Redox Titrations Introduction 1 Redox Titration Based on
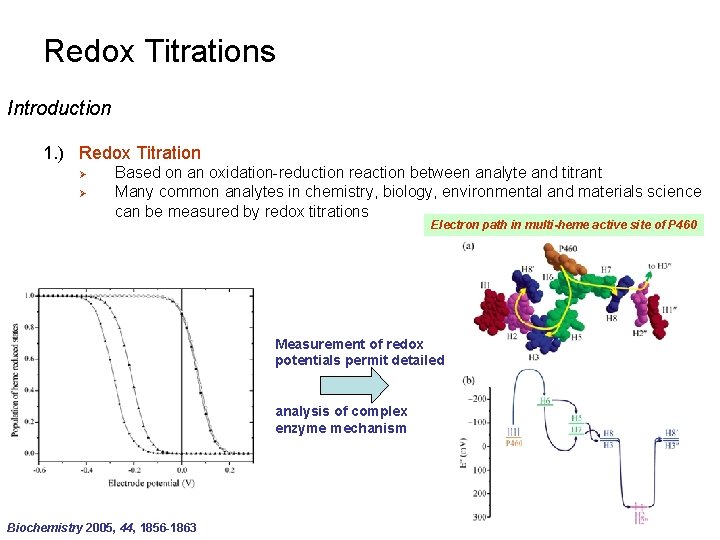
Redox Titrations Introduction 1. ) Redox Titration Ø Ø Based on an oxidation-reduction reaction between analyte and titrant Many common analytes in chemistry, biology, environmental and materials science can be measured by redox titrations Electron path in multi-heme active site of P 460 Measurement of redox potentials permit detailed analysis of complex enzyme mechanism Biochemistry 2005, 44, 1856 -1863

