STATES OF MATTER MATTER Matter is anything that



































- Slides: 39

STATES OF MATTER

MATTER Matter is anything that takes up space and has mass. Matter does not have to be visible. Air is matter.

States of Matter • • Three states of matter common on Earth: Solids Liquids Gases States of matter in the Universe Plasma Bose-Einstein Condensate 1995 Fermionic Condensate 2004

The Fifth State of Matter • • Bose-Einstein Condensation in a gas: a new form of matter at the coldest temperatures in the universe. . . It is the ability to slow down light. Atoms move faster than photons. • • Predicted 1924. . . Created 1995 • A. Einstein S. Bose

The Sixth State of Matter • Scientists have created a new form of matter, which they say could lead to new ways of transmitting electricity and communication to the Moon and other planets. • The fermionic condensate is a cloud of cold potassium (P -40) atoms forced into a state where they behave strangely. It is considered a super cooled, strange superparticle and a superfluid.

Sixth State of Matter The fermionic condensate

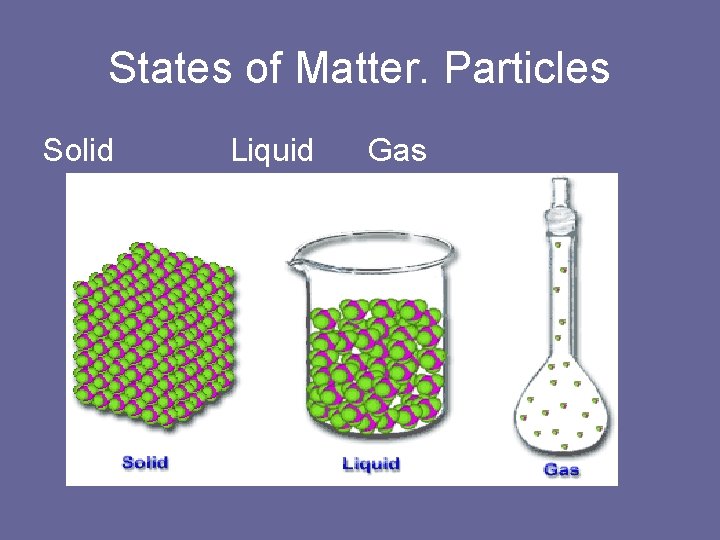
Solid • Solid is matter with a define shape and volume. • Particles that make up all types of matter are in constant motion.

Crystalline Solids • Particles are arranged in repeating threedimensional patterns, called crystal.

Amorphous Solids • Solids with large particles arranged randomly. • Example: plastic, rubber.

Surface Tension • Uneven forces acting on the particles of the surface of the liquid. • The surface of the liquid acts as a thin film were stretched across its surface. Ex: floating an insect on the surface of water.

Gases • Matter does not have a definite shape or volume. Vapor Matter that exist in the gas state but is generally a liquid or solid at room temperature. Ex: water is a liquid. water vapor is a gas.

States of Matter. Particles Solid Liquid Gas

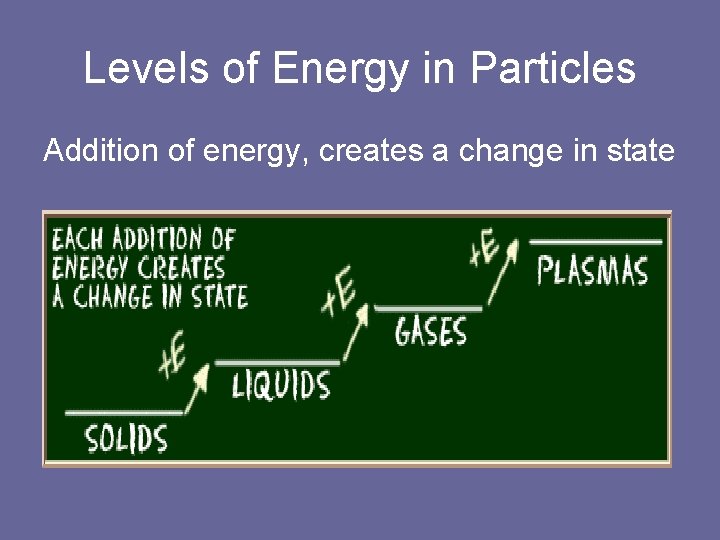
Levels of Energy in Particles Addition of energy, creates a change in state

Changes of State of Matter • Kinetic Energy is the total energy of motion. • Particles of matter are at constant motion. • The amount of motion depends of the kinetic energy of the particles. • Particles moving slowly have less kinetic energy. ( solids) • Particles moving faster have more kinetic energy. ( gases)

Temperature • The average kinetic energy of the particles that make up a substance.

Heat • The movement of thermal energy from a substance at a higher temperature to one at a lower temperature is called heat.

Specific Heat • The specific heat of a substance is the amount of heat required to raise the temperature of 1 Kg. of a substance in 1 Celsius degree.

Matter. - Materials that have a low specific heat • Metals • Aluminum, Gold Sand

Materials that have high specific heat • Water

Melting • The change from a solid to the liquid state.

