Kinetic theory the behaviour of gases Learning outcomes

Kinetic theory & the behaviour of gases

Learning outcomes use a particle model to describe solids, liquids, gases & changes of state explain gas pressure and thermal expansion in terms of kinetic theory describe how a barometer measures atmospheric pressure estimate the height of the atmosphere using a physical model recall and use the gas laws to make quantitative predictions relate the gas laws & absolute zero of temperature to the behaviour of ideal gases illustrate how science works through historical case studies introduce microscopic atoms and molecules through reasoning based on careful observation of macroscopic behaviour use apparatus for relevant demonstration and class experiments convert temperatures between Celsius & Kelvin scales

Misconceptions Students often think that particles • have the properties of bulk matter (particles change in size as the temperature changes, particles can melt and solidify) • have air in between them • have thoughts and intentions e. g. ‘they prefer to move to places that are less crowded’. There is much confusion about the nature of particle motion in solids, liquids and gases.

Teaching challenges Atoms and molecules are far too small to be glimpsed by even the most highly-powered optical microscope. Diagrams of particle arrangements are often static. • Dynamic animations showing the random thermal motion of particles, at all temperatures and in all states of matter, usefully overcome the misconceptions which static diagrams can foster. Students find it difficult • • • to appreciate that gas pressure acts equally in all directions to account for the consequences of pressure differences to convert between units of volume (cm 3, m 3)

All things are made of atoms “If, in some cataclysm, all of scientific knowledge were to be destroyed, and only one sentence passed on to the next generations of creatures, what statement would contain the most information in the fewest words? “I believe it is the atomic hypothesis (or the atomic fact, or whatever you wish to call it) that all things are made of atoms - little particles that move around in perpetual motion, attracting each other when they are a little distance apart, but repelling upon being squeezed into one another. “In that sentence, you will see, there is an enormous amount of information about the world, if just a little imagination and thinking are applied. ” Richard Feynman

Evidence for atoms In pairs: What evidence for atoms can you, or do you, show your pupils?

Evidence for atoms • crystals – regularity of surfaces, cleaving • mixing different liquids • change of volume: solid gas, liquid gas • air occupies space and has mass • diffusion: solid into solid, solid into liquid, gas into gas • Brownian motion See the Practical Physics website, ‘Molecules in motion’ collection
Many states of matter solid, liquid, gas, plasma … superfluids, liquid crystals, solid solutions, aerogels, foams, thin films, colloids, immiscible liquid mixtures, gas dissolved in liquid, condensed matter, biopolymers…. .

Molecular models • solid • liquid • gas Discuss, first in pairs, then in groups: How does each model account for observable physical behaviour – shape, ability to flow, elasticity, surface, changes of state?

Gases: bulk properties pressure = force applied over a unit area. Later: see SPT Forces, episode 8 Pressure

An ideal gas • huge number of point molecules (occupy negligible volume) in continual random motion (and so ‘kinetic’) • colliding elastically with each other and with container walls • no forces between the molecules, except in collision • time in collisions very small compared to time between collisions • distance travelled between collisions (‘mean free path’) depends on gas density • average speed of molecules depends on gas temperature • in a gas composed of different molecules, the average molecular Ek is the same for all, so those with larger mass have smaller speed
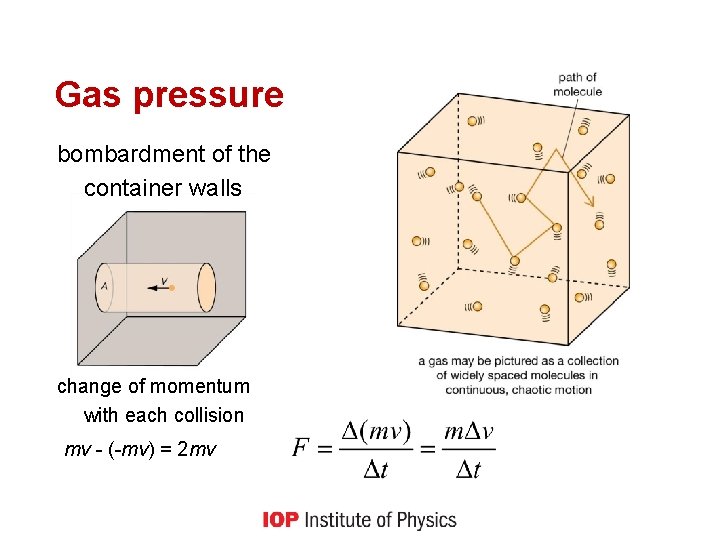
Gas pressure bombardment of the container walls change of momentum with each collision mv - (-mv) = 2 mv

