Starch Glycogen and Cellulose Starch Structure Starch is







- Slides: 12

Starch, Glycogen and Cellulose

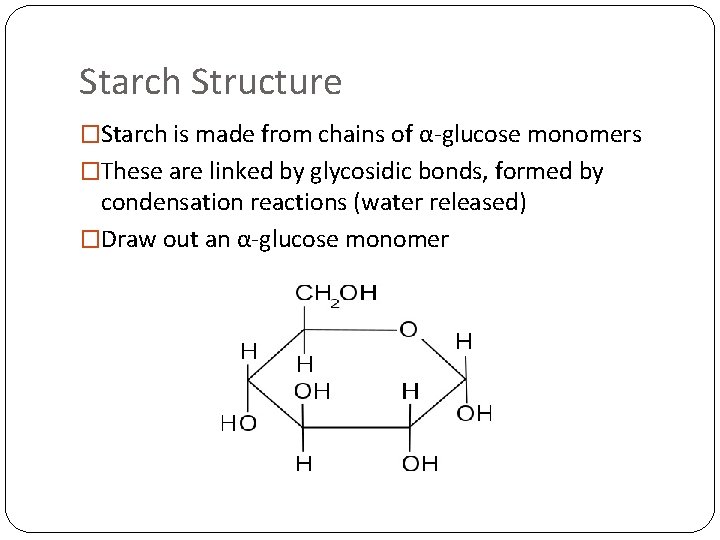
Starch Structure �Starch is made from chains of α-glucose monomers �These are linked by glycosidic bonds, formed by condensation reactions (water released) �Draw out an α-glucose monomer

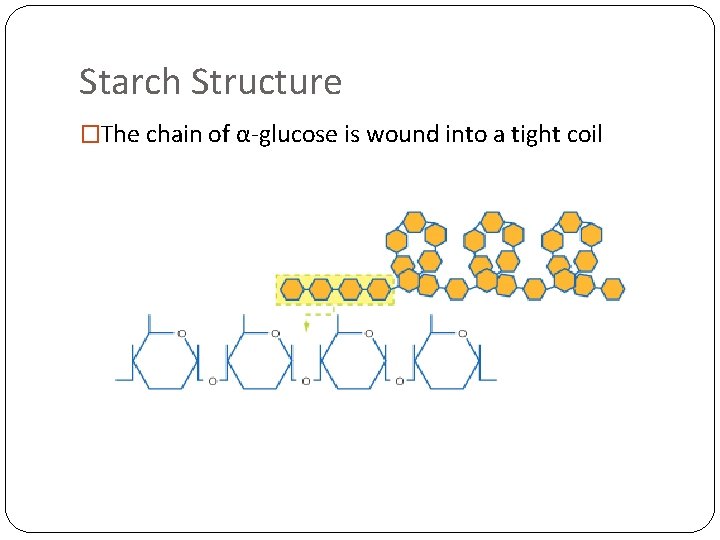
Starch Structure �The chain of α-glucose is wound into a tight coil

Starch �Where is starch found in a plant? �Many parts as starch grains �Seeds �Storage organs e. g. Potato tubers

Why is starch a good molecule for storage in plants? �It is insoluble, so doesn’t draw water into cells by osmosis �Won’t easily diffuse out of cells because it is insoluble �It can be stored in a small space because the tight coils make it compact �Can be easily hydrolysed to give α-glucose, which can be used in respiration

Glycogen �Starch is not found in animal cells, glycogen is used instead �It is similar to starch, but has shorter chains and is more highly branched �Storage molecule for animals, found as small granules in the muscles and the liver �Because of the shorter chains it is even more readily hydrolysed than starch

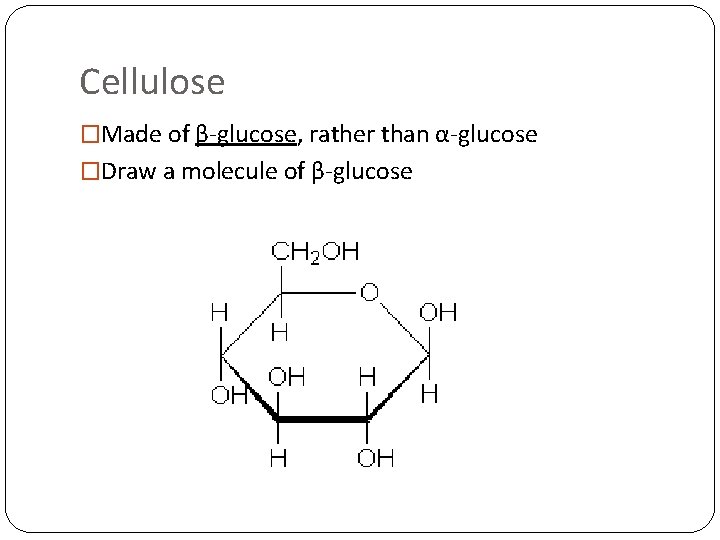
Cellulose �Made of β-glucose, rather than α-glucose �Draw a molecule of β-glucose

