Matter Substance Mixture Matter What Is Matter Anything

Matter Substance Mixture

Matter • What Is Matter? – Anything that has mass and takes up space is matter. • Mass is how much matter there is of an object. • Volume is how much space matter takes up. – Matter is anything made of atoms and molecules.
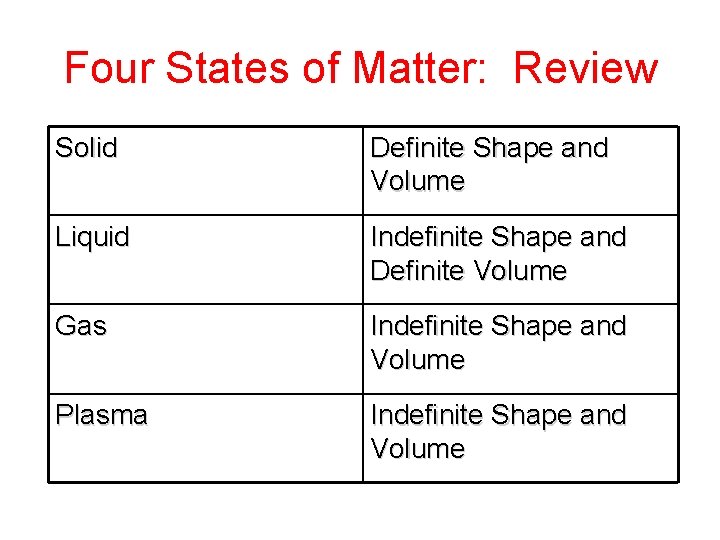
Four States of Matter: Review Solid Definite Shape and Volume Liquid Indefinite Shape and Definite Volume Gas Indefinite Shape and Volume Plasma Indefinite Shape and Volume

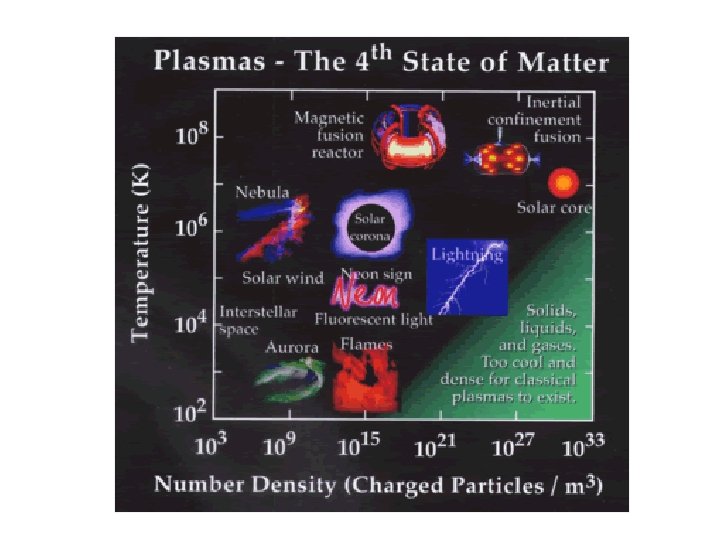
Plasmas • • • Form under conditions of extremely high energy - molecules are ripped apart Plasma made up of free electrons and element ions Very high KE - hot, ionized gases - electrons are ripped off of individual atoms. Gas-like - indefinite shape and volume Very high KE –particles collide with enough energy to break into charged particles Because the atoms in plasma exist as charged ions, plasmas behave differently than gases - free ions cause plasma to be electrically conductive.
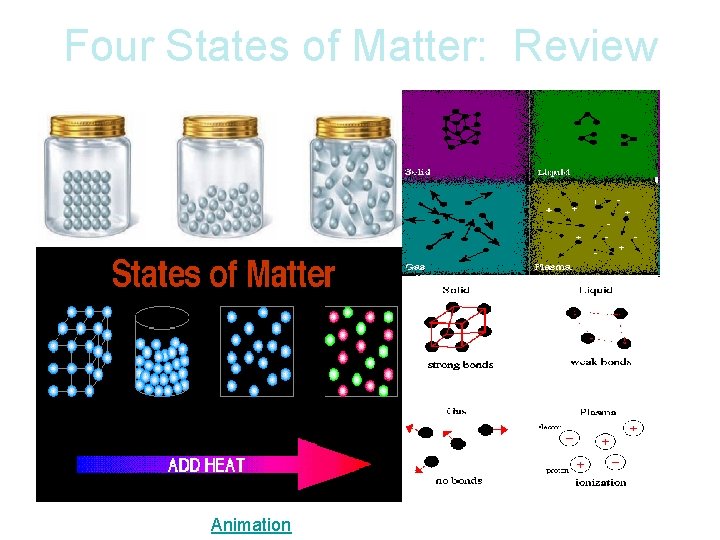
Four States of Matter: Review Animation
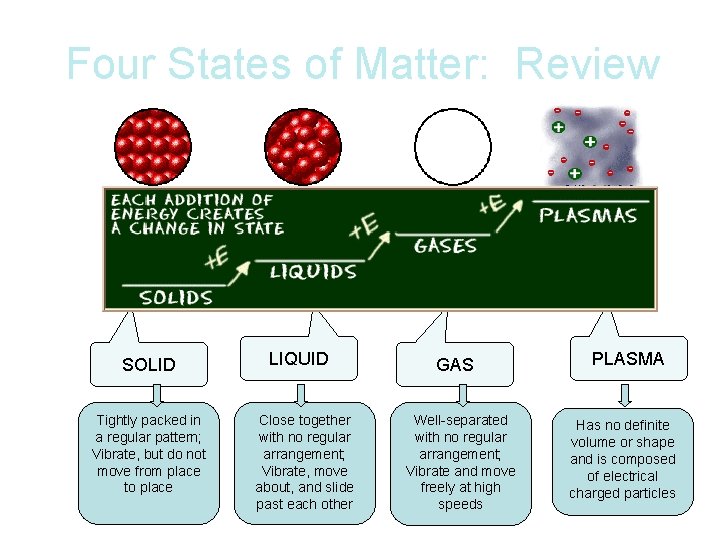
Four States of Matter: Review SOLID Tightly packed in a regular pattern; Vibrate, but do not move from place to place LIQUID Close together with no regular arrangement; Vibrate, move about, and slide past each other GAS Well-separated with no regular arrangement; Vibrate and move freely at high speeds PLASMA Has no definite volume or shape and is composed of electrical charged particles

