Solids Liquids and Gases Kinetic Theory The kinetic




















- Slides: 24

Solids, Liquids, and Gases

Kinetic Theory • The kinetic theory is an explanation of how particles in matter behave. – All matter is composed of small particles (atoms, molecules, ions) – These particles are in constant, random motion. – These particles are colliding with each other and the walls of their container.

Cool Science • http: //youtu. be/b. Ph 1 KXA 8 eg. U

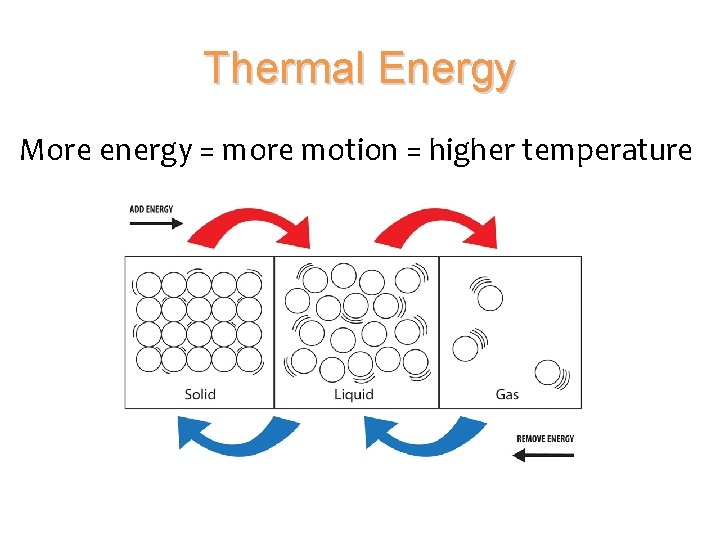
Thermal Energy • All objects have energy • Potential Energy – from the bonds that hold the substance together • Kinetic Energy – from the motion or vibration of the atoms and molecules • The total energy of a material’s particles is called thermal energy.

Thermal Energy More energy = more motion = higher temperature

Temperature • Temperature measures the average kinetic energy of the particles in a substance.

Solids • Particles are closely packed together • Particles have different geometric arrangements. – These arrangements determine the physical and chemical properties.

Carbon

Liquids • Particles have enough energy to break free from the geometric arrangement. • Substance flows and takes the shape of its container. • Melting point – temperature at which a substance goes from solid to liquid • Heat of Fusion – energy required to reach the melting point

Gases • Particles have enough energy to escape the attractive force of the other molecules. • Expand to take the size and shape of their container • Heat of Vaporization – amount of energy required for a liquid to become a gas • Diffusion – spreading out of gas molecules to fill their container.

Boiling • Boiling point – temperature at which a substance goes from a liquid to a gas. • Liquids stay in their container because of air pressure. • Liquids boil because the internal pressure of the liquid becomes equal to the pressure of the air and the molecules are allowed to escape.

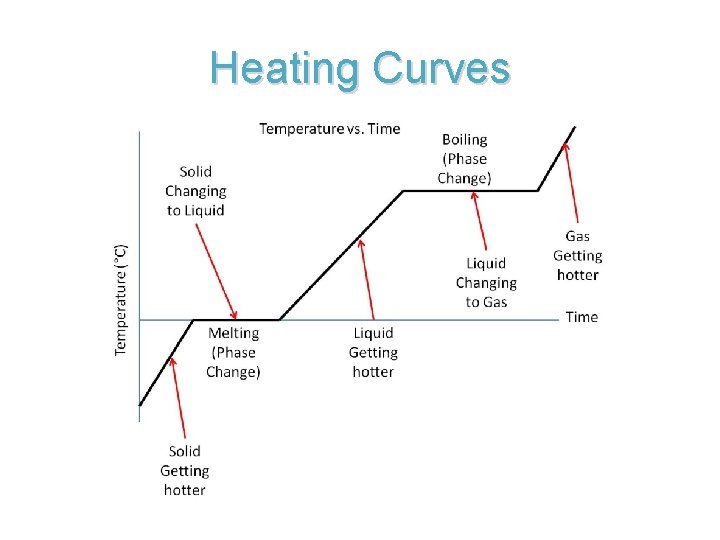
Heating Curves • Graph that shows the states of matter of a substance. • As a solid melts and a liquid vaporizes the temperature remains constant. • The temperature increases after the phase change has happened.

