Lecture 12 Alcohols Thiols Ethers Aldehydes and Ketones












































- Slides: 48

Lecture 12: Alcohols, Thiols, Ethers, Aldehydes and Ketones Course lecturer : Assis. Prof. Dr. Altijana Hromic-Jahjefendic

�Book chapter: 12 �Pages: 398 -433

Introduction �Functional groups – organic compounds with oxygen and sulfur atoms �Anesthetics- Diethyl ether �Aldehydes as perfumes �Alcohols as beverages (drings) and desinfectants

Alcohols � In an alcohol, the functional group known as a hydroxyl group (¬OH) replaces a hydrogen atom in a hydrocarbon.

Naming alcohols �Replacing the e of the corresponding alkane with ol �Common name: alkyl group + alcohol �Example: methyl alcohol (methanol) �Cyclic alcohol – naming depends on substituents �The ring is numbered from C 1 which has OH group

Phenols �In a phenol, the hydroxyl group replaces a hydrogen atom attached to a benzene ring.

Naming phenols �Benzene ring bonded to a hydroxyl group �If there is a second substituent -the benzene ring is numbered starting from C 1 (carbon that has OH group)

Important alcohols �Methanol (methyl alcohol) – the simplest �Found in many solvents and paint removers �If ingested – oxidized to formaldehyde (headache, blindness and death) �Ethanol (ethyl alcohol) – fermentation of grains, sugars and starches �As solvent for perfumes, some medicines (tincture of iodine, propolis) �“Gasohol” – mixture of ethanol and gasoline (production of alternative fuels)

Important phenols �Essential oils of plants �Eugenol – in cloves �Vanillin – vanilla bean �Thymol – thyme and mint �Bispherol A (BPA) – making polycarbonate (manufacturing beverage bottles; also baby bottles) �Washing with certain detergents at high temperatures – disrupts polymer, small amounts of BPA leach from the bottle �Estrogen mimic – harmful effects �Banned – “BPA free”

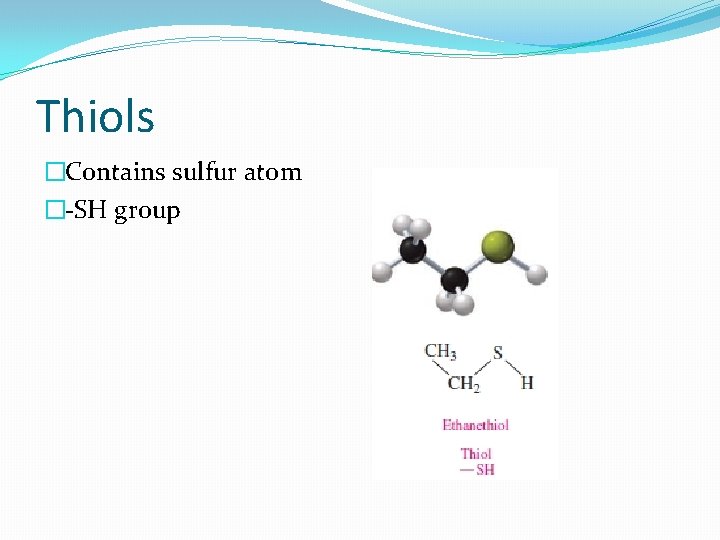
Thiols �Contains sulfur atom �-SH group

Naming thiols �Thiol to the alkane name of the longest carbon chain �Numbering the carbon chain from the end nearer the –SH group �Properties: �Strong, disagreeable odor �Used to detect natural gas leaks (ethanethiol) �Methanethiol – odor of cheddar cheese, onions and garlic

Ether �The functional group consists of an oxygen atom �Attached to 2 C atoms (-O-)

Naming ethers �Most of them have common names �Each alkyl or aromatic group attached to the oxygen atom is written in alphabetical order + ether q. Example: dimethylether (dimethyl group + ether)

Chemistry Link to Health �Ethers as Anesthetics �Anesthesia – loss of sensation and consciousness �Diethyl ether was most widely used for more than a 100 years �Very volatile and highly flammable �Since 1950 s Forane (isofurane), Ethrane and Penthrane were developed – not as flammable �Addition of halogens reduces the flammability �Some of them highly toxic to liver and kidneys replaced

