Food Digestion Carbohydrate s Carbohydrates include sugars and

Food & Digestion

Carbohydrate s Carbohydrates include sugars and starch Used in respiration to produce energy Marathon runners often eat pasta the evening before a race Too much sugar leads to tooth decay, obesity and sometimes diabetes

Testing for Starch Grind up a little food with a little water using a mortar and pestle to produce a slurry. Add 2 drops of iodine to a spotting tile Add 2 drops of the slurry to the iodine A blue-black colour shows that starch is present There is not need to heat the mixture! = starch present = starch absent

Testing for Sugars Grind up a little food with a little water using a mortar and pestle to produce a slurry. Transfer to a test tube to about 1 cm depth Add 5 drops of Benedict’s solution to the tube Heat gently in a water bath (beaker of water) = sugar present

Proteins Used for growth and repair Only used in respiration to during periods of starvation Body builders have a high protein Protein is also needed to produce antibodies which help us to fight disease

Testing for Proteins Grind up a little food with a little water using a mortar and pestle to produce a slurry. Transfer to a test tube to about 1 cm depth Add 5 drops of copper sulphate solution and 5 drops of sodium hydroxide Do not heat! = protein present The stronger the purple colouration, the more protein present.
Good fats Fats Bad fats Fats contain more energy than carbohydrates and proteins They are harder to digest and best not eaten before a marathon! Eating large amounts of fat can lead to obesity and heart disease Polyunsaturated fats are much healthier than saturated fats Unsaturated Fats can be trans fat or cis fat. Trans fats are rarely found in naturally growing food other than small amounts in cattle and sheep. Today the largest amount of trans fat consumed is created by the processed food industry. Trans fats greatly increase the risk of heart disease.

Testing for Fats There are several methods to test for fat. For method 1 smear the food over grease proof paper (or tracing paper). Then shine a light through it (hold up to the window). If it looks translucent, fat is present (spot test) For method 2 grind up a little food with 2 cm 3 ethanol (which dissolves fat) and then transfer the clear liquid to a test tube to 1 cm depth. Take a second tube and add water to 1 cm depth. Mix the contents of the two tubes. The cloudier the resulting mixture, the greater the amount of fat. The fat forms droplets of fat in water/ethanol mixture known as an emulsion. This is sometimes therefore called the emulsion test.