The speed of gas molecules Ways of estimating an average speed in air - from the speed of sound (340 ms-1 at s. t. p. ) - thought experiment: a molecule falls freely from the top of the atmosphere Direct measurement: Zartman (1931) experiment to find the distribution of molecular speeds in a beam emitted from an oven opening. Average speed of N 2 at room temperature ~ 500 ms-1
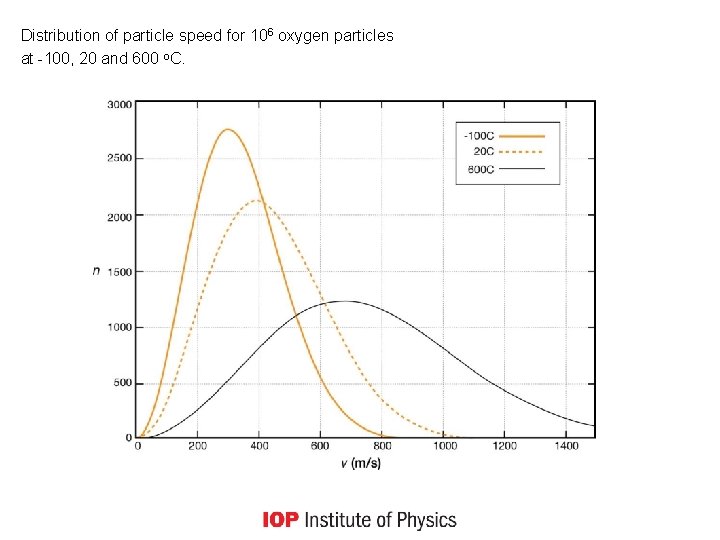
Distribution of particle speed for 106 oxygen particles at -100, 20 and 600 o. C.

The size of a molecule oil film experiment Devised by John William Strutt, Lord Rayleigh, who also explained why the sky looks blue, and many other things! Nobel prizewinner 1904.

The size of atoms ‘If an apple were magnified to the size of Earth, the atoms in it would each be about the size of a regular apple. ’ Richard Feynman AFM showing atoms within hexagonal graphite unit cells. Image size 2 nm × 2 nm.

Models made real metallurgy (e. g. sword-making) existed long before any theory of metal structure. secret techniques to develop toughness & hardness, found by trial and error (e. g. heat treatments, hot working) late 19 th C model (polycrystalline structure of metals - grains) metallurgists studied polished and etched metal surfaces under a microscope, X-ray crystallography from 1911 theory of cracks (dislocations and defects in metal lattice) limiting strength, confirmed by electron microscopy after 1937 materials by design e. g. transistor 1947, pure glass fibre 1970, 1000 s of steel alloys today. Determining properties using computer simulations.