Properties of Alcohols �Classified by the number of alkyl groups attached to the C atom bonded to the OH group Ø Primary (1°) alcohol – one alkyl group Ø Secondary (2°) alcohol – two alkyl groups Ø Tertiary (3°) alcohol – three alkyl groups

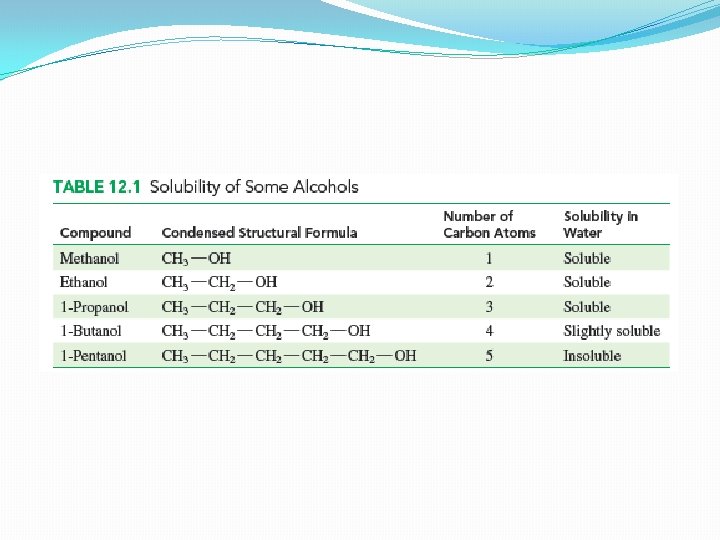
Solubility of Alcohols in water �Polar –OH group �Can form hydrogen bonds with the H and O atoms of water �Makes alcohols more soluble in water �Alcohols with one-three C atoms makes them more miscible in water – any amount of alcohol is completely soluble in water �Solubility decreases as the number of C atoms increases


Chemistry link to health: Hand sanitizers and Ethanol �Hand sanitizers – alternative to washing hands �Kills most bacteria and viruses that spread colds and flu �Use ethanol as active ingredient �Safe for most adults; supervision when used by children �The ethanol amount is 60 % (v/v) but can go up to 85 % �Importnat to rub the hands until they are completely dry – flammable �Keep them in storage away from heat sources

�Some hand sanitizers are ethanol-free �Active ingredient – triclosan �Aromathic, ether and phenol functional groups �Banning it – when mixed with tap water for disposal accumulates in the environment �Promotes the growth of antibiotic-resistant bacteria

Solubility of Phenols �Slightly soluble in water �Weak acids �Very corrosive and highly irritating to the skin �Causes severe burns �Ingestion can be fatal �In the past was used to disinfect wounds to prevent post-surgical infections �Replaced by other disinfectants and later by antibiotics

Summary �Alcohols and naming �Phenols and naming �Ethers and naming �Thiols and naming �Properties of alcohols �Hand sanitizers �Solubility of phenols

Aldehydes and Ketones �Aldehydes and ketones contain a carbonyl group that consists of a carbon– oxygen double bond with two groups of atoms attached to the carbon at angles of 120°. �The double bond is similar to that of alkenes; but carbonyl group has a dipole

�In an aldehyde – carbonyl group is bonded to at least one hydrogen atom �It can be bonded to another hydrogen atom, a carbon of an alkyl group or an aromatic ring

�In a ketone – carbonyl group is bonded to two alkyl groups or aromatic rings

Naming aldehydes �Replacing e of the corresponding alkane with al �No number is needed for the aldehyde group – appears at the end of the chain � 1 -4 C atoms – common names which ends in aldehyde

Naming ketons �Common anmes for unbranched ketones are still in use �The alkyl groups bonded to the carbonyl group are named as substituents �Listed alphabetically followed by ketone �In the IUPAC system – replacing the e in the corresponding alkane name with one q. Example: propanon (propan + one)

�In the cyclic ketone – the carbonyl group is numbered as carbon 1 �The ring is numbered in the direction to give substituents the lowest possible numbers