Redox Titrations Shape of a Redox Titration Curve 1. ) Voltage Change as a Function of Added Titrant Ø Consider the Titration Reaction (essentially goes to completion): K ≈ 1016 Ø Ø Ø Ce 4+ is added with a buret to a solution of Fe 2+ Pt electrode responds to relative concentration of Fe 3+/Fe 2+ & Ce 4+/Ce 3+ Calomel electrode used as reference Indicator half-reactions at Pt electrode: Eo = 0. 767 V Eo = 1. 70 V
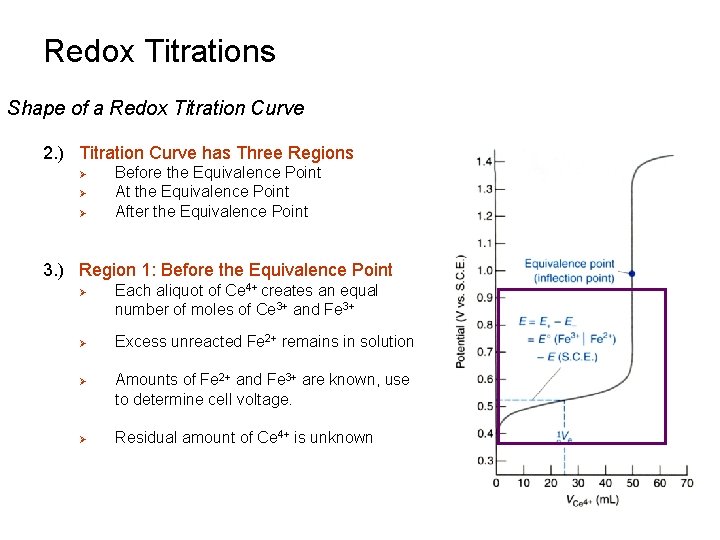
Redox Titrations Shape of a Redox Titration Curve 2. ) Titration Curve has Three Regions Ø Ø Ø Before the Equivalence Point At the Equivalence Point After the Equivalence Point 3. ) Region 1: Before the Equivalence Point Ø Ø Each aliquot of Ce 4+ creates an equal number of moles of Ce 3+ and Fe 3+ Excess unreacted Fe 2+ remains in solution Amounts of Fe 2+ and Fe 3+ are known, use to determine cell voltage. Residual amount of Ce 4+ is unknown
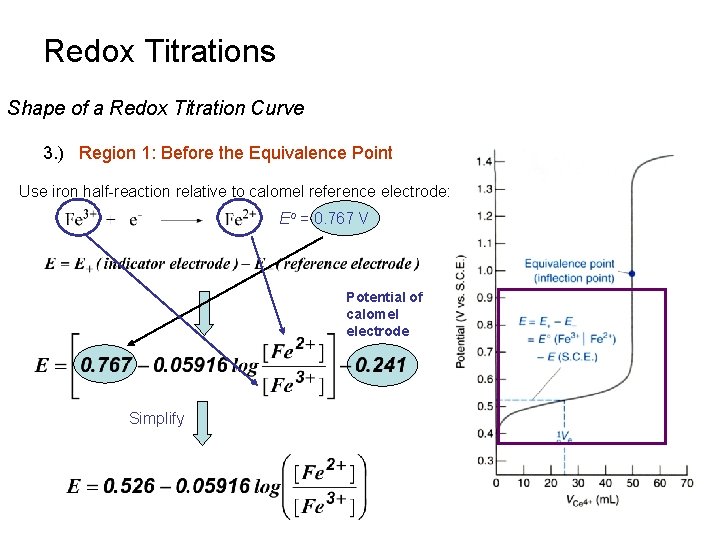
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Use iron half-reaction relative to calomel reference electrode: Eo = 0. 767 V Potential of calomel electrode Simplify
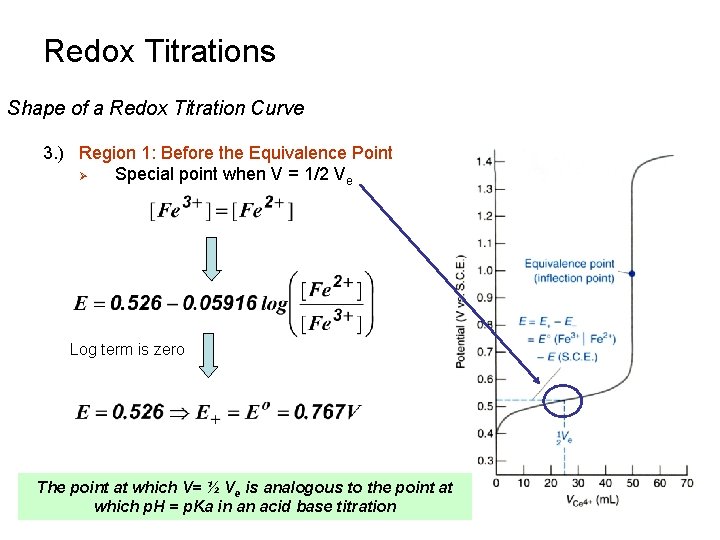
