearrangement of molecules 6. Ligase Catalyze covalent attachment of two substrate molecules

Enzyme Commission Enzyme Nomenclature ® International Union of Biochemistry and Molecular Biology (IUBMB) ® International Union of Pure and Applied Chemistry (IUPAC) ® Joint Commission on Biochemical Nomenclature (JCBN) ® IUPAC-IUBMB-JCBN ® http: //www. chem. qmul. ac. uk/iubmb/enzyme/

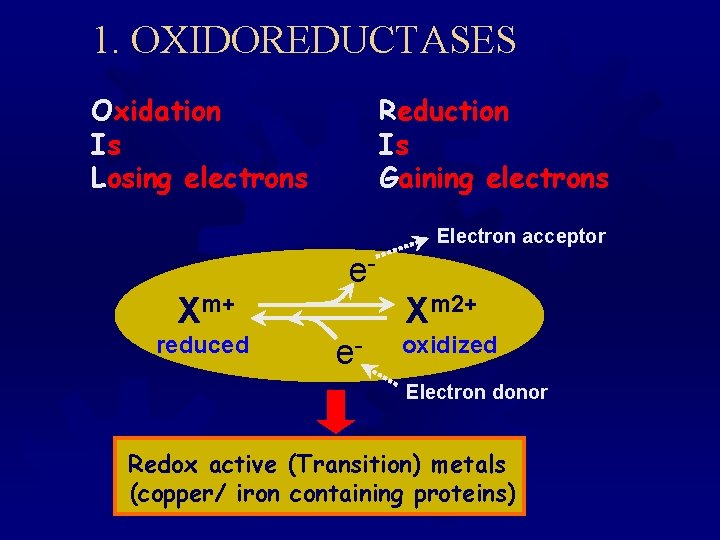
1. OXIDOREDUCTASES Oxidation Is Losing electrons Reduction Is Gaining electrons Electron acceptor e. Xm+ reduced Xm 2+ e- oxidized Electron donor Redox active (Transition) metals (copper/ iron containing proteins)

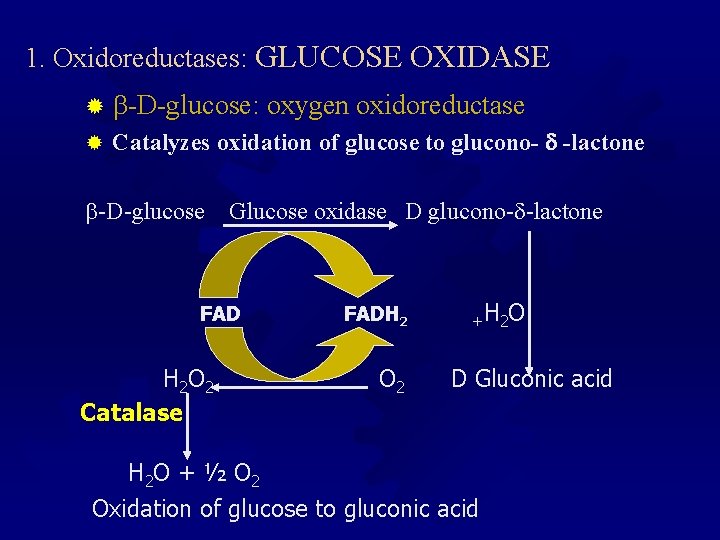
1. Oxidoreductases: GLUCOSE OXIDASE ® -D-glucose: oxygen oxidoreductase ® Catalyzes oxidation of glucose to glucono- -lactone -D-glucose Glucose oxidase D glucono- -lactone FAD H 2 O 2 Catalase FADH 2 O 2 +H 2 O D Gluconic acid H 2 O + ½ O 2 Oxidation of glucose to gluconic acid

