Immunoglobulin Therapy in Primary Immunodeficiencies By Angela Migowa

Immunoglobulin Therapy in Primary Immunodeficiencies By Angela Migowa Pediatric Rheumatologist Aga Khan University EPLS Instructor (RCUK) MBCHB (UON), MMED Pediatrics and Child Health (AKUHN), Pediatric Rheumatology (Mc. Gill)


Objectives • To understand the mechanism of action of Immunoglobulin Therapy in management of Primary Immunodeficiencies • To describe the indications of Immunoglobulin Therapy in primary immunodeficiencies • To outline the formulations and dosing of immunoglobulins

Outline • Historical perspective of Immunoglobulin Therapy • The status of Immunoglobulin Therapy in Africa and Kenya • Mode of action of Immunoglobulin Therapy • Indications of Immunoglobulin Therapy in PID’s • Dosing, Formulation and Toxicity of Immunoglobulin Therapy • Questions • Summary
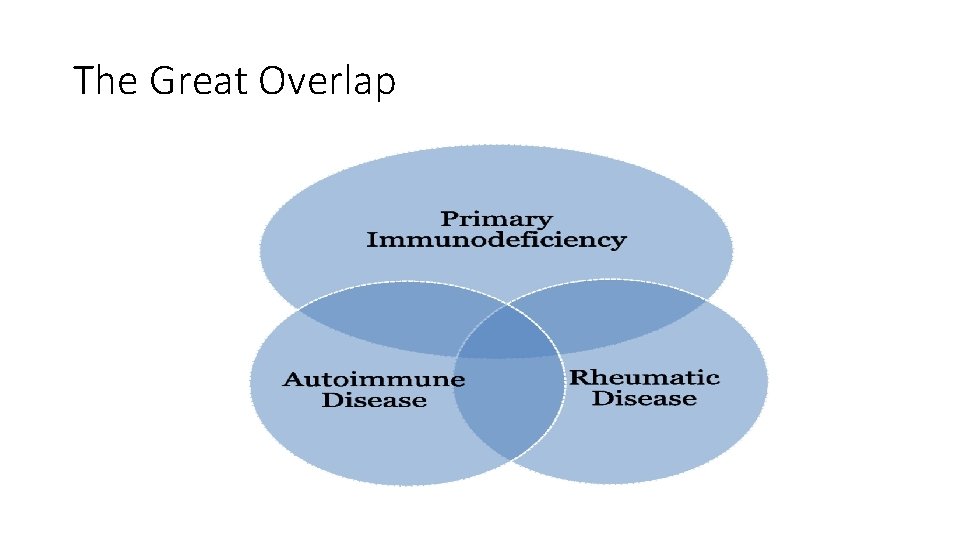
The Great Overlap

Definitions • Therapeutic immunoglobulin is a blood product derived from thousands of healthy, pooled donors and preparations are made up of almost exclusively Ig. G (although commercially available products also contain trace amounts of Ig. A and Ig. M). Schwab I et al. Nat Rev Immunol. 2013; 13(3): 176– 189. Sandomenico A et al. Mol Biotechnol. 2013; 54 (3): 983– 995.

Historical Perspective • Serum from animals or convalescent humans for treatment and prevention by end of the 19 century. • Purification of Immunoglibulin G (Ig. G) in the 1940’s • Human Ig subcut replacement therapy first performed in 1952, by Bruton, in patient with agammaglobulinemia • Since the 1960’s, different intravenous preparations • Subcutaneous administration increasingly used in the last 10 to 15 years Berger M et al. United States: Academic Press; 2014. pp. 307– 377. Wasserman RL et al. Allergy Clin Immunol. 2015; 135(2 Suppl)