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Ø Special point when V = 1/2 Ve Log term is zero The point at which V= ½ Ve is analogous to the point at which p. H = p. Ka in an acid base titration
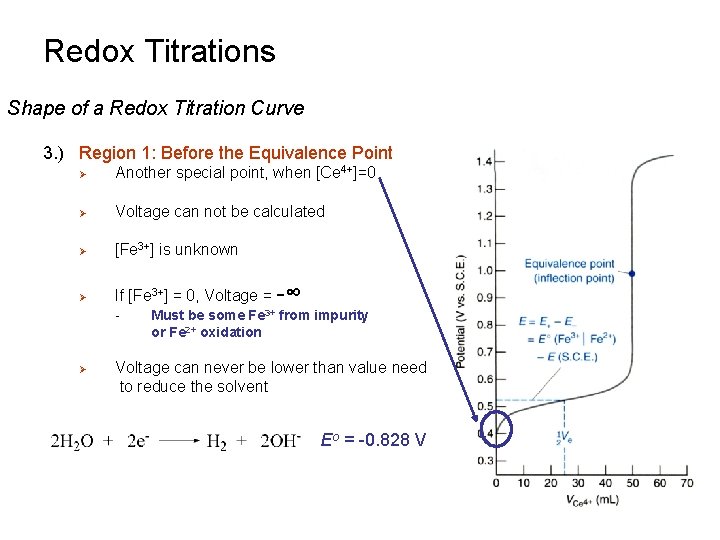
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Ø Another special point, when [Ce 4+]=0 Ø Voltage can not be calculated Ø [Fe 3+] is unknown Ø If [Fe 3+] = 0, Voltage = -∞ - Ø Must be some Fe 3+ from impurity or Fe 2+ oxidation Voltage can never be lower than value need to reduce the solvent Eo = -0. 828 V
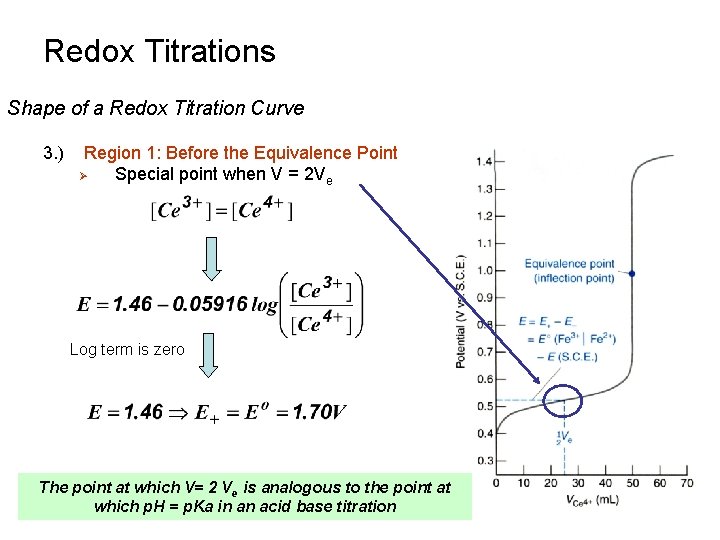
Redox Titrations Shape of a Redox Titration Curve 3. ) Region 1: Before the Equivalence Point Ø Special point when V = 2 Ve Log term is zero The point at which V= 2 Ve is analogous to the point at which p. H = p. Ka in an acid base titration
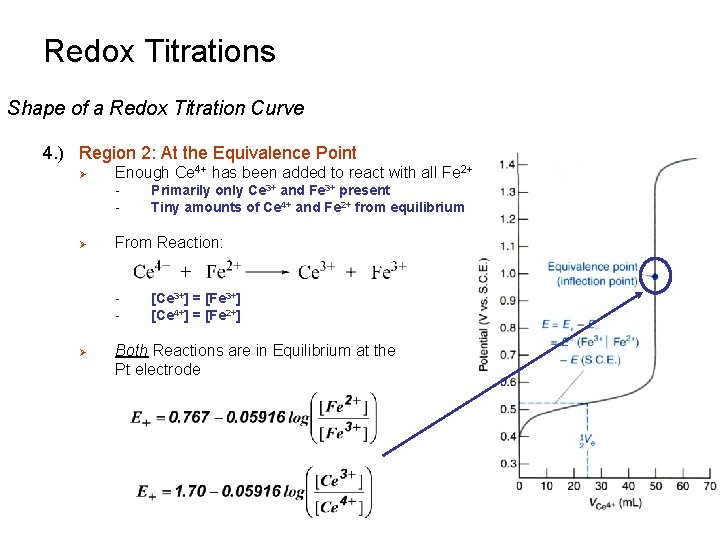
Redox Titrations Shape of a Redox Titration Curve 4. ) Region 2: At the Equivalence Point Ø Enough Ce 4+ has been added to react with all Fe 2+ - Ø From Reaction: - Ø Primarily only Ce 3+ and Fe 3+ present Tiny amounts of Ce 4+ and Fe 2+ from equilibrium [Ce 3+] = [Fe 3+] [Ce 4+] = [Fe 2+] Both Reactions are in Equilibrium at the Pt electrode

