MATTER What is matter Matter is anything that





























- Slides: 37

MATTER ~ What is matter? ~ Matter is anything that has ______ __ MASS & TAKES UP _____ SPACE ~ Name & draw 5 things that ARE matter.

Is AIR matter? Is LIGHT matter? Does air have MASS? Does light have MASS? Does air TAKE UP SPACE? Does light TAKE UP SPACE? Complete the chart in your notes, and draw and label pictures for each item.

Atoms, Elements, Compounds, and Ions • Atom • basic building block of all matter • Element • Substance that consists of only one type of atom. • Molecule has two different atoms. • Compound • substance that consists of more than one type of element. • Ion • substance that has a positive or negative charge


4 Principles of the Particle Theory matter is made of tiny particles. All particles of one substance are the same. - Different substances are made of different particles. The particles are always moving. - The more energy the particles have, the faster they move. There attractive forces between particles. - These forces are stronger when the particles are closer together.

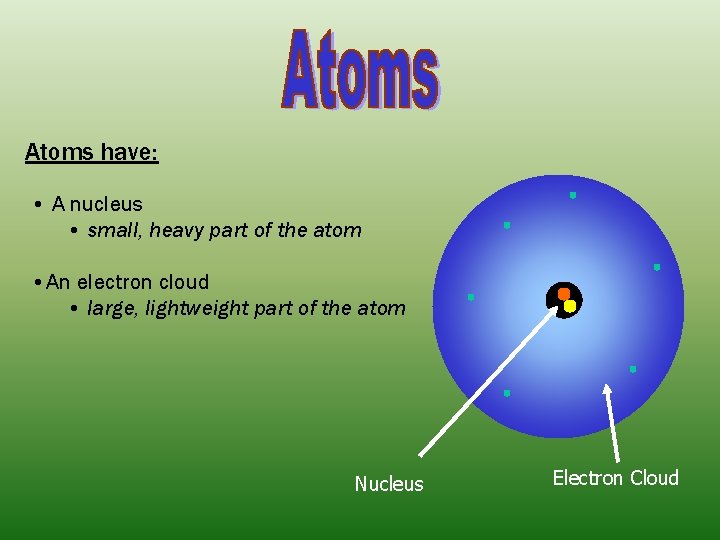
Atoms have: • A nucleus • small, heavy part of the atom • An electron cloud • large, lightweight part of the atom Nucleus Electron Cloud

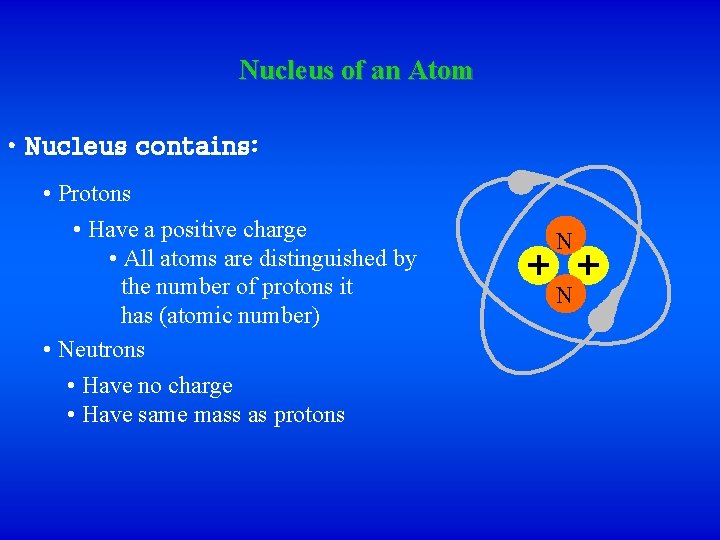
Nucleus of an Atom • Nucleus contains: • Protons • Have a positive charge • All atoms are distinguished by the number of protons it has (atomic number) • Neutrons • Have no charge • Have same mass as protons N N N

Electron Cloud of an Atom An electron cloud contains: • Electrons e- • have a negative charge e- • are contained within shells of the electron cloud • orbit the nucleus of the atom • have a very small mass compared to protons and neutrons • determine bonding properties of substance

