CH 19 Aldehydes and Ketones Renee Y Becker

CH 19: Aldehydes and Ketones Renee Y. Becker Valencia Community College CHM 2211 1
Some Generalizations About Carbonyl Compounds • The most important functional group in organic chemistry. 2

Some Generalizations About Carbonyl Compounds • carbonyl compounds are planar about the double bond with bond angles 120 due to the sp 2 hybridized carbon. • Many types of carbonyl compounds have significant dipole moments. • The polarity of the C-O bond plays a significant role in the reactivity of carbonyl compounds. 3
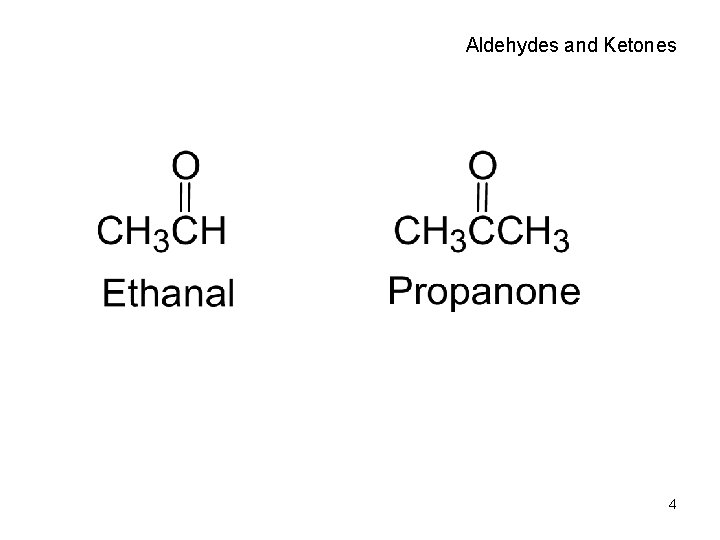
Aldehydes and Ketones 4
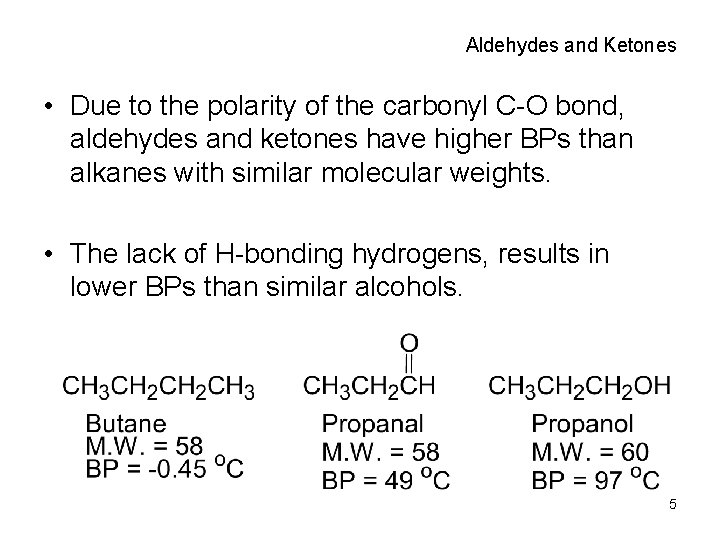
Aldehydes and Ketones • Due to the polarity of the carbonyl C-O bond, aldehydes and ketones have higher BPs than alkanes with similar molecular weights. • The lack of H-bonding hydrogens, results in lower BPs than similar alcohols. 5
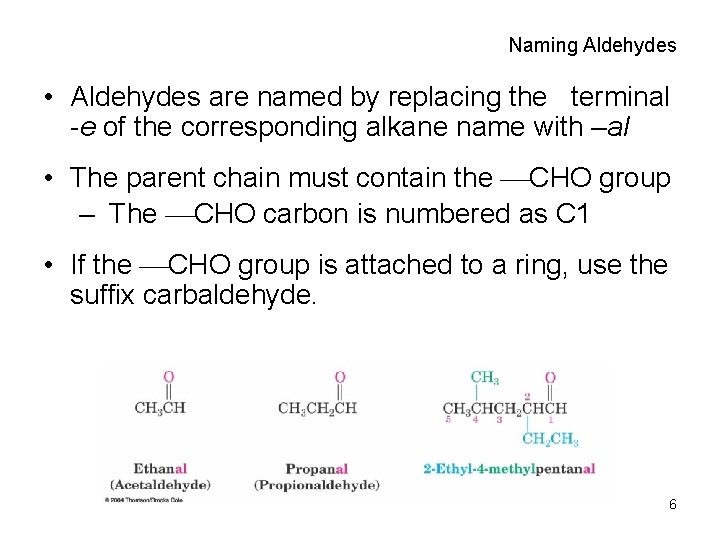
Naming Aldehydes • Aldehydes are named by replacing the terminal -e of the corresponding alkane name with –al • The parent chain must contain the CHO group – The CHO carbon is numbered as C 1 • If the CHO group is attached to a ring, use the suffix carbaldehyde. 6