Summary

Vitamins are substances needed in very small amounts. They cannot be made by the human body (except vitamin D), so they need to be eaten in our diet. Some are water soluble and often found in fruit and vegetables. Some are fat soluble and found in vegetable oils and oily fish.
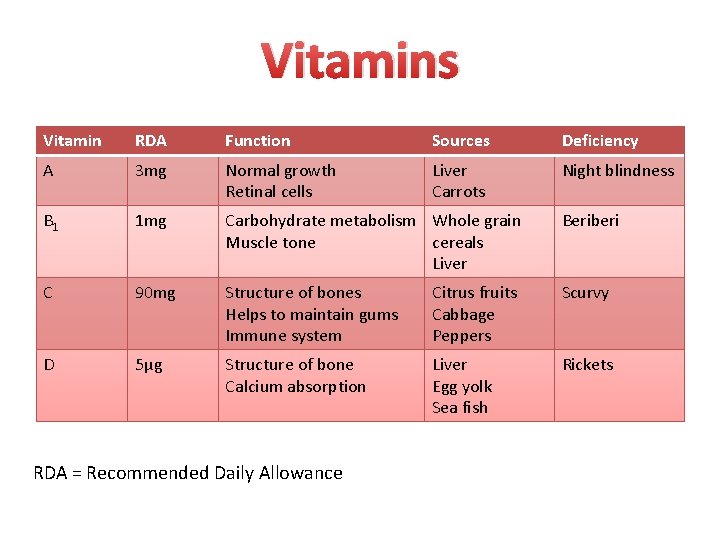
Vitamins Vitamin RDA Function Sources Deficiency A 3 mg Normal growth Retinal cells Liver Carrots Night blindness B 1 1 mg Carbohydrate metabolism Whole grain Muscle tone cereals Liver Beriberi C 90 mg Structure of bones Helps to maintain gums Immune system Citrus fruits Cabbage Peppers Scurvy D 5µg Structure of bone Calcium absorption Liver Egg yolk Sea fish Rickets RDA = Recommended Daily Allowance
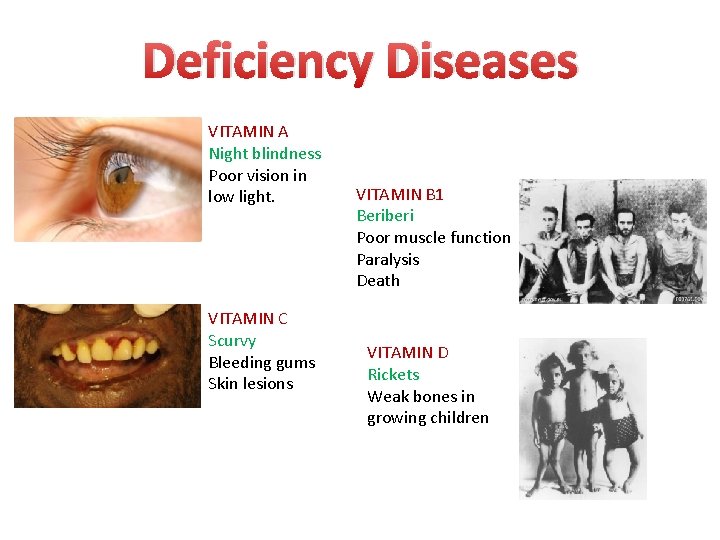
Deficiency Diseases VITAMIN A Night blindness Poor vision in low light. VITAMIN C Scurvy Bleeding gums Skin lesions VITAMIN B 1 Beriberi Poor muscle function Paralysis Death VITAMIN D Rickets Weak bones in growing children

Gowland Hopkins Experiment Group B Group A 2 groups of young rats group A were fed on a diet of purified casein, starch, glucose, lard, minerals and water Group B were fed the same diet but with the addition of milk On day 18 the diets were switched. without milk 1. 2. 3. 