Raising Awareness to improve the current status of PID on the Continent: The A Project • 56 African Countries • Gross undiagnosis and misdiagnosis of PIDs on the continent • Caregivers cannot easily access congresses/resources • Patients have no voice • Urgent need to improve lives of African PID patients • Urgent need to document the African genetic PID landscape Courtesy Prof J. Chipeta
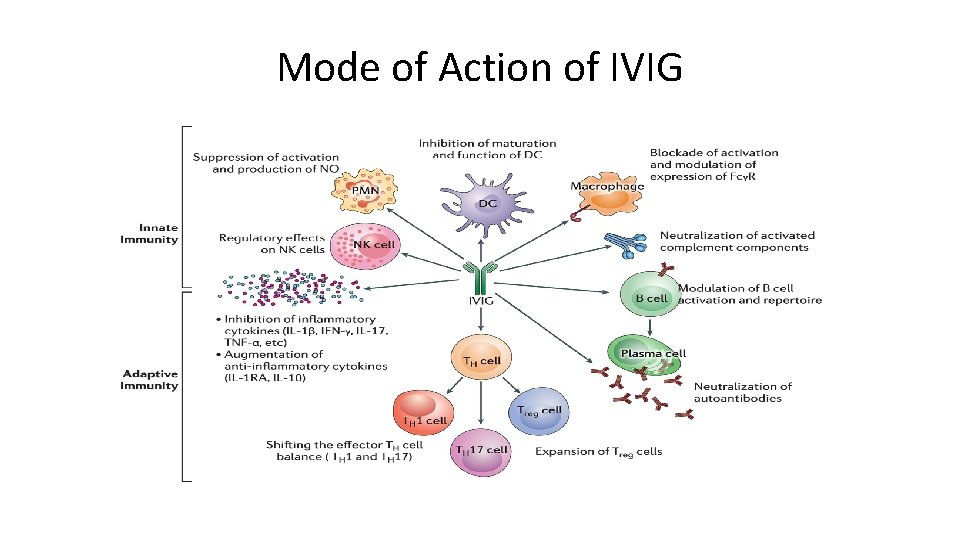
Mode of Action of IVIG


Indications of Therapy

Goal of Therapy • The goal of therapy is to provide levels of functional serum Ig. G expected in normal subjects, and sufficient amounts of passive antibodies capable of neutralization and opsonization of broad categories of infectious pathogens. Albin S et al. Immunotherapy. 2014; 6(10): 1113– 1126.

Pre-requisites to IVIG Administration • Improved education among healthcare workers • Appropriate patient workup before initiation of therapy. • Baseline immunoglobulin levels • Specific antibodies to prior vaccinations • Screening for potential blood borne infections eg HIV, Hepatitis

Administration • Currently two routes • Intravenous (IVIG), first approved for use in 1979 • subcutaneous (SCIG), first approved for use in 2006 [80]. • Biochemical properties should be considered in the context of unique patient profiles. • Starting in doses between 400 and 600 mg/kg every 3– 4 weeks intravenously or twice weekly subcutaneously Immune Deficiency Foundation. Immunoglobulin products. www. primaryimmune. org/treatment-information/immunoglobulin-products/