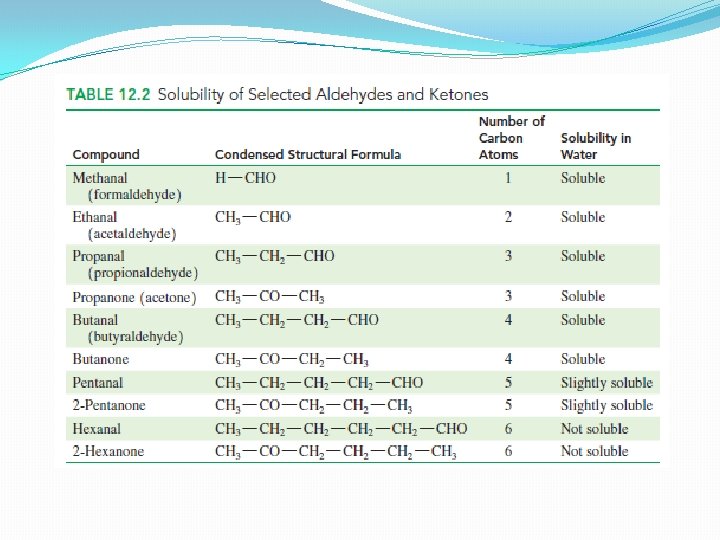
Properties of Aldehydes and Ketones in Water �Both contain polar carbonyl group �Partially negative oxygen atom and partially positive C atom �Electronegative oxygen forms hydrogen bondswith water molecules – both compounds with 1 -4 C atoms are very soluble � 5 and more C atoms – not soluble; longer hydrocarbon chains (nonpolar) diminish the solubility effect of the polar carbonyl group


Chemistry Link to Health: Some important aldehydes and ketones �Formaldehyde – colorless gas, pungent odor �An aqueous solution – formalin (germicide and to preserve biological specimens) �In industry – reactant in the synthesis of polymers (used to make fabrics, insulation materials, carpeting, pressed wood products etc. ) �Exposure to the gas – irritation of eyes, nose and upper respiratory tract, skin rashes, headache, dizziness

�Acetone – colorless liquid with mild odor �Wide use in cleaning fluids, paint, nail polish removers �Very flammable �In the body- produced in uncontrolled diabetes, fasting and high-protein diets �Muscone – used to make musk perfumes �Naturally occuring aromatic aldehydes – used to flavor food and as fragnances in perfumes �Benzaldehyde – almonds

Reactions of Alcohols, Thiols, Aledydes and Ketones �Alcohols – combustion in the presence of oxygen �Example: flamming dessert; prepared by pouring liquor on fruit or ice cream and lighten it

Dehydration of Alcohols �Lose water when they are heated with an acid as catalyst �A double bond forms between the same two C atoms – produces alkenes

Oxidation of alcohols �Addition of oxygen or a loss of hydrogen atoms �Increase in the number of C-O bonds �In the reduction reaction – the product has fewer bonds between carbon and oxygen

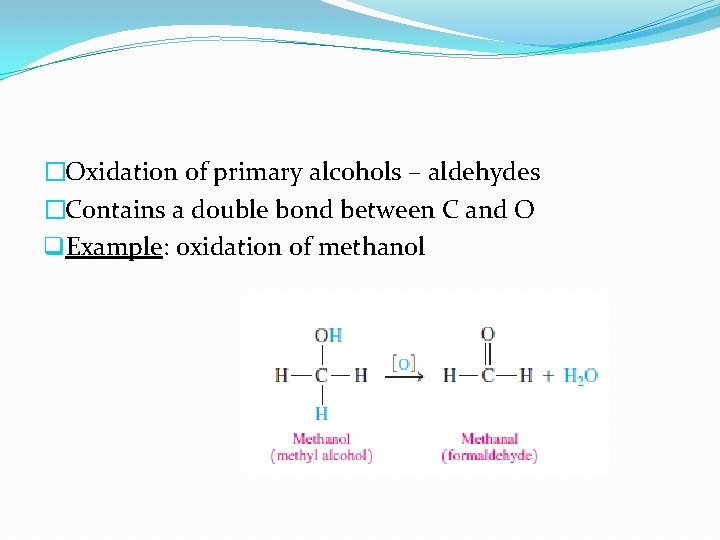
�Oxidation of primary alcohols – aldehydes �Contains a double bond between C and O q. Example: oxidation of methanol