4. With milk For the first 18 days, which was the test group and which was the control? Why were several rats used in each group? Why do you think he switched the diets after 18 days? What conclusions do you think could be drawn from this experiment? Hopkins' experiments transformed nutrition into a clearly recognized branch of biochemistry founded upon scientific principles. The ability to synthesize products in the laboratory that were identical in properties and physiological effect with the natural vitamins spawned a new industry of nutritional supplements.

Vitamin C pre-lab DCPIP is used to detect the presence of vitamin C. It is dark blue but changes colour in the presence of vitamin C. We are going to make an estimate of the amount of vitamin C in a fruit juice by comparing its ability to change the colour of DCPIP compared to a known concentration of ascorbic acid (Vitamin C) 1) Measure out 1 cm 3 of DCPIP into a pipette using a pipette filler 2) Transfer the DCPIP into a test tube 3) Add the 10 mg/100 cm 3 ascorbic acid to the DCPIP drop by drop using a dropper. Carefully count the number of drops that you use. 4) Repeat the procedure using the fruit juice. 5) If time allows, carry out repeated trials 6) Record your results in a suitably designed table and calculate the mean number of drops. The amount of vitamin C in the fruit juice _____ = Number of drops of ascorbic acid Number of drops of fruit juice X 10 mg/100 cm 3
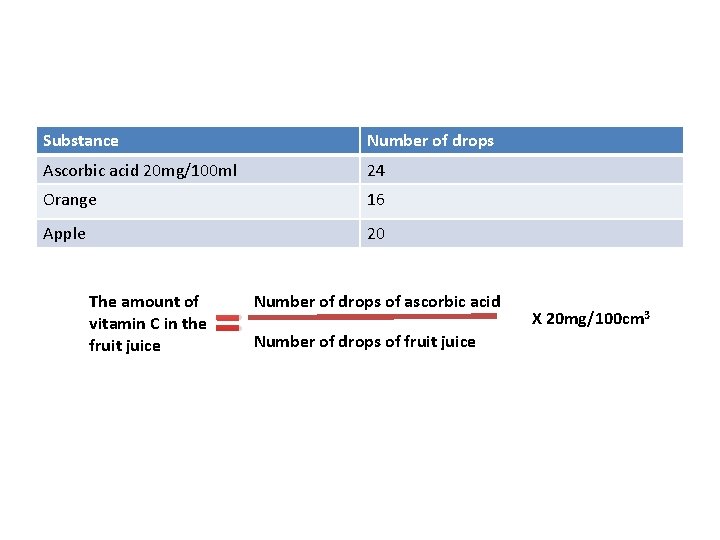
Substance Number of drops Ascorbic acid 20 mg/100 ml 24 Orange 16 Apple 20 The amount of vitamin C in the fruit juice = Number of drops of ascorbic acid Number of drops of fruit juice X 20 mg/100 cm 3