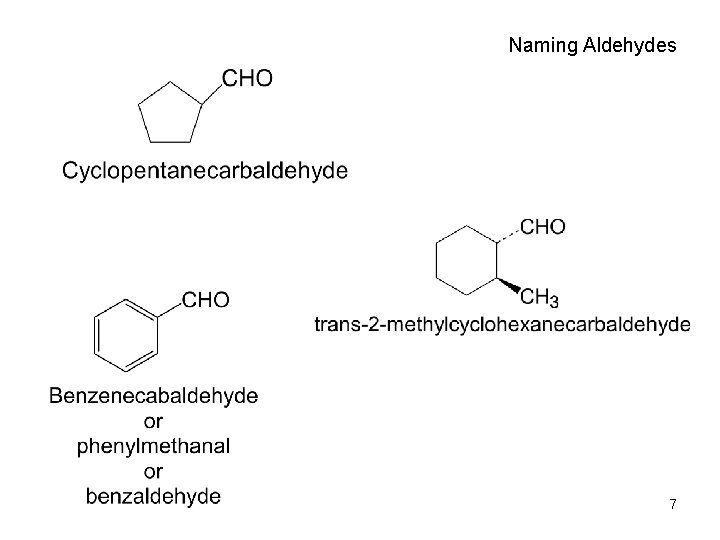
Naming Aldehydes 7
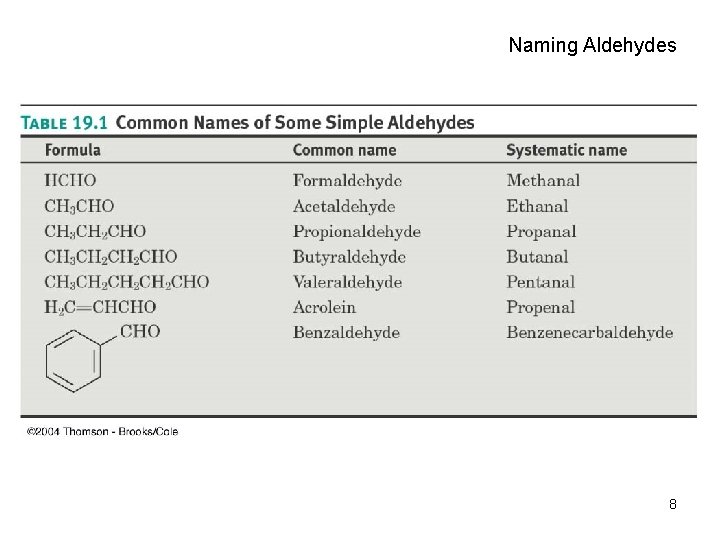
Naming Aldehydes 8
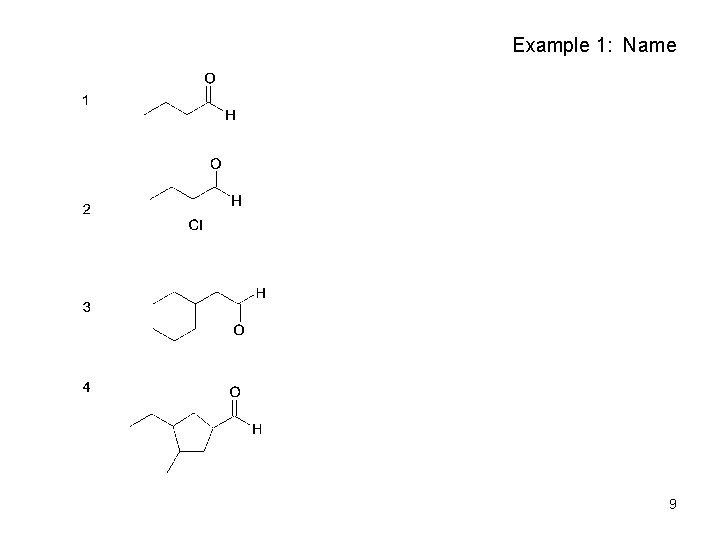
Example 1: Name 9

Example 2: Draw 1. 3 -Methylbutanal 2. 3 -Methyl-3 -butenal 3. cis-3 -tert-Butylcyclohexanecarbaldehyde 10
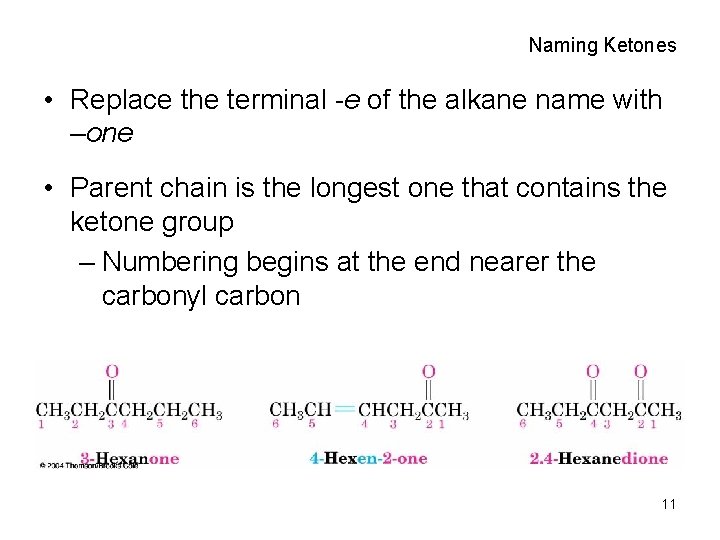
Naming Ketones • Replace the terminal -e of the alkane name with –one • Parent chain is the longest one that contains the ketone group – Numbering begins at the end nearer the carbonyl carbon 11
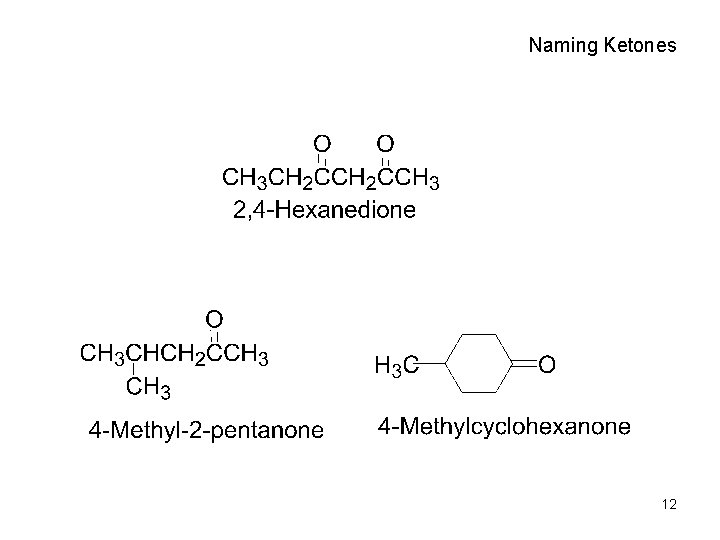
Naming Ketones 12
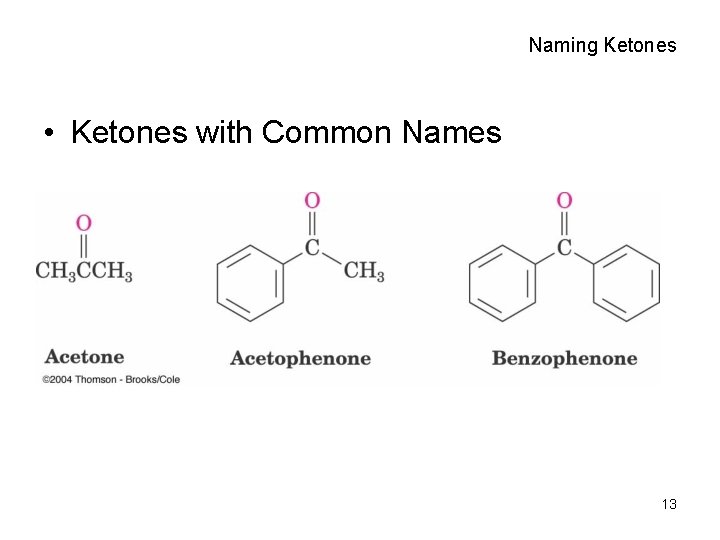
Naming Ketones • Ketones with Common Names 13
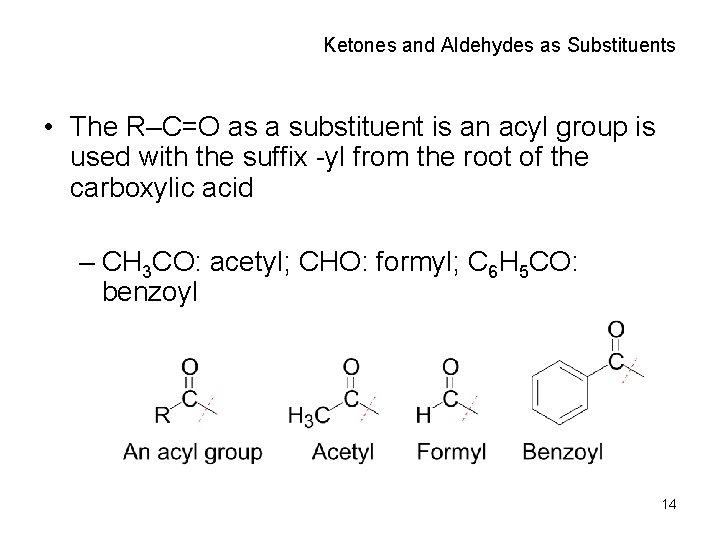
Ketones and Aldehydes as Substituents • The R–C=O as a substituent is an acyl group is used with the suffix -yl from the root of the carboxylic acid – CH 3 CO: acetyl; CHO: formyl; C 6 H 5 CO: benzoyl 14

Ketones and Aldehydes as Substituents • The prefix oxo- is used if other functional groups are present and the doubly bonded oxygen is labeled as a substituent on a parent chain 15
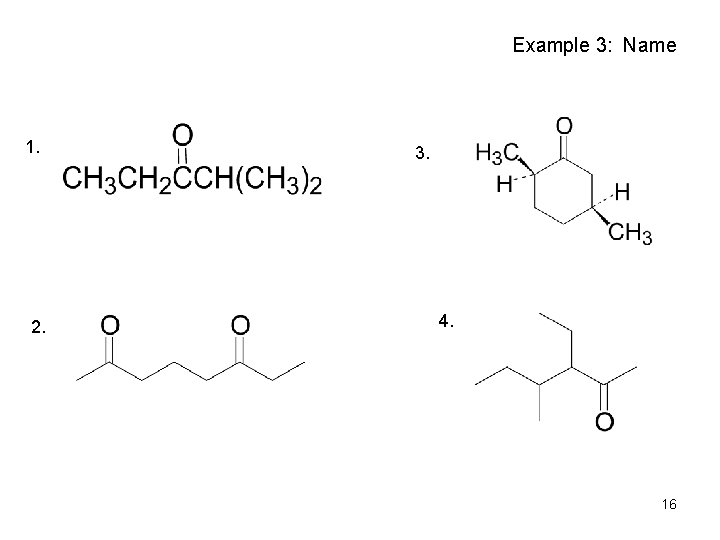