Redox Titrations Shape of a Redox Titration Curve 4. ) Region 2: At the Equivalence Point Ø Ø Ø Don’t Know the Concentration of either Fe 2+ or Ce 4+ Can’t solve either equation independently to determine E+ Instead Add both equations together Add Rearrange
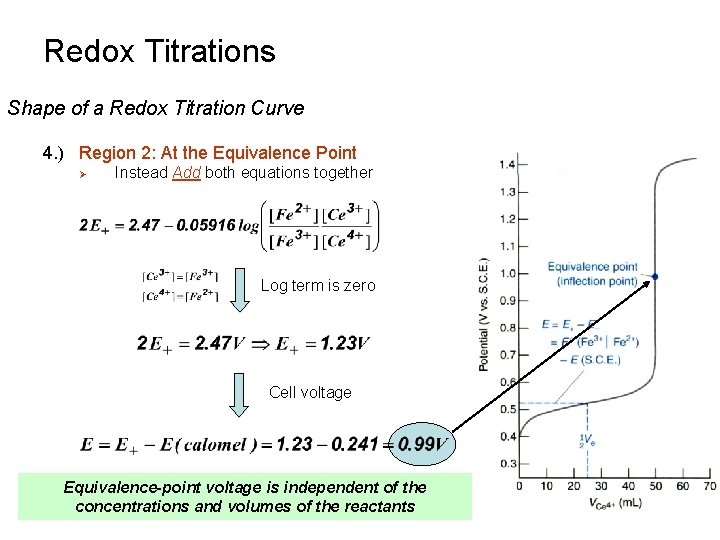
Redox Titrations Shape of a Redox Titration Curve 4. ) Region 2: At the Equivalence Point Ø Instead Add both equations together Log term is zero Cell voltage Equivalence-point voltage is independent of the concentrations and volumes of the reactants
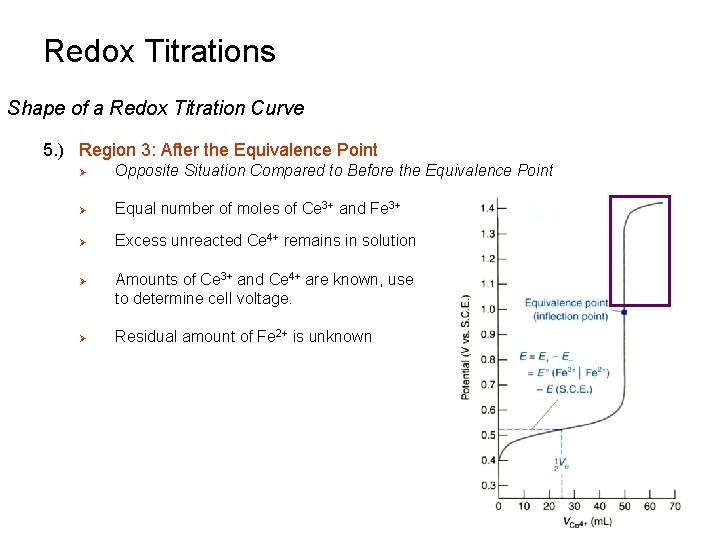
Redox Titrations Shape of a Redox Titration Curve 5. ) Region 3: After the Equivalence Point Ø Opposite Situation Compared to Before the Equivalence Point Ø Equal number of moles of Ce 3+ and Fe 3+ Ø Excess unreacted Ce 4+ remains in solution Ø Ø Amounts of Ce 3+ and Ce 4+ are known, use to determine cell voltage. Residual amount of Fe 2+ is unknown
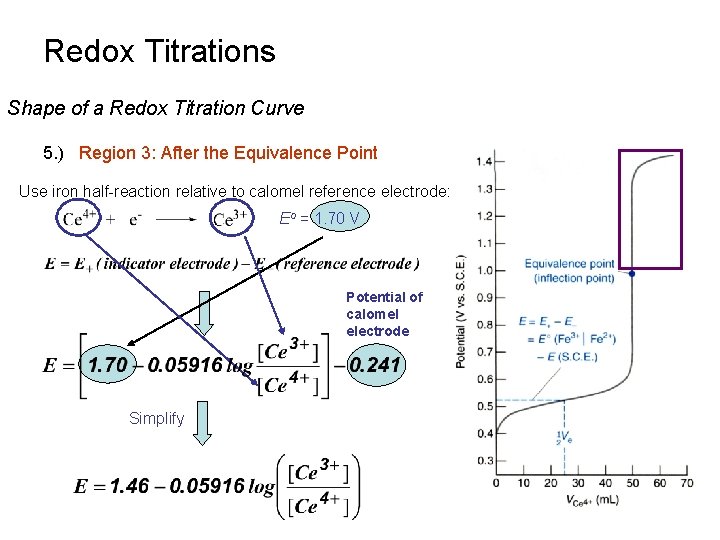
Redox Titrations Shape of a Redox Titration Curve 5. ) Region 3: After the Equivalence Point Use iron half-reaction relative to calomel reference electrode: Eo = 1. 70 V Potential of calomel electrode Simplify
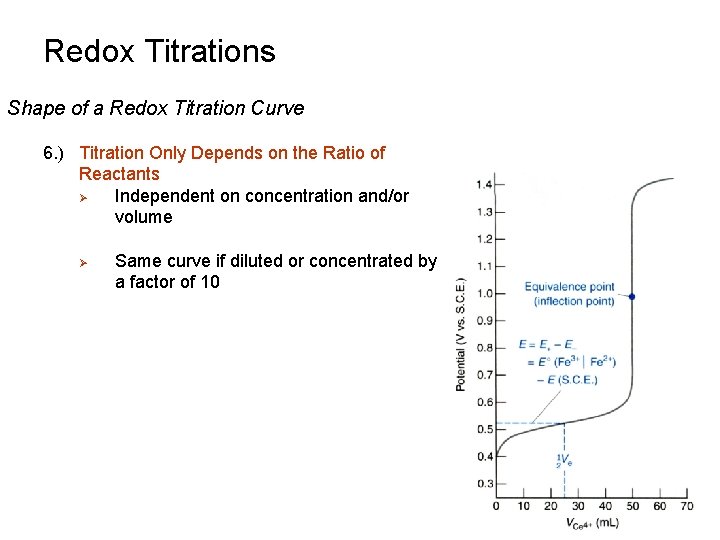
Redox Titrations Shape of a Redox Titration Curve 6. ) Titration Only Depends on the Ratio of Reactants Ø Independent on concentration and/or volume Ø Same curve if diluted or concentrated by a factor of 10
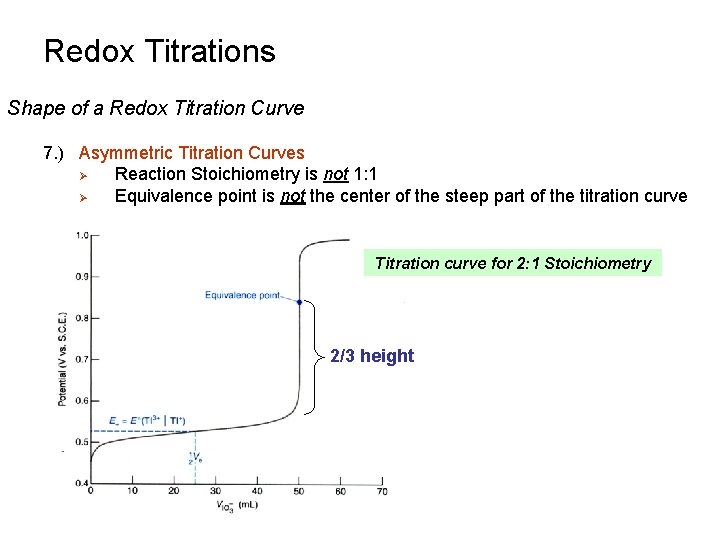
Redox Titrations Shape of a Redox Titration Curve 7. ) Asymmetric Titration Curves Ø Reaction Stoichiometry is not 1: 1 Ø Equivalence point is not the center of the steep part of the titration curve Titration curve for 2: 1 Stoichiometry 2/3 height