How Glucose Oxidase + Catalase Works: GO Reaction 1: Glucose + O 2 Gluconic acid + H 2 O 2 CAT Reaction 2: H 2 O 2 H 2 O + 1/2 O 2 GO/CAT Reaction 3: Glucose + 1/2 O 2 Gluconic acid

1. Oxidoreductases: PEROXIDASE (POD) Donor: Hydrogen peroxide oxidoreductase ® Iron-containing enzyme. Has a heme prosthetic group N N Fe N ® Thermo-resistant – denaturation at ~ 85 o. C ® Since is thermoresistant - indicator of proper blanching (no POD activity in properly blanched vegetables) N

1. Oxidoreductases: Catalase Hydrogen peroxide oxidoreductase ® Catalyzes conversion of 2 molecules of H 2 O 2 into water and O 2: H 2 O 2 ---------- ® ® ® H 2 O +1/2 O 2 Uses H 2 O 2 When coupled with glucose oxidase the net result is uptake of ½ O 2 per molecule of glucose Occurs in MO, plants, animals

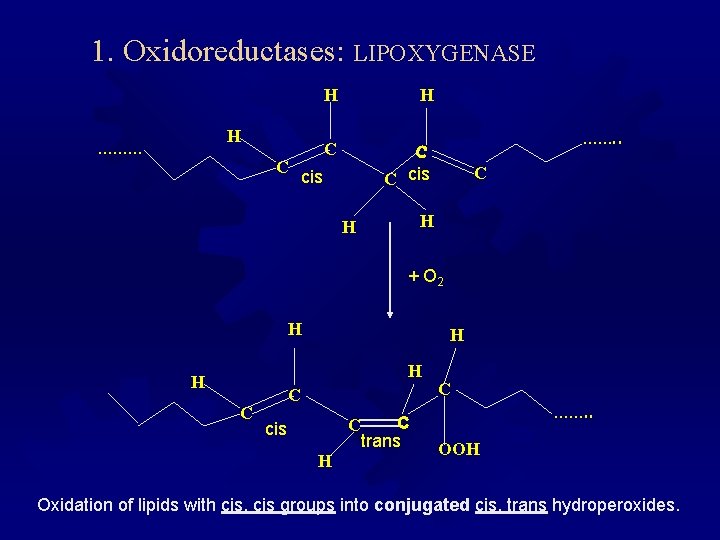
1. Oxidoreductases: LIPOXYGENASE H ……… C H H C C C cis ……. . C H H + O 2 H H C C C cis H C trans C ……. . OOH Oxidation of lipids with cis, cis groups into conjugated cis, trans hydroperoxides.

Enzymatic Determination of Starch or other simple sugars PRINCIPLE ® Starch is hydrolyzed into glucose units by enzymatic conversion ® ® D-glucose can then be quantified by enzymatic methods

1. Oxidoreductases: POLYPHENOLOXIDASES (PPO) Phenolases, PPO ® Copper-containing enzyme ® Oxidizes phenolic compounds to o-quinones: ® Catalyze conversion of mono-phenols to o-diphenols ® In all plants; high level in potato, mushrooms, apples, peaches, bananas, tea leaves, coffee beans Tea leaf tannins Catechins Procyanidins Gallocatechins Catechin gallates PPO O 2 o-Quinone + H 2 O Colored products

Worthington Enzyme Manual http: //www. worthingtonbiochem. com/index/manual. html

Functional Proteins

Protein Functionality ® Hydrodynamic-Aggregation ® Viscosity, Elasticity, Viscoelasticity ® Solubility, Water holding capacity ® Hydrophobic- Surface Active ® Emulsion and foam stabilization ® Flavor binding