Which colour bell peppers contains the most vitamin C? Mr Hennie has been suffering from a cold. He is aware that Vitamin C helps to fight off colds and that bell peppers contain large amounts of vitamin C. He wants to include some freshly sliced bell peppers with his salad for lunch, but which colour should he buy? A slurry can be made by grinding a piece of pepper with water using a mortar and pestle. The slurry can be tested for vitamin C using DCPIP. The more drops required, the less vitamin C there is. Think how the test is to be standardised to ensure a fair test. How do you obtain results that are reliable and you can trust? Can you determine the amount of vitamin C? How can you present the data in a graph?

The Digestive System Draw the diagram in the left half of an A 4 piece of paper in landscape. The mouth area can be simplified. Do not copy the writing below. The liver produces bile which helps in fat digestion. Therefore it is usually included as part of the digestive system The pancreas produces digestive enzymes. Duodenum + Ileum = Small intestine Colon = Large intestine

11 2 7 9 3 1 5 10 8 12 4 6 Write out the information to the left in the correct sequence, approximately in line with the relevant structures on your drawing of the digestive system.

Digestion

Learning objectives 1. Explain why food needs to be digested 2. State the role of enzymes in digestion 3. Identify the types of enzymes that digest food 4. Explain the importance of villi and microvilli in crating a large surface area in the intestines
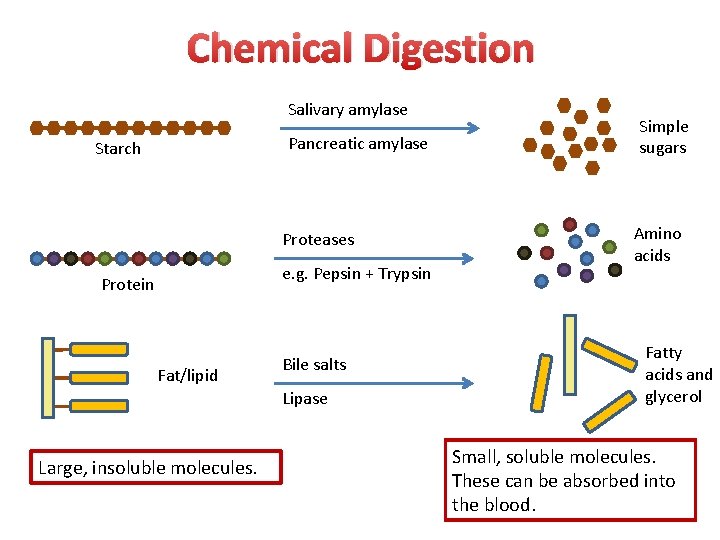
Chemical Digestion Salivary amylase Pancreatic amylase Starch Proteases e. g. Pepsin + Trypsin Protein Fat/lipid Bile salts Lipase Large, insoluble molecules. Simple sugars Amino acids Fatty acids and glycerol Small, soluble molecules. These can be absorbed into the blood.
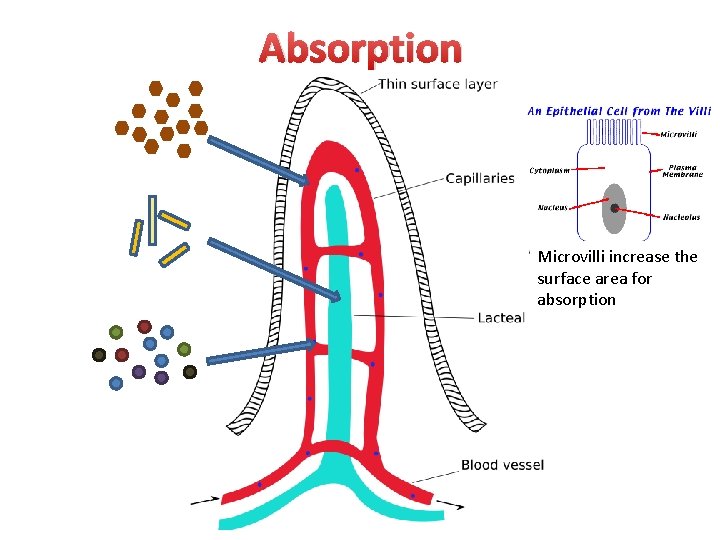
Absorption Microvilli increase the surface area for absorption
Summary Food substance Enzyme End product Where transported?
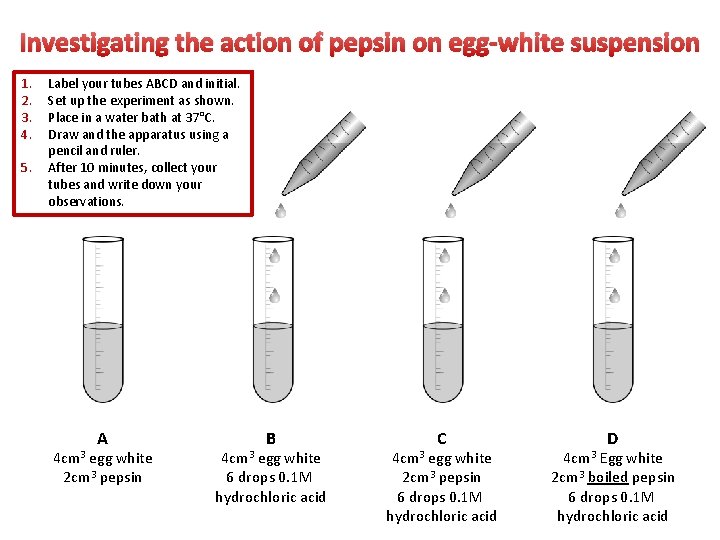
Investigating the action of pepsin on egg-white suspension 1. 2. 3. 4. 5. Label your tubes ABCD and initial. Set up the experiment as shown. Place in a water bath at 37°C. Draw and the apparatus using a pencil and ruler. After 10 minutes, collect your tubes and write down your observations. A 4 cm 3 egg white 2 cm 3 pepsin B 4 cm 3 egg white 6 drops 0. 1 M hydrochloric acid C 4 cm 3 egg white 2 cm 3 pepsin 6 drops 0. 1 M hydrochloric acid D 4 cm 3 Egg white 2 cm 3 boiled pepsin 6 drops 0. 1 M hydrochloric acid