Indications

X-Linked Agammaglobulinemia • Caused by a mutation in the gene for Bruton’s tyrosine kinase, which leads to a marked reduction in B cells and agammaglobulinemia. • Present with recurrent bacterial infections such as otitis, sinusitis and pneumonia, with physical exam findings of absent or barely detectable tonsillar lymph node tissue. • Immunoglobulin replacement proven successful in preventing infections and allowing patients to lead healthy and productive lives. Debost-Legrand A et al. Gene. 2013; 527(1): 426– 428.
Common Variable Immunodeficiency • Hypogammaglobulinemia with a variety of T cell abnormalities • Clinical manifestations include infections, autoimmunity and cancer. • Infections from encapsulated, atypical bacteria and parasites eg Giardia • Immunoglobulin replacement may be used in conjunction with prophylactic antibiotics. • Immunoglobulin does not primarily treat the noninfectious manifestations of CVID Cunningham-Rundles C et al. Blood. 2010; 116(1): 7– 15.
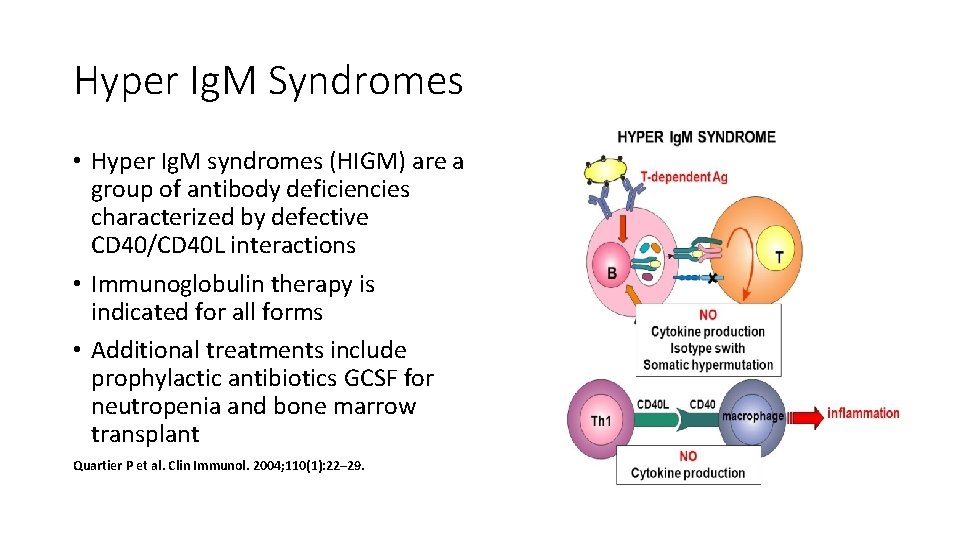
Hyper Ig. M Syndromes • Hyper Ig. M syndromes (HIGM) are a group of antibody deficiencies characterized by defective CD 40/CD 40 L interactions • Immunoglobulin therapy is indicated for all forms • Additional treatments include prophylactic antibiotics GCSF for neutropenia and bone marrow transplant Quartier P et al. Clin Immunol. 2004; 110(1): 22– 29.

Wiskott Aldrich Syndrome • Wiskott-Aldrich syndrome is a triad of recurrent infections, microthrombocytopenia and eczema • Patients also are predisposed to autoimmune disorders and malignancies • It is an X-linked disease caused by mutations in the WASP gene. WASP protein is involved in the transduction of signals from receptors • Bone marrow transplant is the only cure • Supportive treatment includes immunoglobulin therapy, antimicrobials, platelet transfusions Massaad MJ et al. Ann NY Acad Sci. 2013; 1285: 26– 43.
Severe Combined Immunodeficiency • Severe combined immunodeficiency (SCID) is a heterogeneous group of genetic disorders characterized by significant T-cell deficiency, with variable production and function of B and NK cells [67]. • Infants born are normal at birth, but infection and failure to thrive manifest as passively transferred maternal antibodies begin to wane • Considered a pediatric emergency • While awaiting transplant, patients are given prophylactic antibiotics and immunoglobulin replacement. Bousifa A et al. Front Immunol. 2011; 2: 54. Dvorak CC et al. J Clin Immunol. 2013; 33(7): 1156– 1164. The Bubble Boy

When to be cautious with IVIG therapy

Selective Ig A Deficiency • Asymptomatic patients with selective Ig. A deficiency do not need immunoglobulin treatment • Recurrent infections should be treated as needed • Selective Ig. A deficiency is not an indication for immunoglobulin, and administration may lead to anaphylaxis or similarly severe reactions for Ig. A-deficient patients who have Ig. G or Ig. E anti-Ig. A antibodies Burks AW et al. N Engl J Med. 1986; 314(9): 560– 564.

Toxicity

Adverse Effects Symptoms and signs Frequency Related to infusion rate Chills Frequent Headache Frequent Dyspnea Frequent Chest pain or tightness Frequent Back pain Frequent Fatigue and malaise Frequent Fever Frequent Hypotension or hypertension Frequent Myalgia Frequent Nausea and vomiting Frequent Pruritus Frequent Skin rash and hives Frequent Flu-like symptoms Frequent Tachycardia Frequent https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 5823061