�Oxidation of secondary alcohols – ketones �C-O double bond attached to alkyl groups on both sides �No further oxidation of ketones – no hydrogen atoms attached to the carbonyl group

�Tertiary alcohols do not oxidize! �There is no hydrogen atom on the carbon bonded to the –OH group �C-C bonds are too strong to oxidize

Chemistry Link to Health: Methanol Poisoning �Methanol (methyl alcohol) – highly toxic �Present in products such as winshield washer fluid, paint strippers �Rapidly absorbed in the gastrointestinal tract �In the liver – oxidized to formaldehyde and formic acid �Causes severe abdominal pain and blurred vision �Blindness – intermediate products destroy the retina �Formic acid – lowers blood p. H so severly; 30 m. L of Me. OH can lead to coma and death

�Treatment: Ø Sodium bicarbonate – neutralize the formic acid in the blood Ø Ethanol – intravenously �Enzymes in the liver pick up ethanol molecules to oxidize it instead of methanol �Gives time for the methanol to be eliminated via the lungs �Without formation of its dangerous oxidation products

Oxidation of Thiols �Loss of hydrogen atoms from each of two –SH groups �Oxidized product- disulfide bond –S-S�Proteins in hair – cross-linked by disulfide bonds �Occur mostly between side chains of cysteins �Reducing substance is used to break disulfide bonds – permanent curling or straightening �When hair is wrapped – oxidizing substance is applied to cause new disulfide bonds to form between different parts of the protein hair strands – forming a new shape

Oxidation of aldehydes �Adding O to aldehydes – to form a carboxylic acid �Has a carboxyl functional group �This step occurs readily – often difficult to isolate the aldehyde product during the oxidation reaction �In contrast, ketones do not undergo further oxidation

Chemistry Link to Health: Oxidation of Alcohol in the Body �Ethanol – most commonly abused drug in the USA �Ingestion of small amounts – feeling of euphoria despite the fact that it is a depressant �In the liver – enzymes (alcohol dehydrogenase) oxidize Et. OH to acetaldehyde �Impairs mental and physical coordination �Concentration abowe 0. 4 % - coma or death

�The acetaldehyde – further oxidized to acetic acid �Converted to CO 2 and water in the citric acid cycle �Enzymes in the liver – can break down ethanol �However, aldehyde and carboxylic acid intermediates can cause considerable damage within the cells of the liver �Rate of Et. OH metabolism varies between drinkers and nondrinkers �Some effects of alcohol metabolism include an increase in liver lipids, gastritis, alcoholic hepatitis and psychological disturbances

�Alcohol in the blood evaporates through the lungs �Percentage of it in the lungs – used to calculate the blood alcohol concentration (BAC) �Police devices �Driver exhales into a solution containing the orange Cr 6+ ion �Alcohol reduces the orange Cr 6+ to a green Cr 3+

�Treatment: �Antabuse (disulfiram) �Prevents oxidation of acetaldehyde to acetic acid �Result: acetaldehyde accumulates in the blood �Causes nausea, sweating, headache, dizziness and respiratory difficulties �Because of side effects – the person is less likely to use alcohol

Tollens’ Test �Uses solution of Ag+ (Ag. NO 3) and ammonia �Oxidizes aldehydes but not ketones �The silver ion is reduced and forms a “silver mirror” �Comertially, similar process is used to make mirrors by applying a solution of Ag. NO 3 and ammonia on a glass (spray gun)

Reduction of Aldehydes and Ketones �Reduced by sodium borohydride (Na. BH 4) or hydrogen (H 2) �Decrease in the number of C-O bonds by the addition of hydrogen or the loss of oxygen �Aldehydes reduce to primary alcohols �Ketones reduce to secondary alcohols �Catalyst: Ni, Pt, or palladium is needed

Summary �Aldehydes and naming �Ketones and naming �Properties in water �Aldehydes and ketones in everyday life �Reactions of alcohols, thiols, aldehydes and ketones �Methanol poisoning �Oxidation of alcohol in the body �Tollens Test �Reduction of aldehydes and ketones