Galileo and Torricelli Task: Read chapters 3 & 4 from Joan Solomon (1989) The big squeeze. ASE booklets series The Nature of Science. Discuss: Could you use an extract from this in your teaching? ………………………………. . . How to make a water barometer in school, for a participative demonstration.
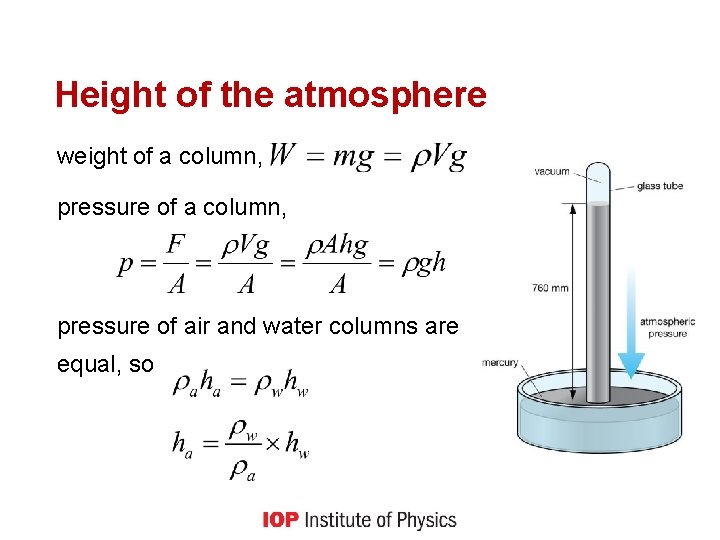
Height of the atmosphere weight of a column, pressure of a column, pressure of air and water columns are equal, so

Hydraulic machines exploit these facts: • pressure is the same throughout a fluid (at same height). • liquids are incompressible. How do hydraulic systems work? In pairs: Explore SPT Forces episode 8. (25 min)
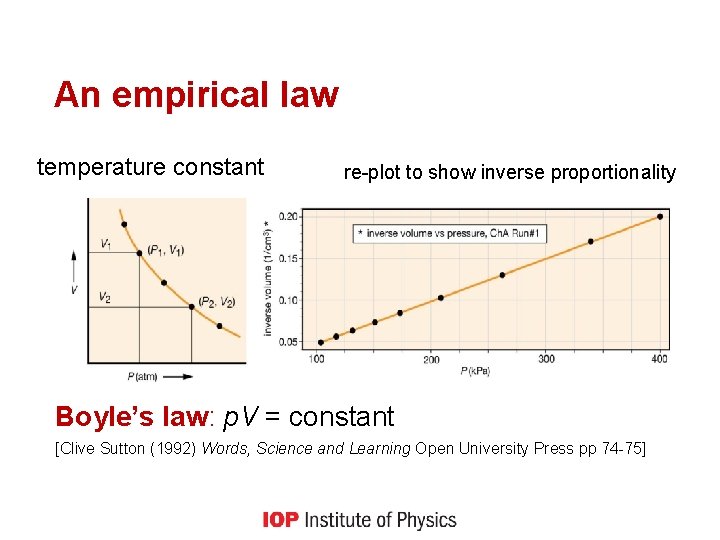
An empirical law temperature constant re-plot to show inverse proportionality Boyle’s law: p. V = constant [Clive Sutton (1992) Words, Science and Learning Open University Press pp 74 -75]
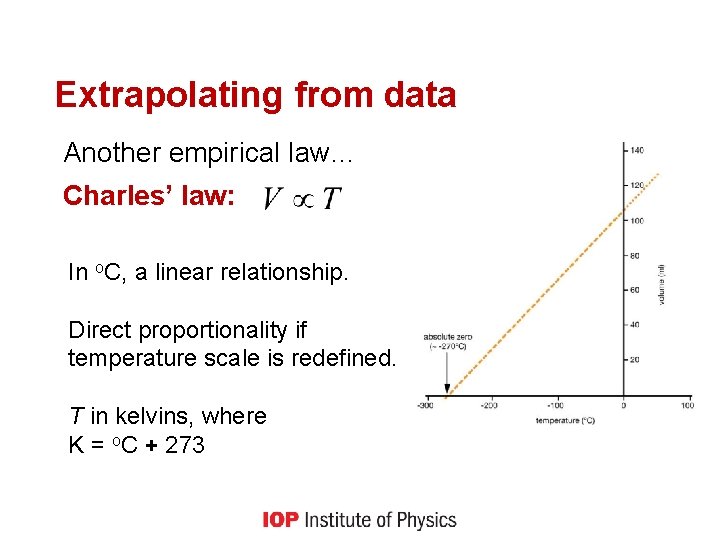
Extrapolating from data Another empirical law… Charles’ law: In o. C, a linear relationship. Direct proportionality if temperature scale is redefined. T in kelvins, where K = o. C + 273