Redox Titrations Finding the End Point 1. ) Indicators or Electrodes Ø Ø Ø Similar to Acid-Base Titrations Electrochemical measurements (current or potential) can be used to determine the endpoint of a redox titration Redox Indicator is a chemical compound that undergoes a color change as it goes from its oxidized form to its reduced form - Similar to acid-base indicators that change color with a change in protonation state

Redox Titrations Finding the End Point 2. ) Redox Indicators Ø Color Change for a Redox Indicator occurs mostly over the range: where Eo is the standard reduction potential for the indicator and n is the number of electrons involved in the reduction

Redox Titrations Finding the End Point 2. ) Redox Indicators Ø Ø Color Change for a Redox Indicator occurs over a potential range Illustration: For Ferroin with Eo = 1. 147 V, the range of color change relative to SHE: Relative to SCE is:
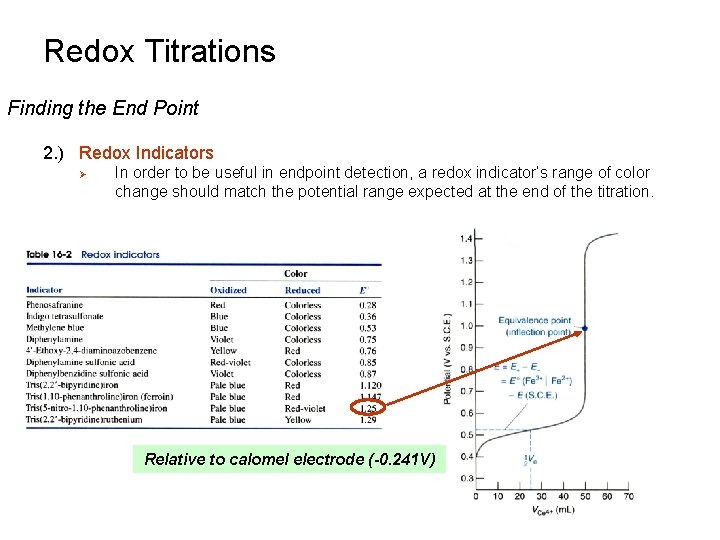
Redox Titrations Finding the End Point 2. ) Redox Indicators Ø In order to be useful in endpoint detection, a redox indicator’s range of color change should match the potential range expected at the end of the titration. Relative to calomel electrode (-0. 241 V)