Example 3: Name 1. 2. 3. 4. 16
Example 4: Draw 1. 4 -Chloro-2 -pentanone 2. P-bromoacetophenone 3. 3 -ethyl-4 -methyl-2 -hexanone 17
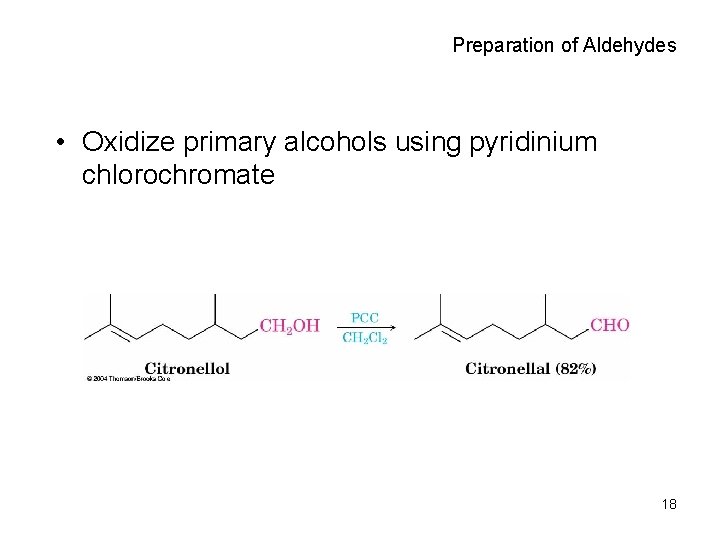
Preparation of Aldehydes • Oxidize primary alcohols using pyridinium chlorochromate 18
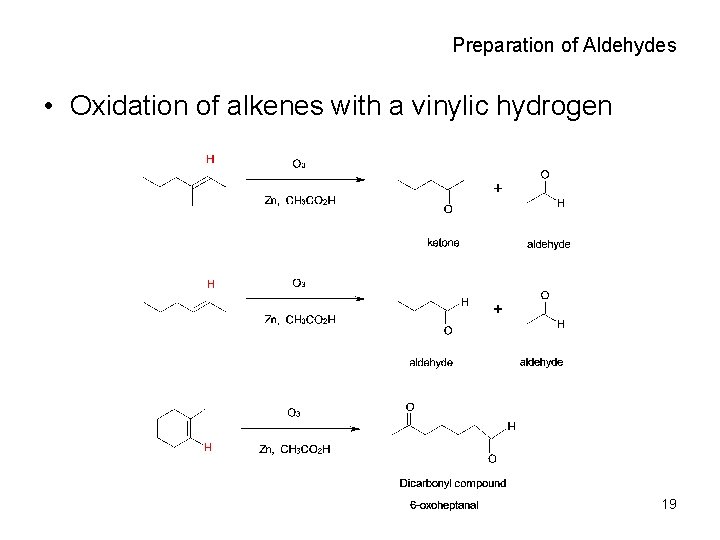
Preparation of Aldehydes • Oxidation of alkenes with a vinylic hydrogen 19
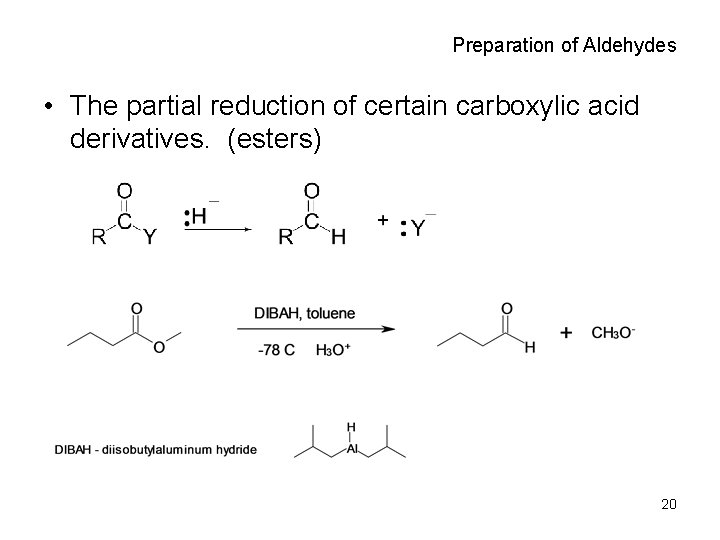
Preparation of Aldehydes • The partial reduction of certain carboxylic acid derivatives. (esters) 20
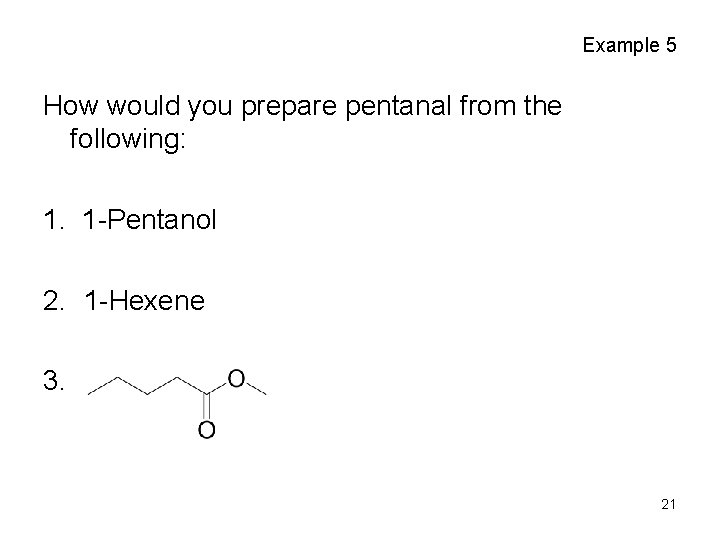
Example 5 How would you prepare pentanal from the following: 1. 1 -Pentanol 2. 1 -Hexene 3. 21
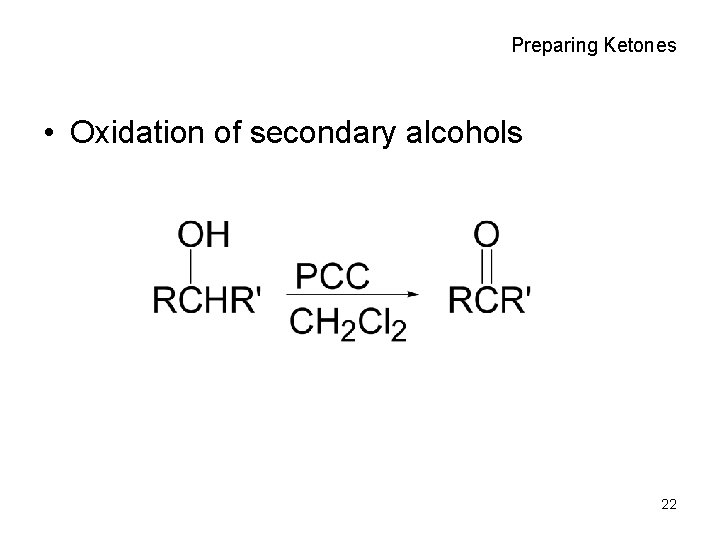
Preparing Ketones • Oxidation of secondary alcohols 22
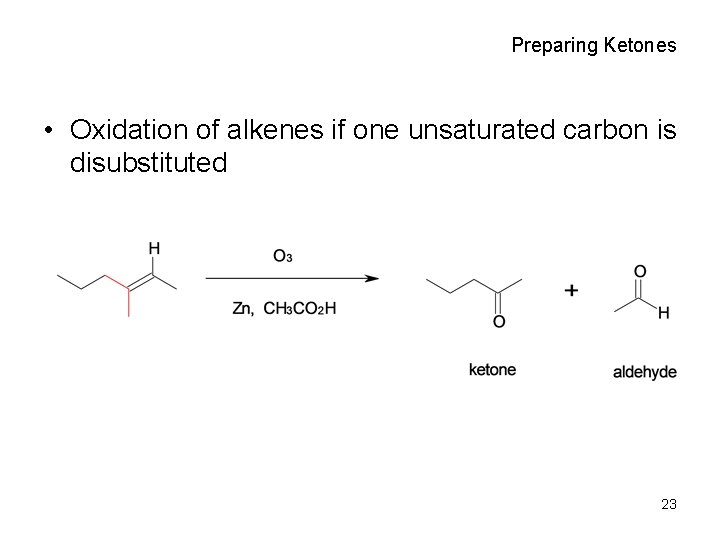
Preparing Ketones • Oxidation of alkenes if one unsaturated carbon is disubstituted 23
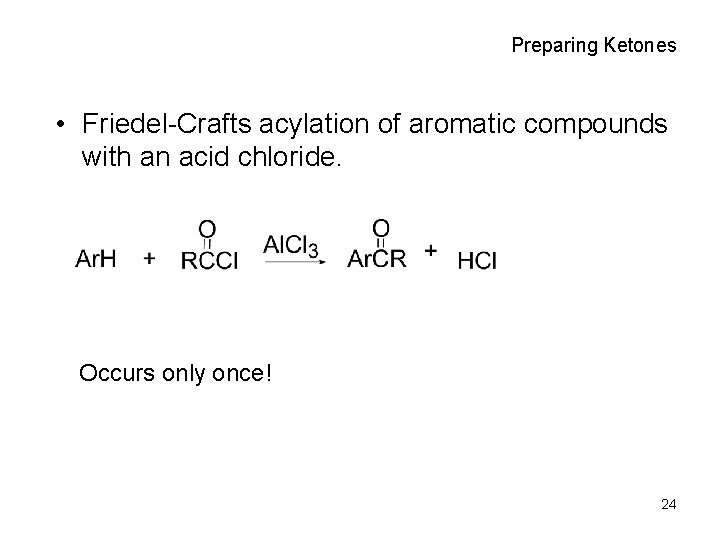
Preparing Ketones • Friedel-Crafts acylation of aromatic compounds with an acid chloride. Occurs only once! 24
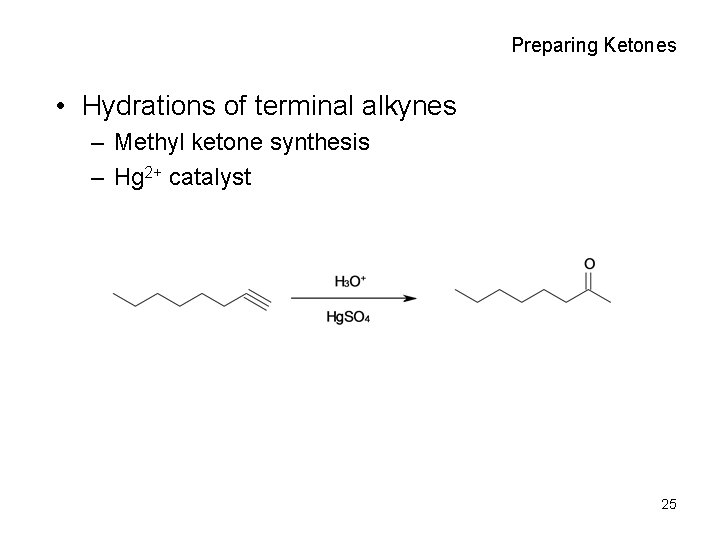
Preparing Ketones • Hydrations of terminal alkynes – Methyl ketone synthesis – Hg 2+ catalyst 25