Freezing • The change from the liquid state to the solid state.

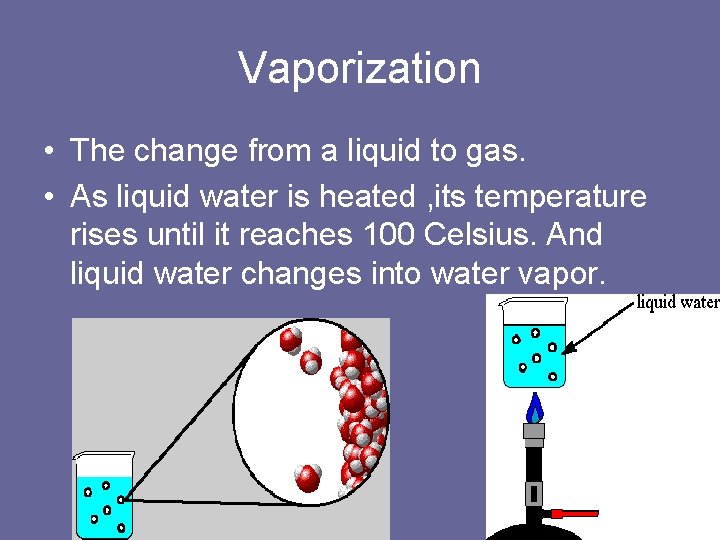
Vaporization • The change from a liquid to gas. • As liquid water is heated , its temperature rises until it reaches 100 Celsius. And liquid water changes into water vapor.

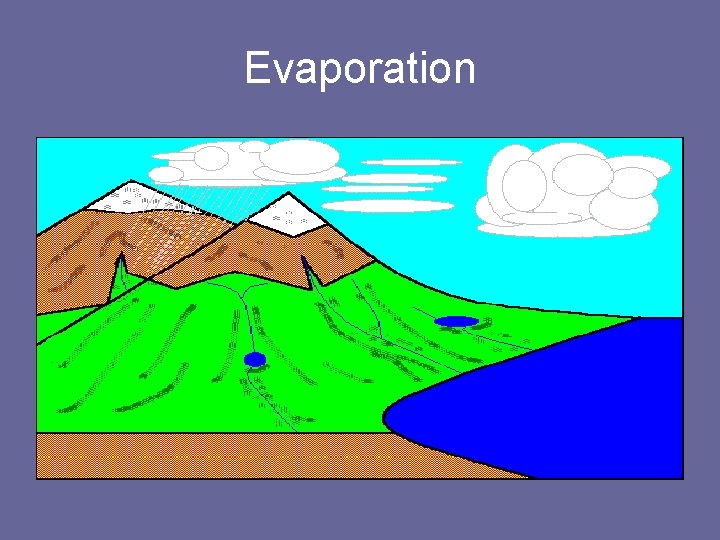
Evaporation • The conversion of water from a liquid into a gas. • Water is transferred from the surface to the atmosphere through evaporation, the process by which water changes from a liquid to a gas.

Evaporation

Condensation • It is the opposite process of vaporization. • A gas condenses to a liquid. • The decrease in energy changes the arrangements of the particles.

Sublimation • This is a drawing of what the surface of a comet might look like. This is a picture of dry ice (frozen CO 2) sublimating.

Sublimation • Sublimation of an element or compound is a transition from the solid to gas phase so rapidly that the liquid phase cannot be observed. Sublimation is a phase of transition where solid gain enough energy to transform in gas.

Deposition • The process of a gas changing directly into a solid is called deposition or desublimation. • For example, water vapor in sub-freezing air can transform into ice • without going through • the liquid phase, which is • how snow and frost are formed.

Viscosity • Some liquids flow more easily than others. • A liquid resistance to flow is known as viscosity. • Slower the liquid flow, higher the viscosity. • High viscosity pure honey.

Behavior of Fluids • Pressure is the force exerted on a surface divided by the total area over which the force is exerted. • • P= F/ A Force is measure in Newtons N. Area is measured in m 2 ( square meter) N/m 2 the unit is called Pascal ( Pa)

Pressure • Steam machine. Pumps water out of mines. ( Thomas Savery 17 th century )

Pressure • Crushing a can is applying pressure.

Pascal Principle • Atmospheric pressure at the sea level is 101. 3 KPa ( kilopascals). This means that air exerts a force about 101, 000 N ( Newtons).

Atmospheric Pressure • Changes with altitude. • Altitude is the height above the sea level. • As altitude increases, atmospheric pressure decreases.


Buoyant Force/Archimede’s Principle • The difference in pressure results in an upward force on an object immerse in a fluid. • The buoyant force is equal to the weight of the object.

Buoyancy • Sink or float

Archimedes’Principle • Density is mass divided by volume • D= m/v • Understanding density can help you predict whether an object will float or sink.

Pascal’s principle • When a force is applied to a confined fluid, an increase in pressure is transmitted equally to all parts of the fluid. • This relationship is known as Pascal’s Principle.