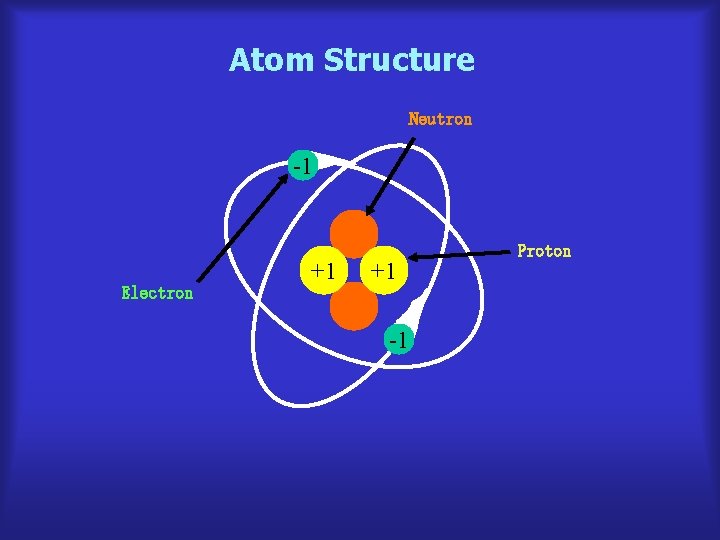
Atom Structure Neutron -1 Electron +1 +1 -1 Proton

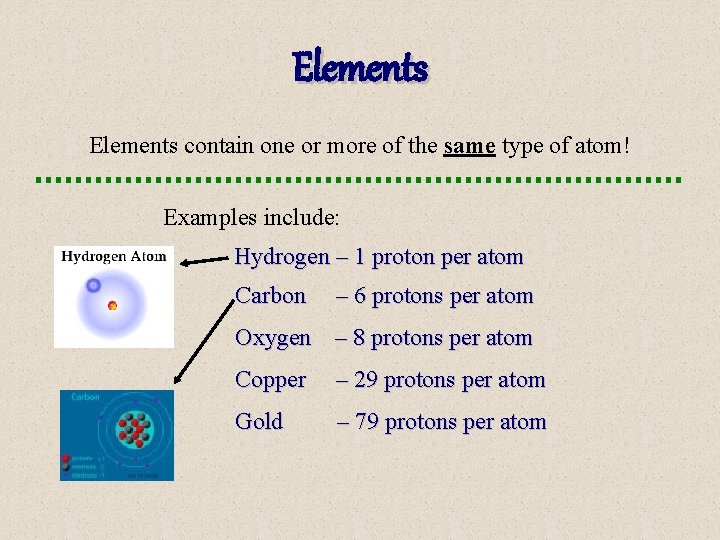
Elements contain one or more of the same type of atom! Examples include: Hydrogen – 1 proton per atom Carbon – 6 protons per atom Oxygen – 8 protons per atom Copper – 29 protons per atom Gold – 79 protons per atom

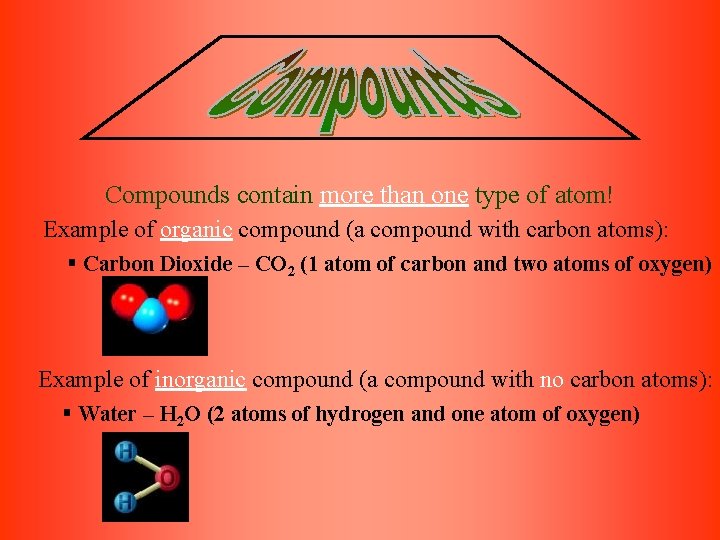
Compounds contain more than one type of atom! Example of organic compound (a compound with carbon atoms): § Carbon Dioxide – CO 2 (1 atom of carbon and two atoms of oxygen) Example of inorganic compound (a compound with no carbon atoms): § Water – H 2 O (2 atoms of hydrogen and one atom of oxygen)

- + An ion is an atom or group of atoms with a positive or negative charge!! § A particle with a neutral charge has the same number of protons and electrons. § An ion does not have the same number of electrons and protons. § Examples of ions: • He+ - A helium atom that is missing one electron. The atom has one more proton than electron, and must have a positive charge. • CO 32 - - Carbonate has two more electrons than protons