Example 6 How would you carry out the following reactions? More than 1 step might be necessary. 1. 3 -Hexyne 3 -Hexanone 2. Benzene m-Bromoacetophenone 3. Bromobenzene Acetophenone 26

Reactions of Aldehydes and Ketones • Oxidation reactions • Nucleophilic addition reactions • Conjugate nucleophilic addition reactions 27
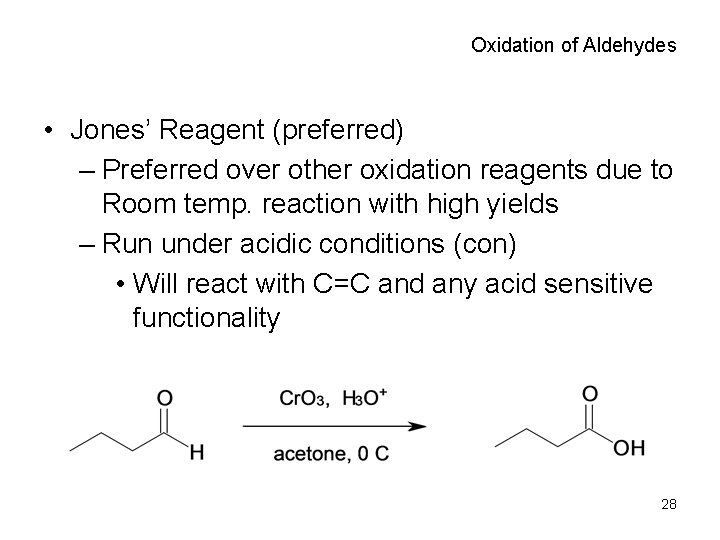
Oxidation of Aldehydes • Jones’ Reagent (preferred) – Preferred over other oxidation reagents due to Room temp. reaction with high yields – Run under acidic conditions (con) • Will react with C=C and any acid sensitive functionality 28
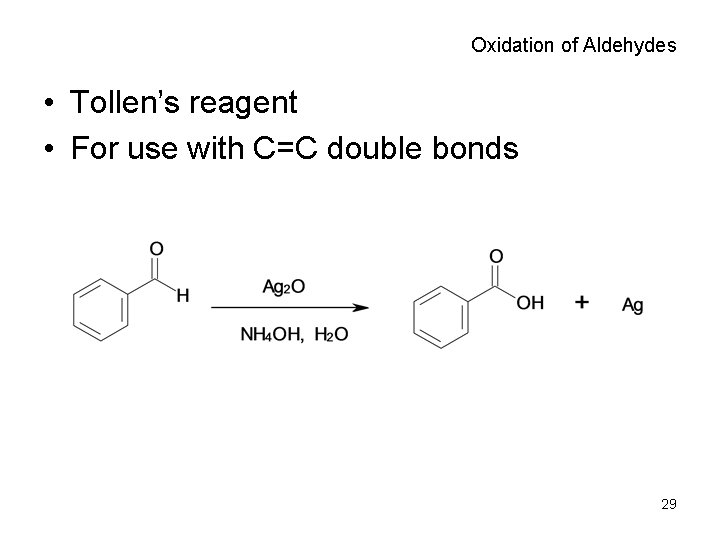
Oxidation of Aldehydes • Tollen’s reagent • For use with C=C double bonds 29
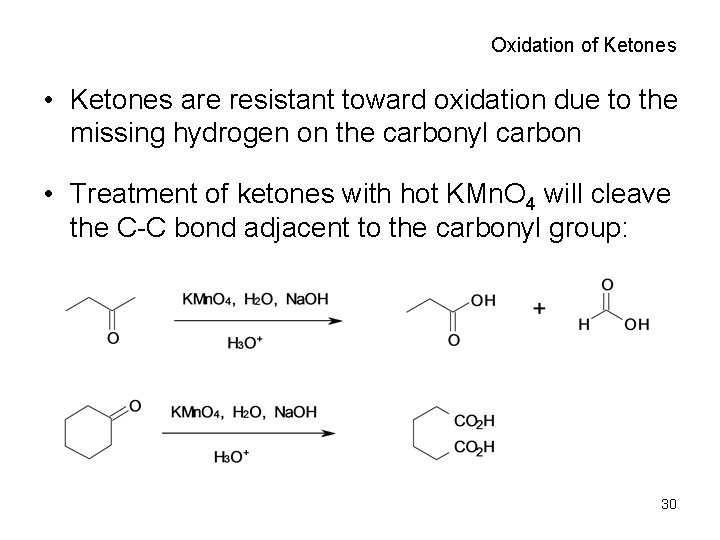
Oxidation of Ketones • Ketones are resistant toward oxidation due to the missing hydrogen on the carbonyl carbon • Treatment of ketones with hot KMn. O 4 will cleave the C-C bond adjacent to the carbonyl group: 30
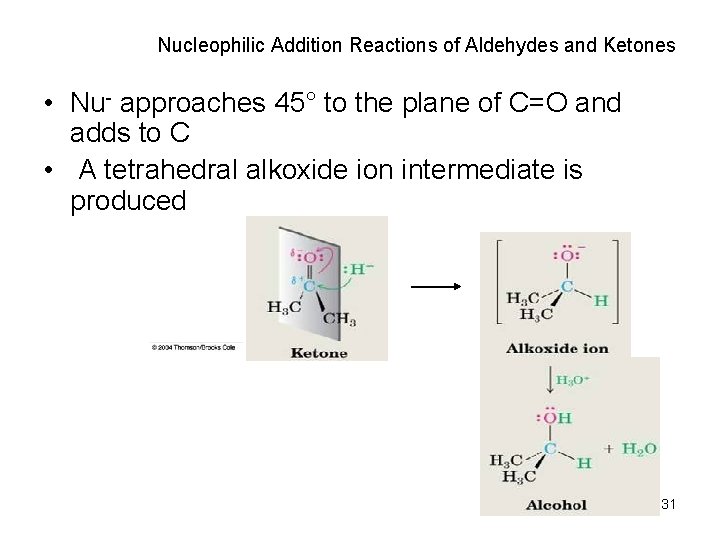
Nucleophilic Addition Reactions of Aldehydes and Ketones • Nu- approaches 45° to the plane of C=O and adds to C • A tetrahedral alkoxide ion intermediate is produced 31