Redox Titrations Common Redox Reagents 1. ) Starch Ø Commonly used as an indicator in redox titrations involving iodine Ø Reacts with iodine to form an intensely blue colored complex Ø Starch is not a redox indicator - Does not undergo a change in redox potential I 6 bound in center of starch helix Repeating unit

Redox Titrations Common Redox Reagents 2. ) Adjustment of Analyte Oxidation State Ø Before many compounds can be determined by Redox Titrations, must be converted into a known oxidation state - Ø Reagents for prereduction or preoxidation must: - Ø This step in the procedure is known as prereduction or preoxidation Totally convert analyte into desired form Be easy to remove from the reaction mixture Avoid interfering in the titration Examples: - Preoxidation: a) Peroxydisulfate or persulfate (S 2 O 82 -) with Ag+ catalyst Powerful oxidants Oxidizes Mn 2+, Ce 3+, Cr 3+, VO 2+ excess S 2 O 82 - and Ag+ removed by boiling the solution

Redox Titrations Common Redox Reagents 2. ) Adjustment of Analyte Oxidation State Ø Examples: - Preoxidation: b) Silver(II) oxide (Ag. O) in concentrated mineral acids also yields Ag 2+ excess removed by boiling c) - Hydrogen peroxide (H 2 O 2) is a good oxidant to use in basic solutions Oxidizes Co 2+, Fe 2+, Mn 2+ Reduces Cr 2 O 72 -, Mn. O 4 excess removed by boiling Prereduction: a) Stannous chloride (Sn. Cl 2) in hot HCl Reduce Fe 3+ to Fe 2+ excess removed by adding Hg. Cl 2 b) Jones reductor (Zn + Zn amalgam – anything in mercury)

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Potassium Permanganate (KMn. O 4) - Strong oxidant Own indicator Titration of VO 2+ with KMn. O 4 p. H ≤ 1 Eo = 1. 507 V Violet colorless p. H neutral or alkaline Eo = 1. 692 V Violet Before Near After Equivalence point brown p. H strolngly alkaline Eo = 0. 56 V Violet green

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Potassium Permanganate (KMn. O 4) - Application of KMn. O 4 in Redox Titrations

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Cerium (IV) (Ce 4+) - Commonly used in place of KMn. O 4 Works best in acidic solution Can be used in most applications in previous table Used to analyze some organic compounds Color change not distinct to be its own indicator Yellow colorless Ce 4+ binds anions very strongly results in variation of formal potential Formal potential 1. 70 V in 1 F HCl. O 4 1. 61 V in 1 F HNO 3 1. 47 V in 1 F HCl 1. 44 V in 1 F H 2 SO 4 Measure activity not concentration

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Potassium Dichromate (K 2 Cr 2 O 7) - Powerful oxidant in strong acid Not as Strong as KMn. O 4 or Ce 4+ Primarily used for the determination of Fe 2+ Not an oxidant in basic solution Color change not distinct to be its own indicator Eo = 1. 36 V orange green to violet

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Iodine (Solution of I 2 + I-) - I 3 - is actual species used in titrations with iodine K = 7 x 102 - Either starch of Sodium Thiosulfate (Na 2 S 2 O 3) are used as indicator I 3 - + S 2 O 32 - I 3 - + Starch Before At endpoint

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Iodine (Solution of I 2 + I-) - Application of Iodine in Redox Titrations

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Iodine (Solution of I 2 + I-) - Application for Redox Titrations that Produce I 3 -

Redox Titrations Common Redox Reagents 3. ) Common Titrants for Oxidation Reactions Ø Periodic Acid (HIO 4) - Commonly used in titration of organic compounds (especially carbohydrates) 4. ) Titrations with Reducing Agents Ø Not as common as titrations using oxidizing agents - Ø Available titrants are not very stable in the presence of atmospheric O 2 Reagents can be generated directly in solution by means of chemical or electrochemical reactions

Redox Titrations Common Redox Reagents 5. ) Example A 50. 00 m. L sample containing La 3+ was titrated with sodium oxalate to precipitate La 2(C 2 O 4)3, which washed, dissolved in acid, and titrated with 18. 0 m. L of 0. 006363 M KMn. O 4. Calculate the molarity of La 3+ in the unknown.
- Slides: 30