Lets take a look deep inside

Lets take a look deep inside

Lets take a look deep inside

Lets take a look deep inside





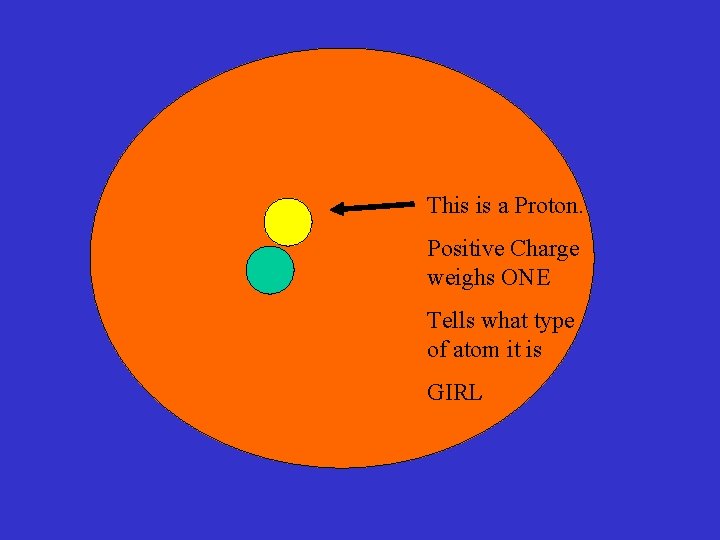
This is a Proton. Positive Charge weighs ONE Tells what type of atom it is GIRL

This is a Proton. Positive Charge weighs ONE Tells what type of atom it is GIRL

This is a Proton. Positive Charge weighs ONE Tells what type of atom it is GIRL

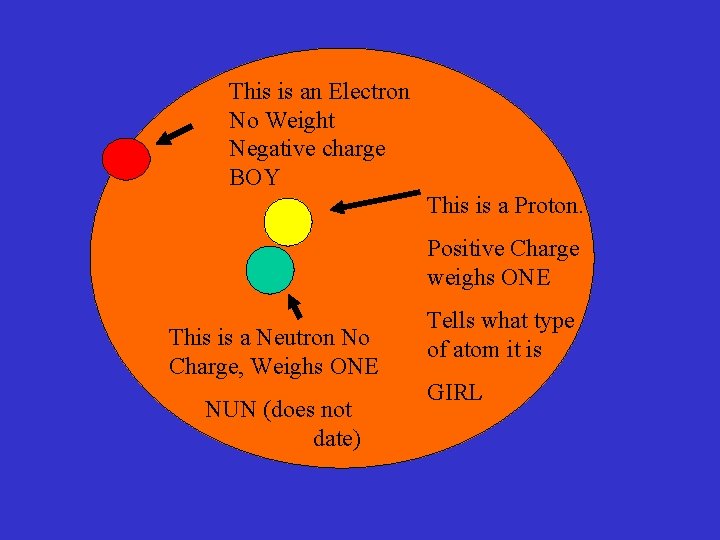
This is a Proton. Positive Charge weighs ONE This is a Neutron No Charge, Weighs One NUN (does not date) Tells what type of atom it is GIRL

This is an Electron No Weight Negative charge BOY This is a Proton. Positive Charge weighs ONE This is a Neutron No Charge, Weighs ONE NUN (does not date) Tells what type of atom it is GIRL


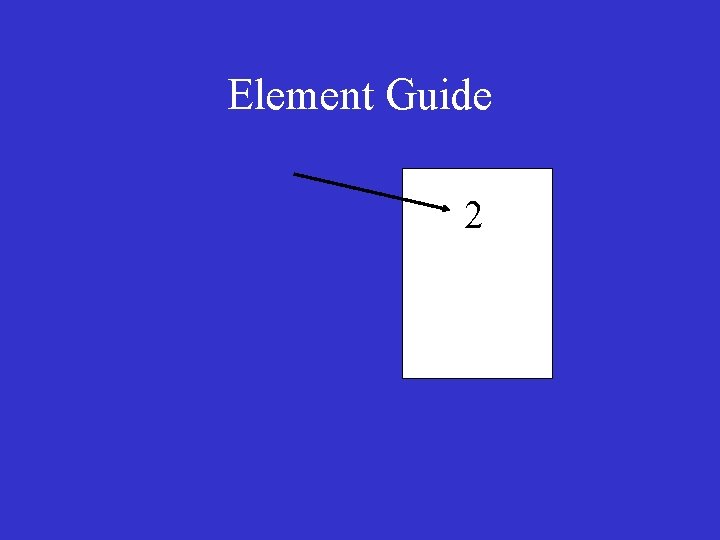
Element Guide

Element Guide 2

Element Guide 2

Element Guide Proton Number-tells what element it is. (also the atomic number) 2

Element Guide Proton Number-tells what element it is. (also the atomic number) 2 He

Element Guide Proton Number-tells what element it is. (also the atomic number) 2 He

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. 2 He

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. 2 He 4

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. 2 He 4

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. Explains the total atomic weight-The number of protons PLUS the number of neutrons. 2 He 4

Element Guide Proton Number-tells what element it is. (also the atomic number) Explains name of the element. Explains the total atomic weight-The number of protons PLUS the number of neutrons. 2 He 4