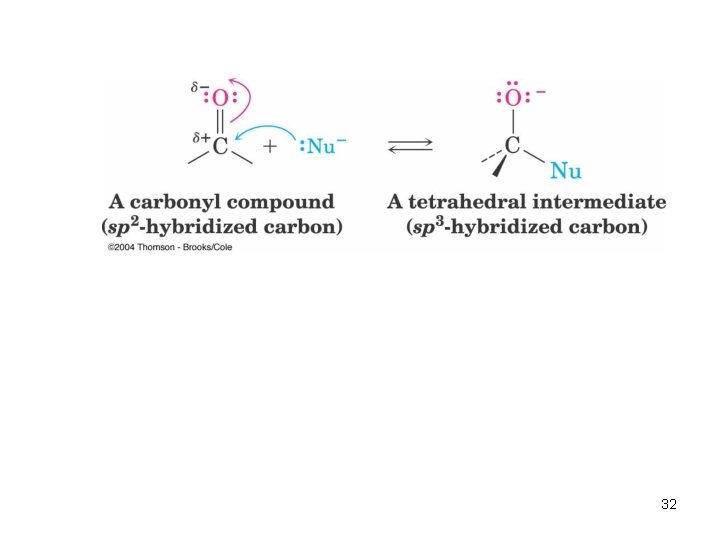
32

Nucleophiles • Nucleophiles can be negatively charged ( : Nu ) or neutral ( : Nu) at the reaction site • The overall charge on the nucleophilic species is not considered 33
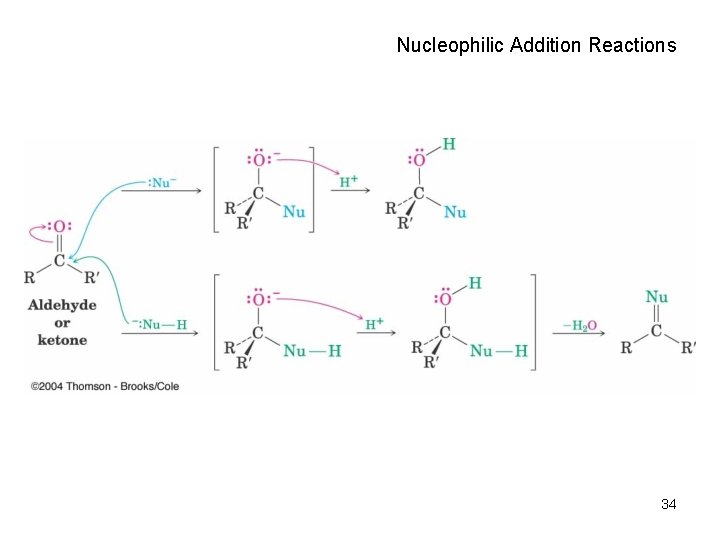
Nucleophilic Addition Reactions 34
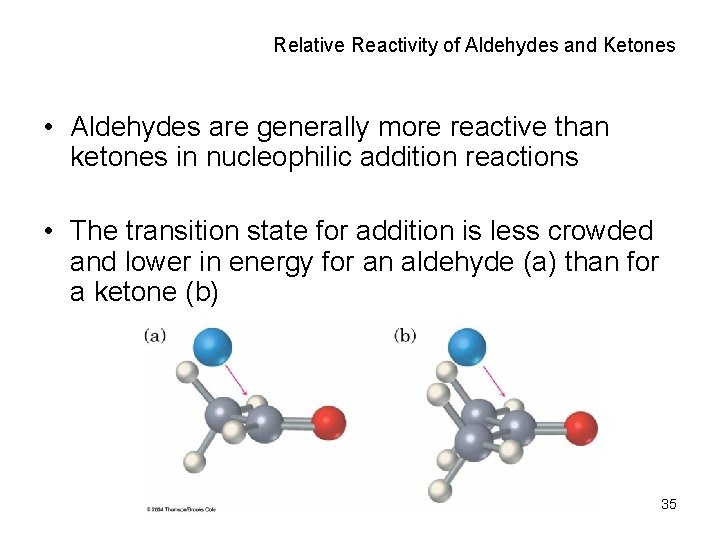
Relative Reactivity of Aldehydes and Ketones • Aldehydes are generally more reactive than ketones in nucleophilic addition reactions • The transition state for addition is less crowded and lower in energy for an aldehyde (a) than for a ketone (b) 35
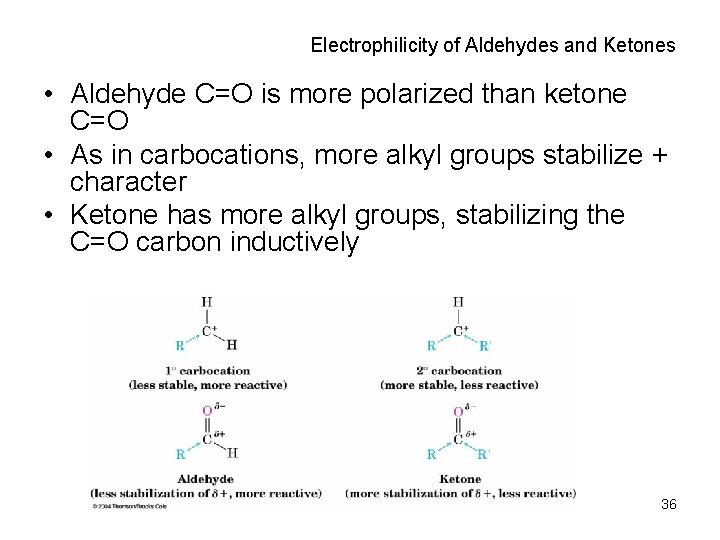
Electrophilicity of Aldehydes and Ketones • Aldehyde C=O is more polarized than ketone C=O • As in carbocations, more alkyl groups stabilize + character • Ketone has more alkyl groups, stabilizing the C=O carbon inductively 36
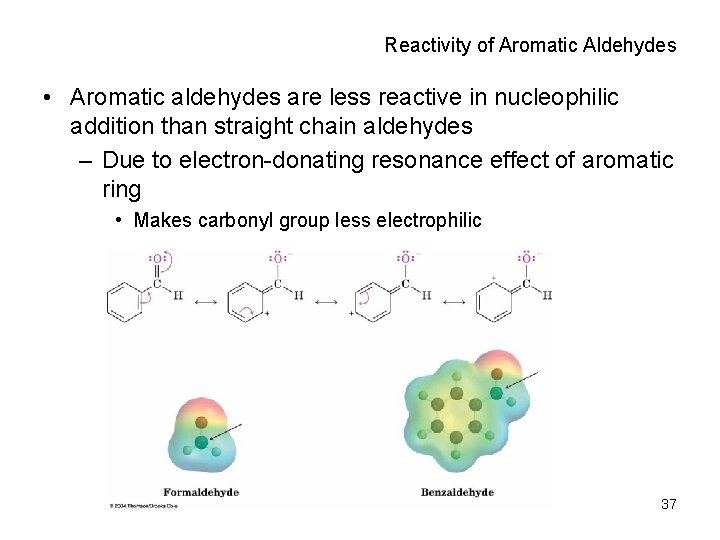
Reactivity of Aromatic Aldehydes • Aromatic aldehydes are less reactive in nucleophilic addition than straight chain aldehydes – Due to electron-donating resonance effect of aromatic ring • Makes carbonyl group less electrophilic 37
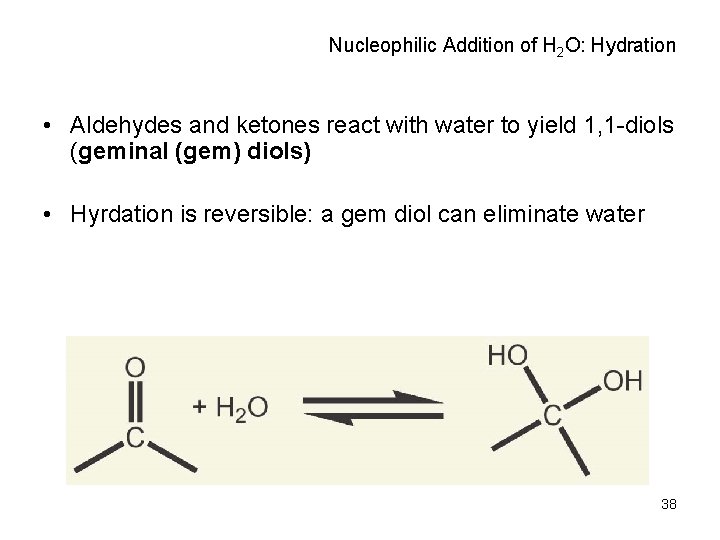