Year 8 Digestion of Starch Practical
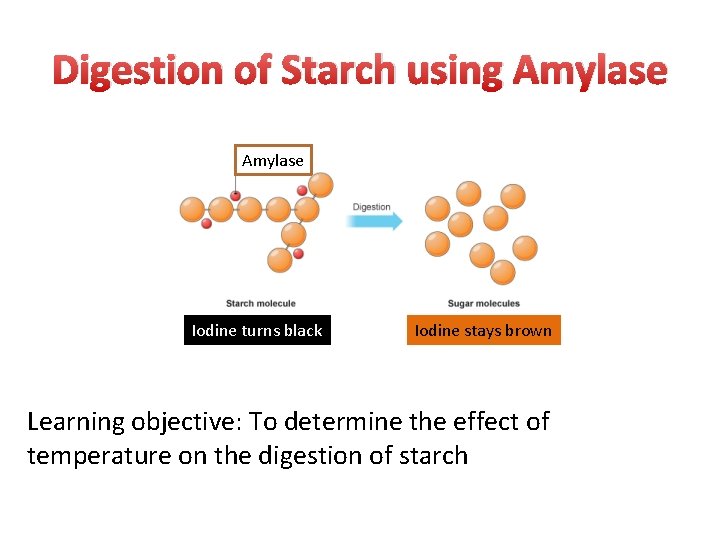
Digestion of Starch using Amylase Iodine turns black Iodine stays brown Learning objective: To determine the effect of temperature on the digestion of starch
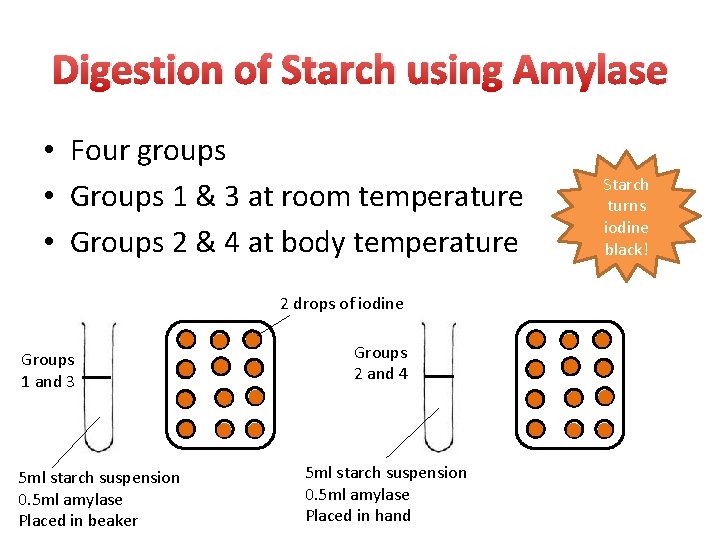
Digestion of Starch using Amylase • Four groups • Groups 1 & 3 at room temperature • Groups 2 & 4 at body temperature 2 drops of iodine Groups 1 and 3 5 ml starch suspension 0. 5 ml amylase Placed in beaker Groups 2 and 4 5 ml starch suspension 0. 5 ml amylase Placed in hand Starch turns iodine black!
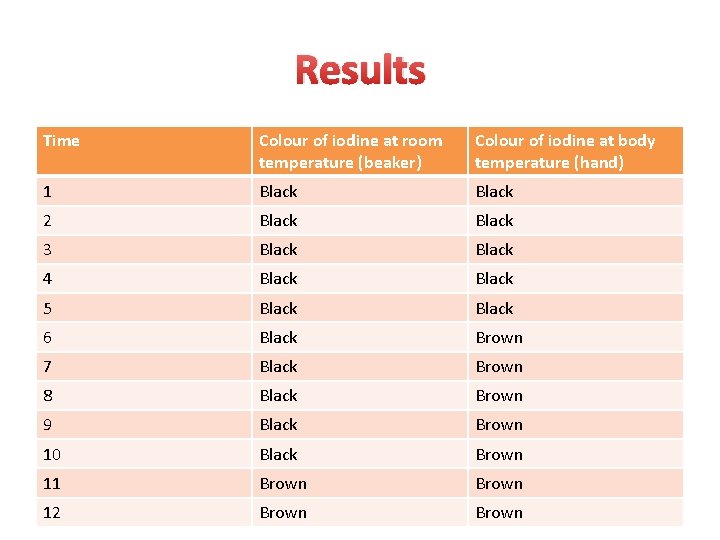
Results Time Colour of iodine at room temperature (beaker) Colour of iodine at body temperature (hand) 1 Black 2 Black 3 Black 4 Black 5 Black 6 Black Brown 7 Black Brown 8 Black Brown 9 Black Brown 10 Black Brown 11 Brown 12 Brown
- Slides: 28