Other gas laws pressure law: (T in kelvins) All three relationships combined: where n is number of moles, gas constant R = 8. 31 J K-1 This means

Kinetic theory – a chronology c. 420 BC – atomic theory (Democritus: matter ultimately uncut-able) 1662 – Boyle’s law 1738 – Bernoulli Hydrodynamica (molecular collisions gas pressure) 1787 – Charles’ law 1798 – atomic theory of heat 1827 – Brownian motion 1834 – ideal gas law 1849 – kinetic theory k = 1. 38 x 10 -23 J K-1 (Boltzmann constant)

Gas properties Ph. ET simulation Gas properties Pump gas molecules to a box and see what happens as you change the volume, add or remove heat, change gravity, and more. Measure the temperature and pressure, and discover how the properties of the gas vary in relation to each other.
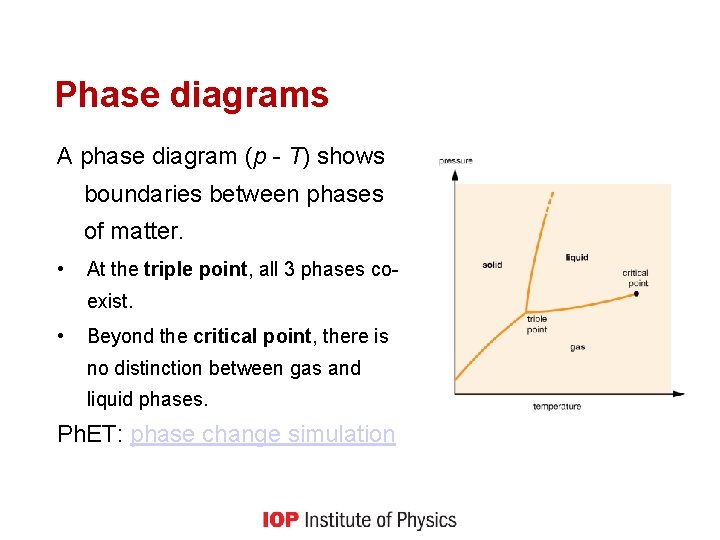
Phase diagrams A phase diagram (p - T) shows boundaries between phases of matter. • At the triple point, all 3 phases coexist. • Beyond the critical point, there is no distinction between gas and liquid phases. Ph. ET: phase change simulation

Speed of sound When a sound wave travels in a medium, particles in the medium are subjected to varying stresses, with resulting strains. The speed of sound is partly affected by the elasticity (stiffness) of the medium, E, and partly by the density, , of the medium. In a gas, the sound (pressure) wave is an adiabatic process (no thermal transfers), so

Physics of society? measuring society e. g. • • Thomas Hobbes (1651) Leviathan. political philosophy (science) based on ‘natural philosophy’ Enlightenment (especially in France): social statistics to measure society (large numbers: order emerges from random behaviour) physics e. g. • • kinetic theory & phases changes (statistical mechanics) growth and form in nature – fractal patterns, chaos theory physics-based modelling human behaviour e. g. • • • modelling geography – e. g. growth of cities, transport networks modelling road traffic – changes of state financial forecasting Philip Ball (2004) Critical mass: how one thing leads to another

Educational constructivism Four valuable ideas: • the importance of the pupil’s active involvement in thinking, if anything like understanding is to be reached • the importance of respect for the child and for the child’s own ideas • that science consists of ideas created by human beings • that the design of teaching should give high priority to making sense to pupils, capitalising and using what they know and addressing difficulties that may arise from how they imagine things to be ‘To make knowledge one’s own is crucial for satisfying learning, though not at all a simple or quick process. ” Ogborn J (1997) ‘Constructivist Metaphors of Learning Science’, Science and Education 6: 121 -133
- Slides: 30