Nucleophilic Addition of H 2 O: Hydration • Aldehydes and ketones react with water to yield 1, 1 -diols (geminal (gem) diols) • Hyrdation is reversible: a gem diol can eliminate water 38
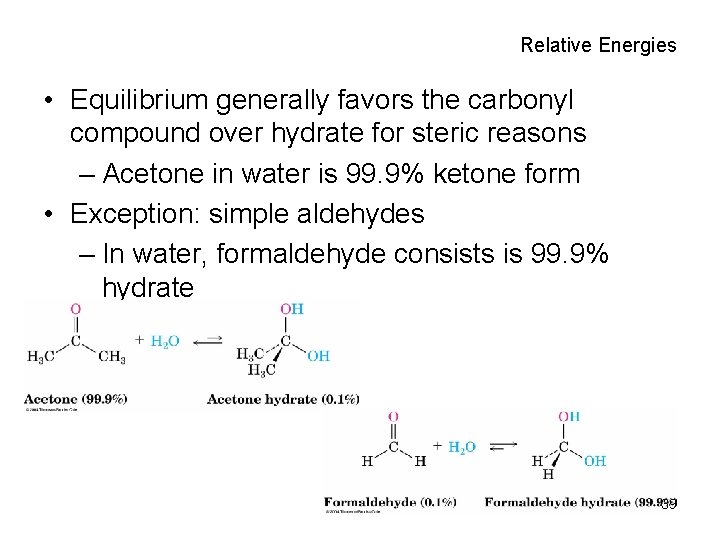
Relative Energies • Equilibrium generally favors the carbonyl compound over hydrate for steric reasons – Acetone in water is 99. 9% ketone form • Exception: simple aldehydes – In water, formaldehyde consists is 99. 9% hydrate 39

Acid & Base-Catalyzed Addition of Water • Addition of water is catalyzed by both acid and base • The base-catalyzed hydration nucleophile is the hydroxide ion, which is a much stronger nucleophile than water • Acid-Catalyzed Addition of Water • Protonation of C=O makes it more electrophilic 40
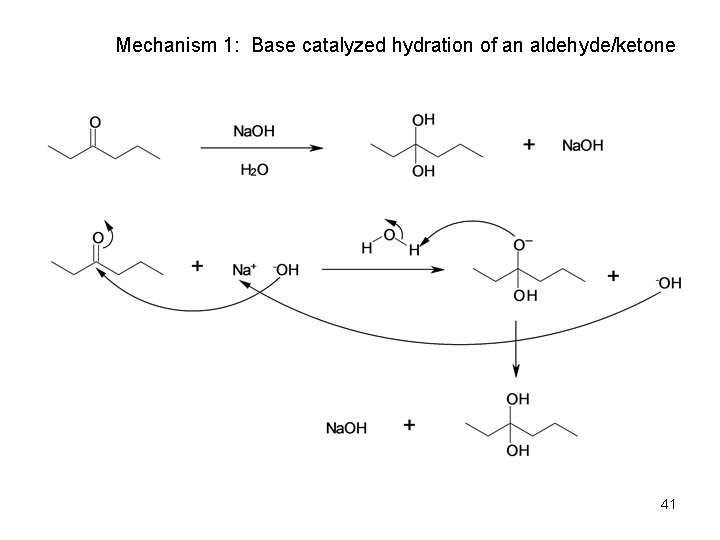
Mechanism 1: Base catalyzed hydration of an aldehyde/ketone 41
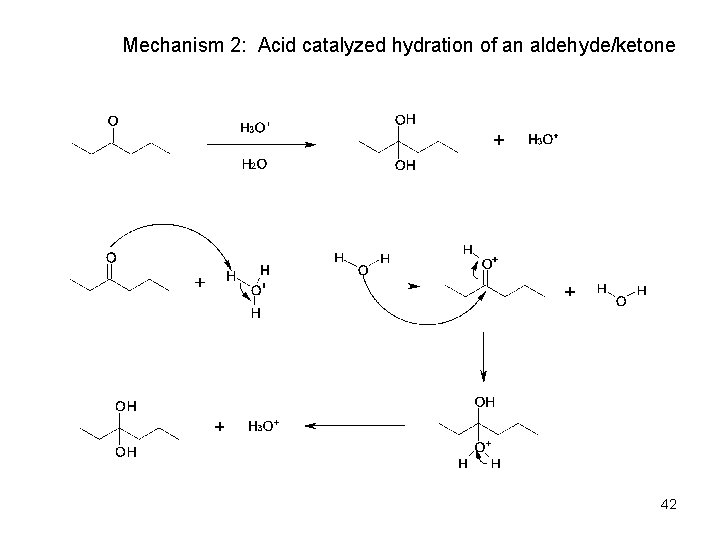
Mechanism 2: Acid catalyzed hydration of an aldehyde/ketone 42
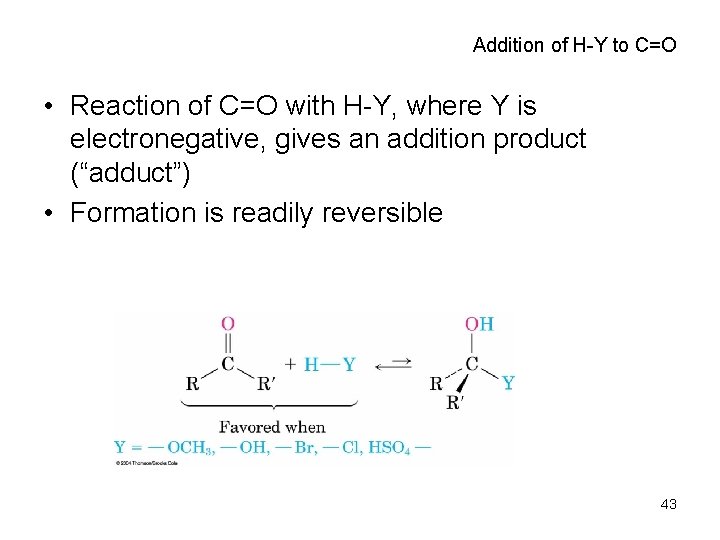
Addition of H-Y to C=O • Reaction of C=O with H-Y, where Y is electronegative, gives an addition product (“adduct”) • Formation is readily reversible 43
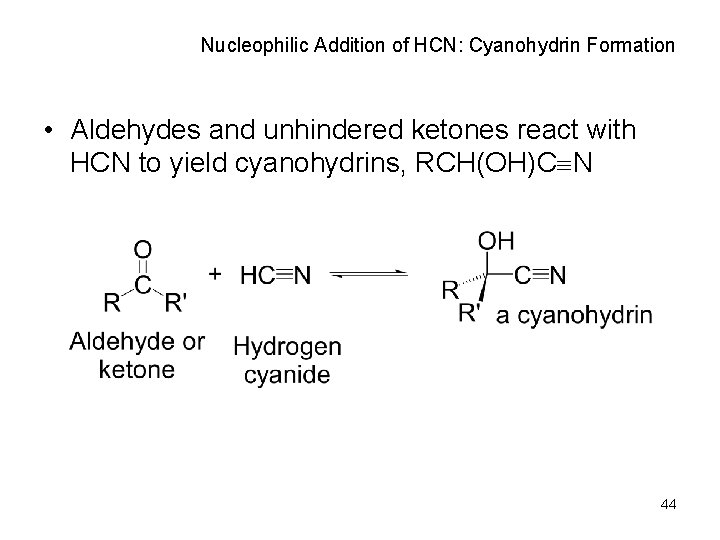