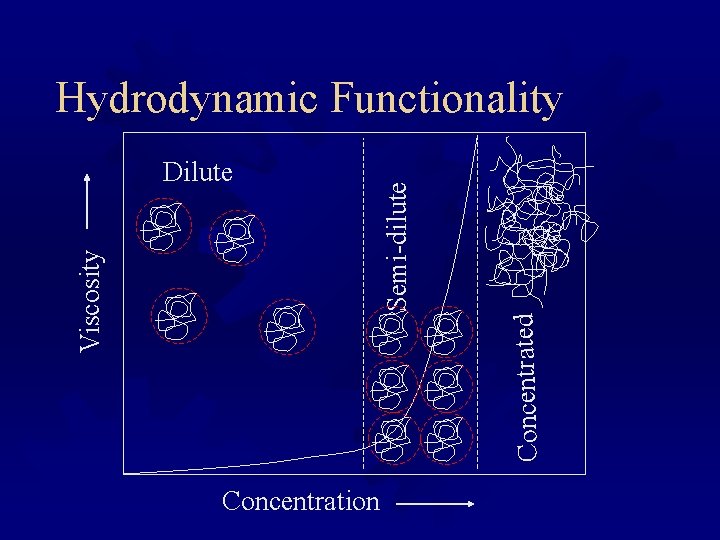
Concentrated Viscosity Dilute Semi-dilute Hydrodynamic Functionality Concentration

Viscosity ® A property of liquids ® Viscosity is the resistance to flow. The amount of energy you need to expend to get a given flow rate. ® Stress (force per unit area) is proportional to rate of strain (i. e. , flow rate) ® Particles of any type in a fluid will increase its viscosity ® Large, well hydrated polymers contribute most to viscosity

Elasticity ® A property of solids ® Elasticity is the force to achieve a given percentage change in length ® Stress (force per unit area) is proportional to strain (fractional deformation) ® An elastic material must have some solid-like network throughout the structure ® The more load bearing structures the more elastic ® The more inter-structure links the more elastic

Viscoelasticity ® Many materials simultaneously show solid and liquid like properties ® If they are stretched they will partly and slowly return to their original shape ® Elastic solids would completely recover ® Viscous liquids would retain their shape

Water Binding ® Gels contain pores ® Water can flow out of the pores ® If the gel contracts it may expel liquid SYNERESIS ® Due to closer association of protein with protein

Solubility Emulsoid Suspensoid

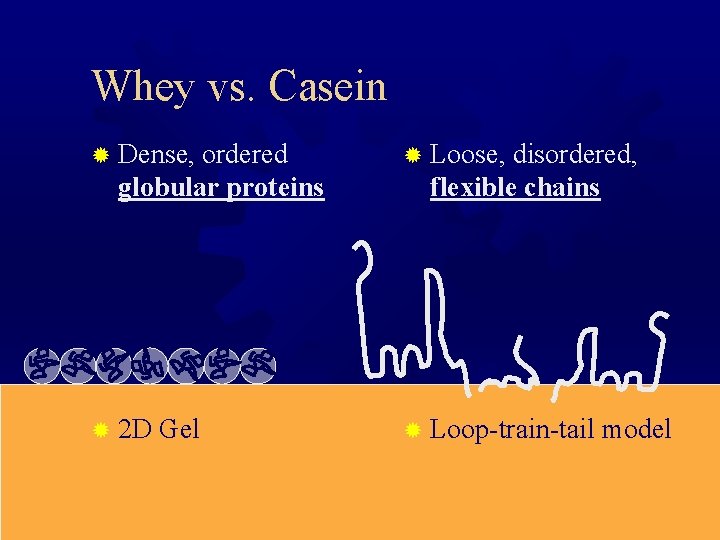
Whey vs. Casein ® Dense, ordered globular proteins ® 2 D Gel ® Loose, disordered, flexible chains ® Loop-train-tail model

Practical Applications A quick stroll through the literature…

WH= whole hydrolysate




Story Behind the Story ® Amy-Acrylamide ® Andrea-Maillard ingredients

Effect of Citric Acid and Glycine Addition on Acrylamide and Flavor in a Potato Model System Class discussion; Bianca and Cassie

A quick review Protein Analysis Methods

















