SOLIDS LIQUIDS AND GASES KINETIC THEORY explanation of


























- Slides: 33

SOLIDS, LIQUIDS, AND GASES

KINETIC THEORY • explanation of the behavior of particles in gases: • 1. gas is made of tiny particles • 2. particles are in constant, random motion • 3. collisions between particles are elastic--no kinetic energy lost

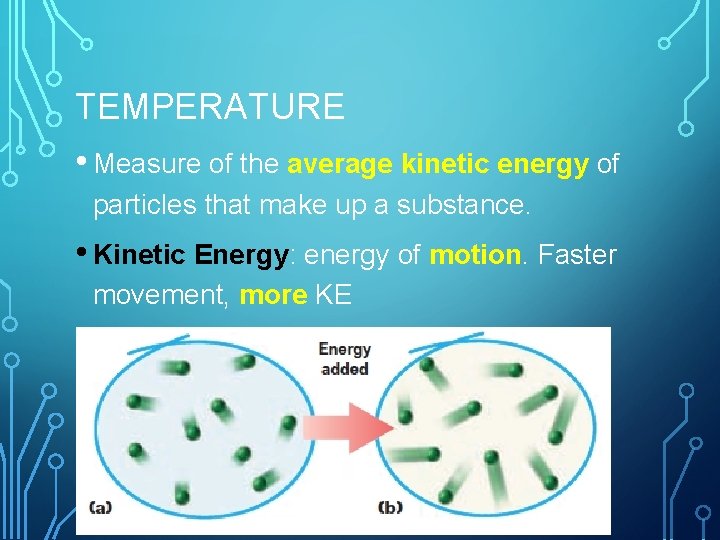
TEMPERATURE • Measure of the average kinetic energy of particles that make up a substance. • Kinetic Energy: energy of motion. Faster movement, more KE


GAS • Substance made of quickly-moving, mostly non-interacting particles • fills up the volume and shape of a container holding it.

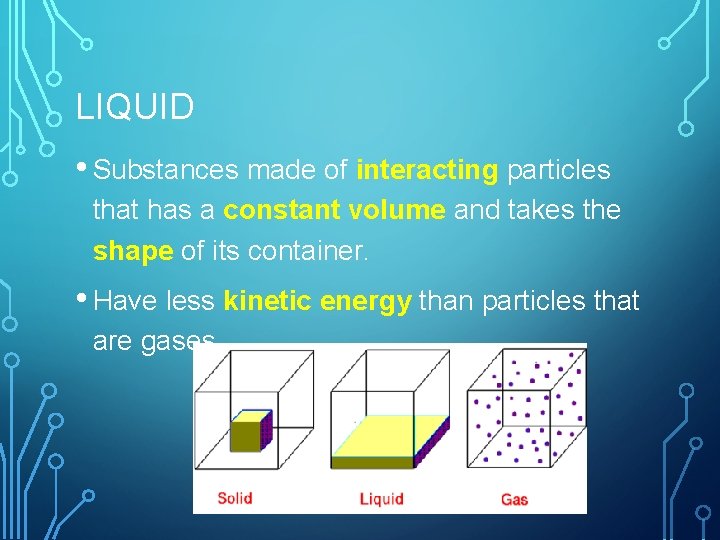
LIQUID • Substances made of interacting particles that has a constant volume and takes the shape of its container. • Have less kinetic energy than particles that are gases.

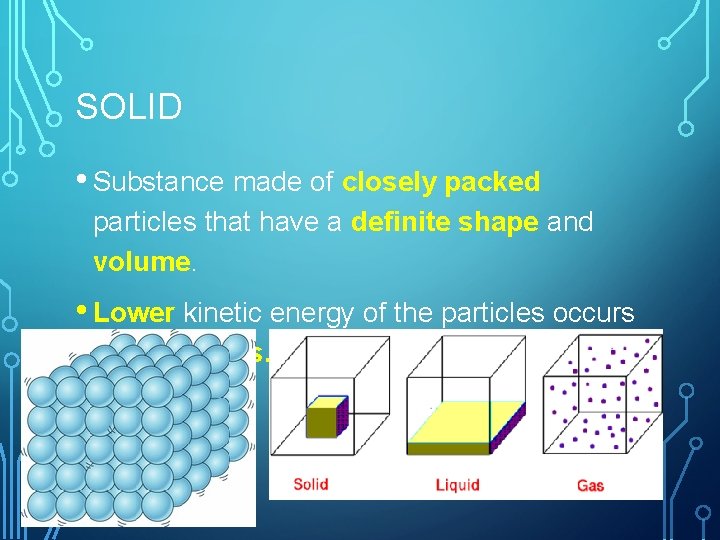
SOLID • Substance made of closely packed particles that have a definite shape and volume. • Lower kinetic energy of the particles occurs as vibrations.