Heating Curves

Plasmas • High temperature gas with positively and negatively charged particles. • Found in lightning and neon signs

Thermal Expansion • Increase in the size of a substance when it is heated. • Occurs in most solids, liquids, and gases. • Water is the exception.

Weird Things • Some substances do not react as expected when changing states. – Amorphous Solids • Lack the geometric pattern of other solids • Glass and Plastic • No definite melting/boiling point – Liquid Crystals • Keep their geometric arrangement when liquid • Liquid Crystal Displays – Non-Newtonian Substances • Acts as different states based on certain conditions

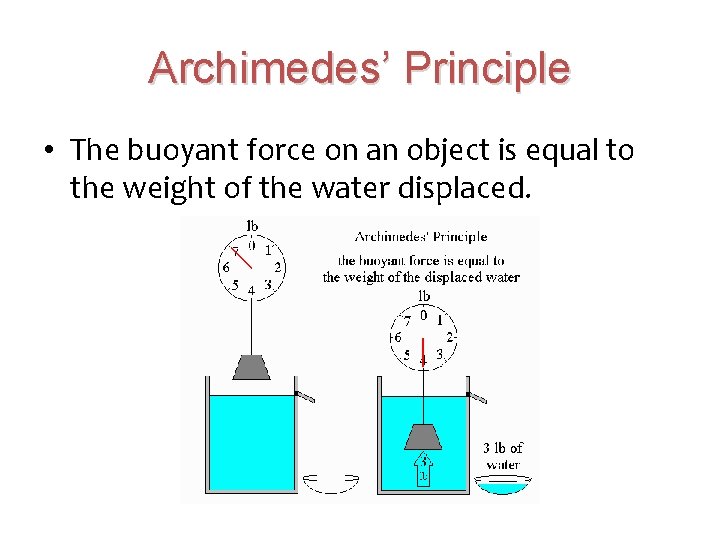
Buoyancy • The ability of a fluid (liquid or gas) to exert an upward force on an object immersed in it. • If the buoyant force is equal to the weight of the object, the object will float. • If the buoyant force is less than the weight of the object, the object will sink.

Archimedes’ Principle • The buoyant force on an object is equal to the weight of the water displaced.

Density • • Mass ÷ Volume More dense objects sink Less dense objects float To increase the density you can: – Decrease the volume – Increase the mass • To decrease the density you can: – Increase the volume – Decrease the mass

Pressure • Pressure = Force / Area • To increase the pressure: – Increase the force – Decrease the area • To decrease the pressure: – Decrease the force – Increase the area

Pascal’s Principle • Pressure is constant throughout a fluid • In simple terms, pressure is the same in a fluid. • Force 1 / Area 1 = Force 2 / Area 2 • Hydraulic Lifts

Bernoulli’s Principle • As the velocity of a fluid increases, the pressure decreases. • Fast air = less pressure • Slow air = more pressure • Airplane wings are designed so that the air moves faster on the top. That means there is a small pressure pushing down and a bigger pressure pushing up. The airplane is then able to fly.

Viscosity • The resistance of a fluid to flow • Low viscosity = flows easily (water, milk) • High viscosity = does not flow easily (syrup, ketchup)

Assignment • Section 2 Reinforcement • On the back, answer these questions: – What are two opposing forces that act on an object floating in water? – Why do helium balloons float and non helium balloons sink? – Why are roofs lifted off houses during tornados? – How is it possible for a heavy boat to float on water?