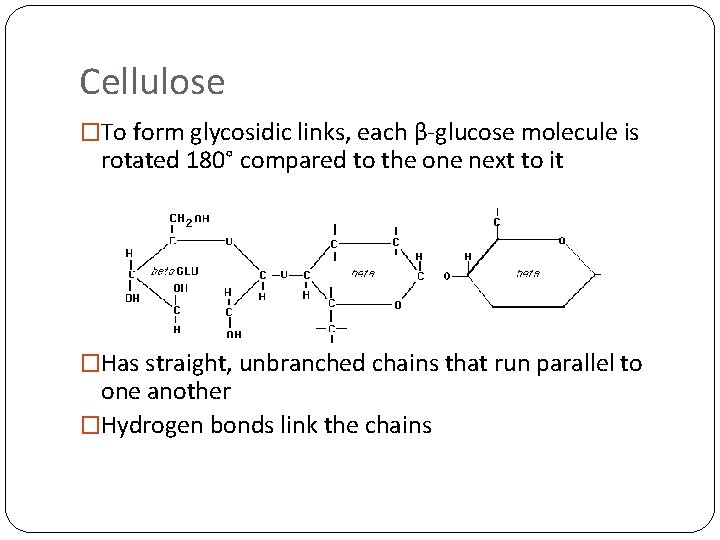
Cellulose �To form glycosidic links, each β-glucose molecule is rotated 180° compared to the one next to it �Has straight, unbranched chains that run parallel to one another �Hydrogen bonds link the chains

Cellulose �So many hydrogen bonds help to strengthen cellulose �This makes cellulose a good structural material, hence its use in plant cell walls to aid rigidity �Cellulose does this by grouping together to form microfibrils fibres �Cellulose also prevents cells bursting, so they are turgid when full with water. This helps support stems

The main structural difference between starch glycogen cellulose comes from? Starch, glycogen and cellulose are all polymers of glucose. They differ in the type of glucose present and the bonds which link the glucose monomers together. Starch and glycogen are made from alphaglucose. This is an isomer of glucose in which the hydroxyl (-OH) group attached to carbon number 1 is below the plane of the ring.

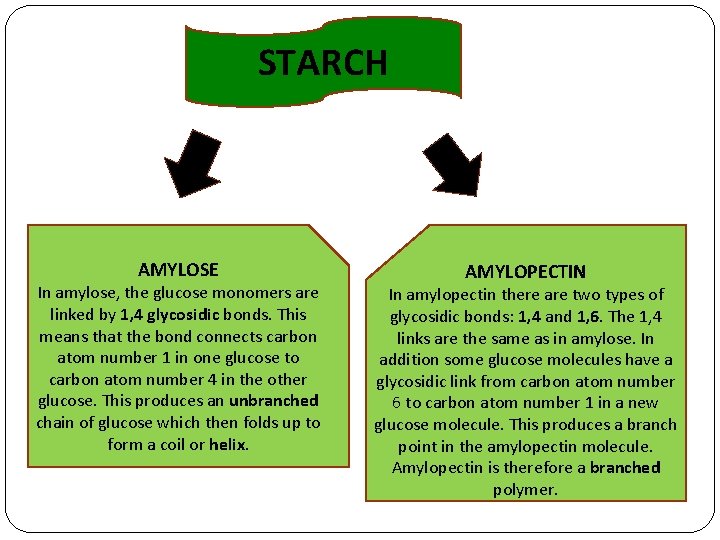
STARCH AMYLOSE In amylose, the glucose monomers are linked by 1, 4 glycosidic bonds. This means that the bond connects carbon atom number 1 in one glucose to carbon atom number 4 in the other glucose. This produces an unbranched chain of glucose which then folds up to form a coil or helix. AMYLOPECTIN In amylopectin there are two types of glycosidic bonds: 1, 4 and 1, 6. The 1, 4 links are the same as in amylose. In addition some glucose molecules have a glycosidic link from carbon atom number 6 to carbon atom number 1 in a new glucose molecule. This produces a branch point in the amylopectin molecule. Amylopectin is therefore a branched polymer.

Glycogen is similar in structure to amylopectin, but branches more frequently. Cellulose is an unbranched polymer composed of beta glucose molecules. Beta glucose is an isomer of glucose in which the hydroxyl group attached to carbon 1 is above the plane of the ring. The glucose monomers are linked by 1, 4 glycosidic bonds. Hydrogen bonds between adjacent cellulose molecules allow them to form strong fibres, which suite them to their role as the main structural component of plant cell walls.