Nucleophilic Addition of HCN: Cyanohydrin Formation • Aldehydes and unhindered ketones react with HCN to yield cyanohydrins, RCH(OH)C N 44

Mechanism of Formation of Cyanohydrins • Addition of HCN is reversible and basecatalyzed, generating nucleophilic cyanide ion, CN • Addition of CN to C=O yields a tetrahedral intermediate, which is then protonated • Equilibrium favors adduct 45
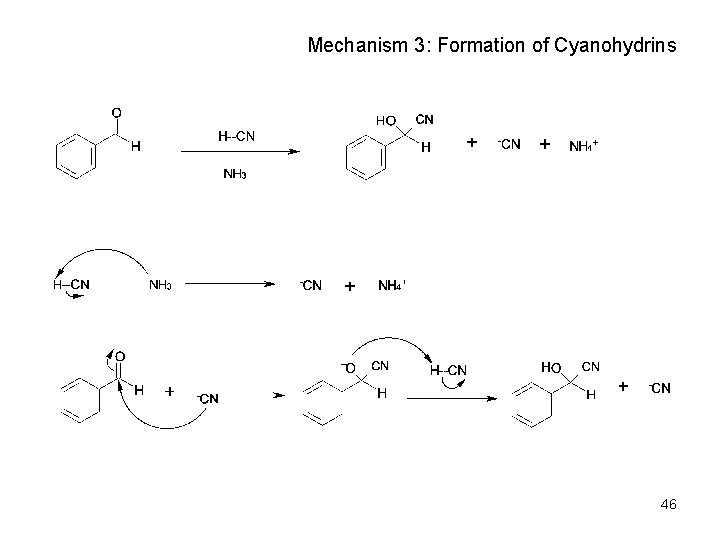
Mechanism 3: Formation of Cyanohydrins 46
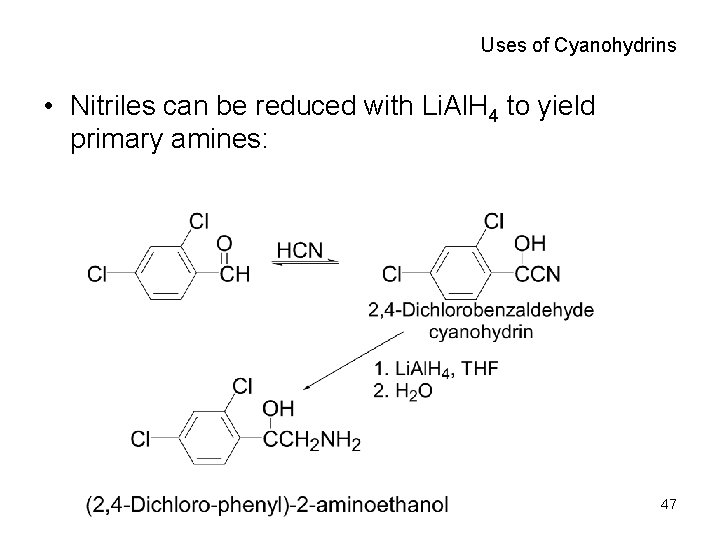
Uses of Cyanohydrins • Nitriles can be reduced with Li. Al. H 4 to yield primary amines: 47
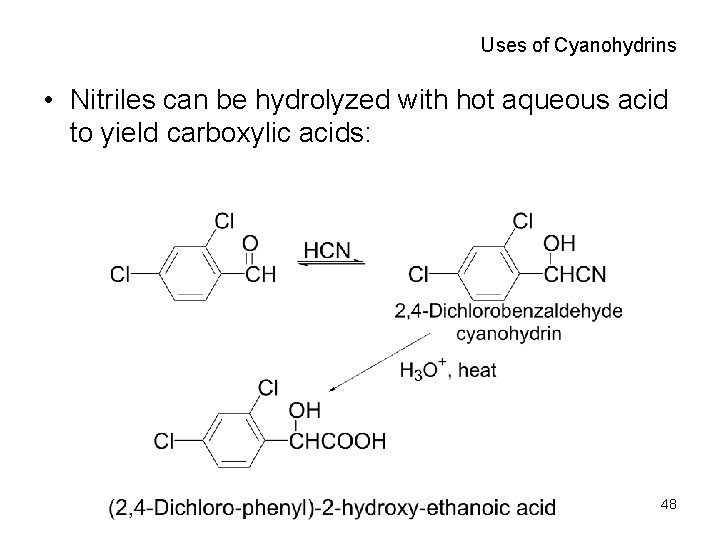
Uses of Cyanohydrins • Nitriles can be hydrolyzed with hot aqueous acid to yield carboxylic acids: 48

Nucleophilic Addition of Grignard Reagents and Hydride Reagents: Alcohol Formation • Treatment of aldehydes or ketones with Grignard reagents yields an alcohol – Nucleophilic addition of the equivalent of a carbon anion, or carbanion. A carbon–magnesium bond is strongly polarized, so a Grignard reagent reacts for all practical purposes as R : Mg. X +. 49