PLASMA • the fourth state of matter consisting of particles with kinetic energy high enough such that electrons are stripped off of atoms • Most of the matter in the universe is in this state.

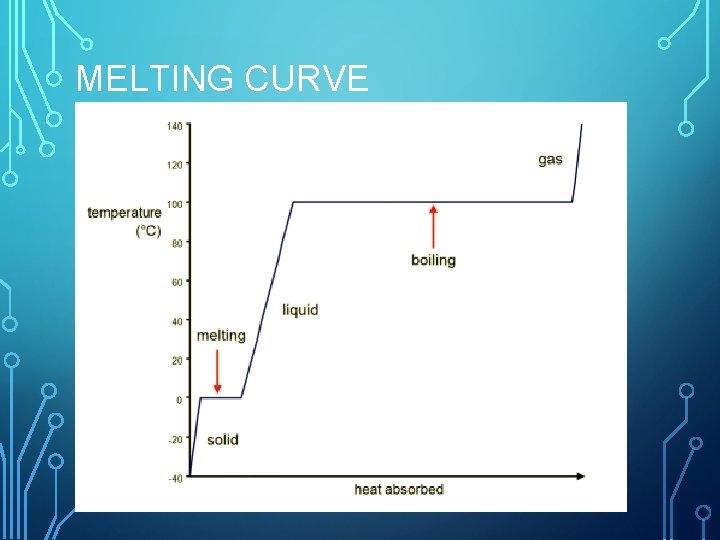
MELTING POINT • temperature at which a solid begins to liquefy. • Water: 0°C • At this temperature particles have the kinetic energy to break free from tighter interactions of the solid state

HEAT OF FUSION • amount of energy required to change a substance from the solid phase to the liquid phase. • Any energy absorbed by the solid goes to phase change, and NOT temperature change.

BOILING POINT • the temperature at which the pressure of vapor of a liquid is equal to the external pressure acting on the surface of the liquid. 100°C for water. • Vapor pressure is how much force per area particles exert on their surroundings. • Increased temperature increased K. E. increased vapor pressure

HEAT OF VAPORIZATION • amount of energy required for a liquid at its boiling point to become a gas. • Water may reach 100°C and not boil. • MORE energy is needed to give particles enough K. E. to escape interactions • Boiling fluid will remain at the boiling point temp until all particles are in gas state


MELTING CURVE

• Think: • What would happen to the boiling point of water if the atmospheric pressure were lowered? • Hint: look at boiling point definition • Turn and Talk • Share

SUBLIMATION • the process of a solid changing directly to a vapor without forming a liquid.

DENSITY •

DENSITY • Generally, a solid is more dense than a liquid, which is more dense than a gas • Water is an exception: ice is less dense than liquid water • Density of water = 1 g/m. L (exactly) at 4°C.

THERMAL EXPANSION • increase in the volume of a substance when the temperature is increased • Density decreases

DENSITY PRACTICE PROBLEMS • Pure solid gold has a density of 19. 32 g/cm 3. What is the mass of a cubic meter (or 1, 000 cm 3) of gold? • What is the density of ice if 5. 33 cm 3 of ice has a mass of 4. 886 g?

AMORPHOUS SOLIDS • solids lacking a crystalline structure that do not have a specific melting point • Examples: • Butter, solid grease, petroleum jelly

BUOYANCY • ability of a fluid (liquid or gas) to exert an upward force on an object immersed in it.