What is the 5 th state of matter? Bose- Einstein Condensate 2016 Experiment on the ISS Practical Applications in the real world Cold Atom Lab
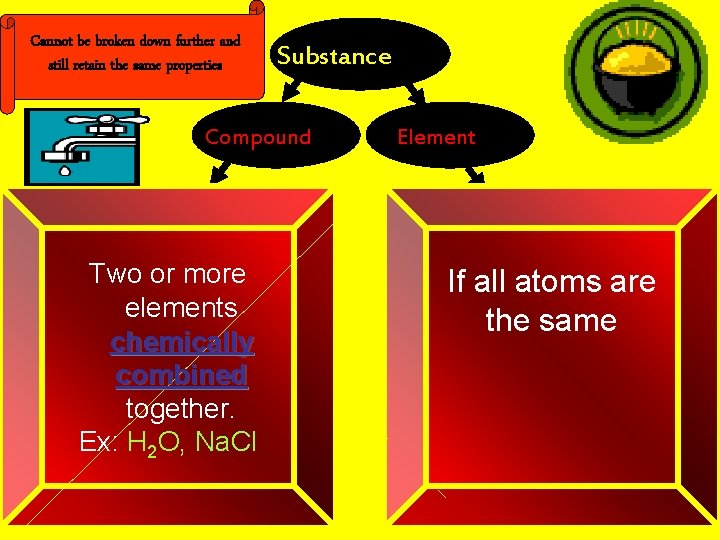
Cannot be broken down further and still retain the same properties Substance Compound Two or more elements chemically combined together. Ex: H 2 O, Na. Cl Element If all atoms are the same

Two or more substances that can be separated by physical means. Heterogenous the substances in the mixture are not evenly mixed Salad dressings, potting soil Mixture Not Chemically combined Homogenous the substances are in the same amount in all parts of the mixture also known as solutions Tea, perfume

5 A solution is made up of two parts—a solute and a solvent A solute is the substance being dissolved, while a solvent is the substance doing the dissolving. For example, in a saltwater solution, salt is the solute and water is the solvent. The ability of a substance to dissolve in another substance is called solubility

6

7 Chemical Properties • The property that describes how a substance can change to form new substances is called a chemical property. ØAbility to Burn ØAbility to Corrode ØAbility to Support Burning ØReactivity with Chemicals ØReactivity with Light

Physical Properties Physical properties can be observed or measured without changing the identity of the substance � Boiling Point � Color � Conductivity � Density � Malleability � Mass � Melting Point � Odor � Shape � State of Matter � Weight • Color, shape, and odor are properties that can be observed using your senses. • Other physical properties, like mass, weight, volume, density, and conductivity can be measured.


Chemical Changes � Combustion In a chemical � Corrosion change, one or � Electrolysis more substances � Fermentation combine or � Metabolism Photosynthesis break down to � form new substances. • Sometimes energy in the form of heat, light, or electricity is given off. • You may see bubbles and there may be a color change. • In some chemical changes, energy is required for the chemical change to take place.

Physical Changes v. A physical change alters the form of a substance, but not the composition of the substance v. Any change in the state of matter is a physical change üBoiling ü Condensation ü Dissolving ü Evaporation ü Freezing ü Melting ü Sublimation

Is the Change Permanent? To help you remember the difference between physical change and chemical change, ask yourself this question, “Can I get the original substances back after the change? ” If so, it is a physical change. If not, it is changed forever and is a chemical change.

Colloids and Suspensions Colloid • Mixture that never settles. • Jello • Tyndall Effect- light scatters when it goes through a colloid Suspension • Mixtures that settle out. • EX: Muddy River Water
- Slides: 20