Adverse Effects Symptoms and signs Frequency Tachycardia Frequent Central nervous system Aseptic meningitis Rare Severe headache Rare Renal Acute renal failure (acute tubular necrosis) Azotemia Rare (usually associated with sucrose as a stabilizer) Rare Thromboembolic events Thrombosis and cerebral infarction Rare Myocardial infarction Rare Pulmonary thromboembolism Rare Posterior leukoencephalopathy syndrome Rare https: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 5823061/

Imperatives for the Primary Care Provider • Accurate record of immunizations • Maintain accurate and ongoing growth charts • Complete family history with genogram • Follow-up after antibiotic treatment (i. e. did what you prescribe work? )

Questions?
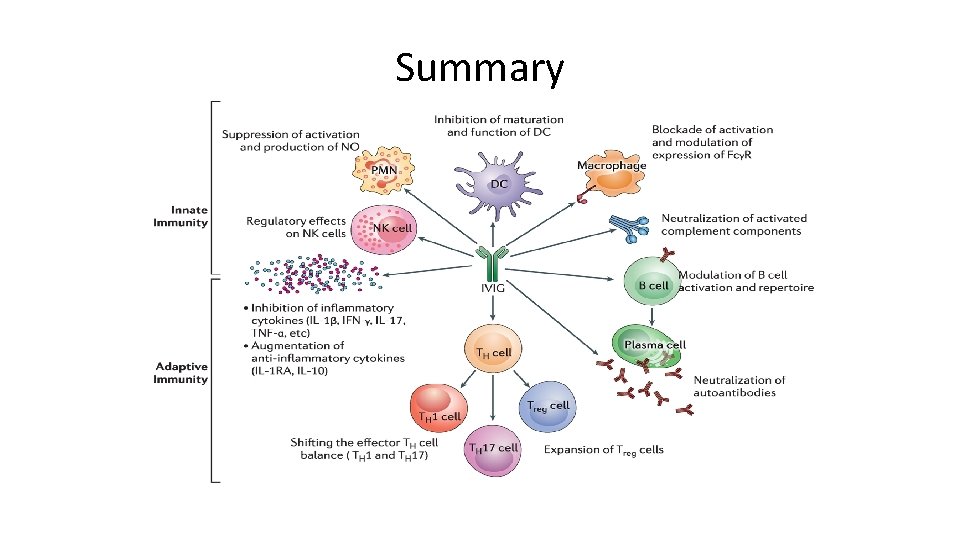
Summary


Thank you
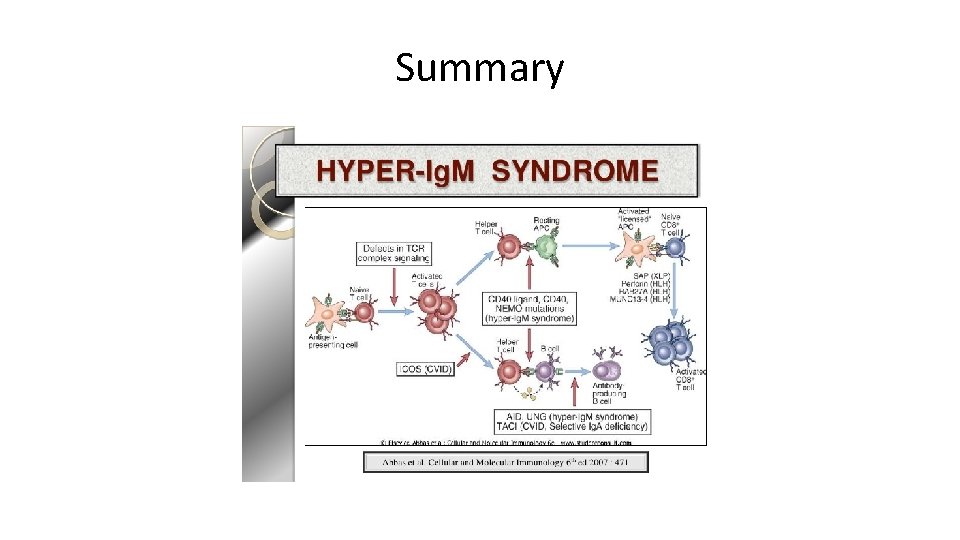
Summary
- Slides: 32