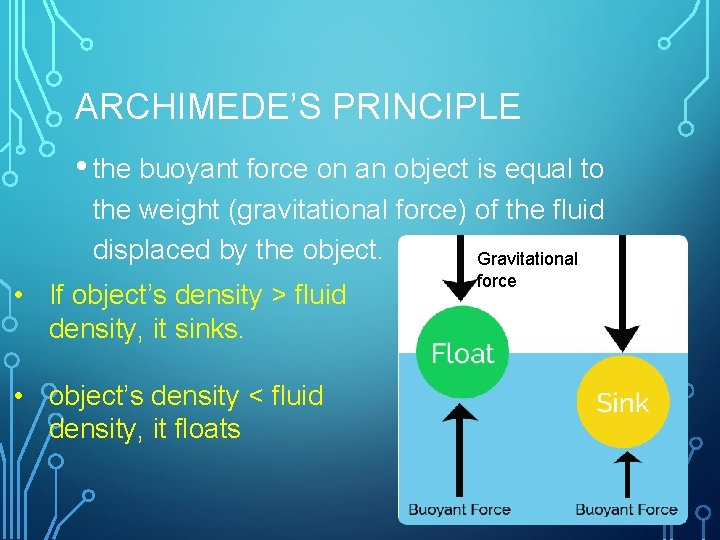
ARCHIMEDE’S PRINCIPLE • the buoyant force on an object is equal to the weight (gravitational force) of the fluid displaced by the object. Gravitational • If object’s density > fluid density, it sinks. • object’s density < fluid density, it floats force

IF AN OBJECT IS FLOATING… • Weight of object (gravitational force) is balanced with the opposing weight of the water displaced (buoyant force) • Weight of object = weight of water displaced • Use the density equation to convert from volume of water displaced to mass/weight of water displaced.

WHY DO WE FLOAT IN THE DEAD SEA?

ARCHIMEDES' PRINCIPLE PRACTICE 1. A man is floating in water. The buoyant force acting on his body is 210 lbs (pounds are a unit of force). What is the man’s weight? 2. A river barge weighs 5, 340 N. What is the weight of the water it displaces when it floats? 3. A floating object displaces 10. 0 cubic feet (ft 3) of water. How much does the object weigh if the weight density of water is 62. 43 lbs/ft 3?

PRESSURE • amount of force exerted per unit area.

UNITS OF FORCE • Newtons (N) and pounds (lbs)

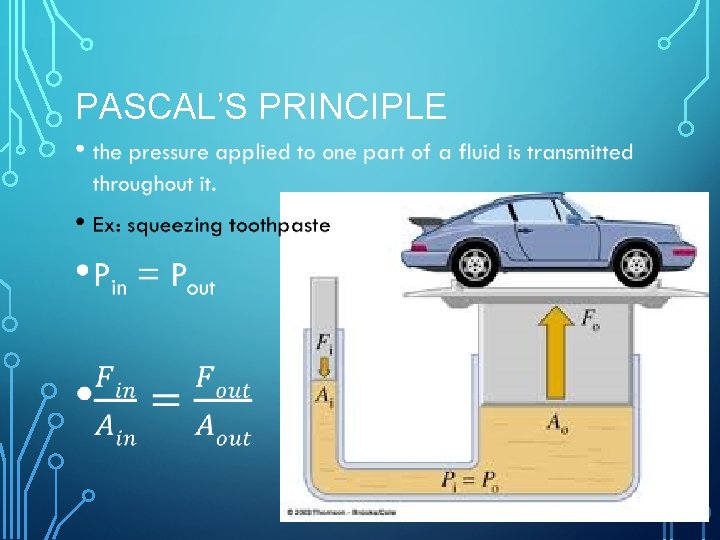
PASCAL’S PRINCIPLE •

BERNOULLI’S PRINCIPLE • as the velocity of a fluid increases, the pressure exerted by that fluid decreases. Higher pressure

BOYLE’S LAW •

KELVIN (UNITS) • units of temperature equal in magnitude to Celsius, that begin (zero value) at -273. 15 C. • 0 K is called absolute zero. NO kinetic energy.

CHARLES’ LAW •