Mechanism of Addition of Grignard Reagents • Complexation of C=O by Mg 2+, Nucleophilic addition of R : , protonation by dilute acid yields the neutral alcohol • Grignard additions are irreversible because a carbanion is not a leaving group 50
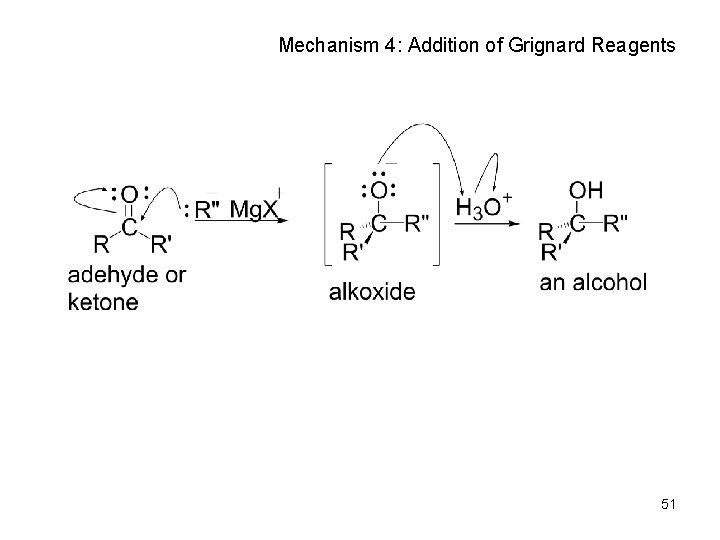
Mechanism 4: Addition of Grignard Reagents 51
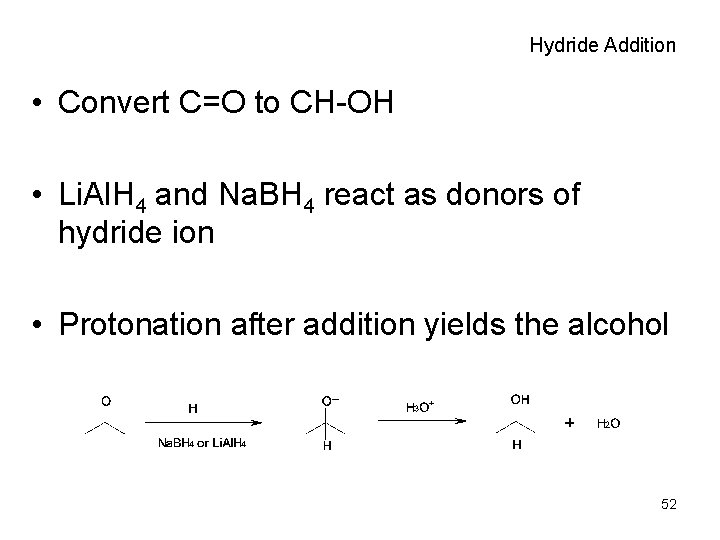
Hydride Addition • Convert C=O to CH-OH • Li. Al. H 4 and Na. BH 4 react as donors of hydride ion • Protonation after addition yields the alcohol 52

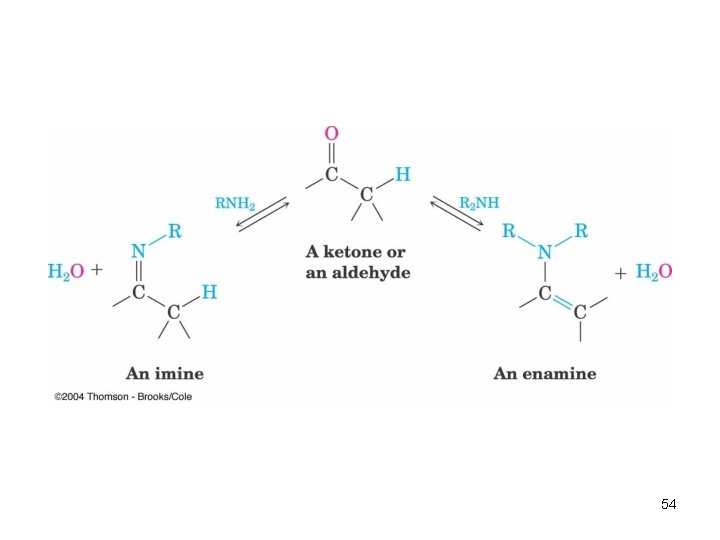
Nucleophilic Addition of Amines: Imine and Enamine Formation RNH 2 (primary amines) adds to C=O to form imines, R 2 C=NR (after loss of HOH) R 2 NH (secondary amines) yields enamines, R 2 N CR=CR 2 (after loss of HOH) (ene + amine = unsaturated amine) 53
54

Mechanism of Formation of Imines • Primary amine adds to C=O • Proton is lost from N and adds to O to yield a neutral amino alcohol (carbinolamine) • Protonation of OH converts into water as the leaving group • Result is iminium ion, which loses proton • Acid is required for loss of OH – too much acid blocks RNH 2 Note that overall reaction is substitution of RN for O 55

Mechanism 5: Imine Formation 56

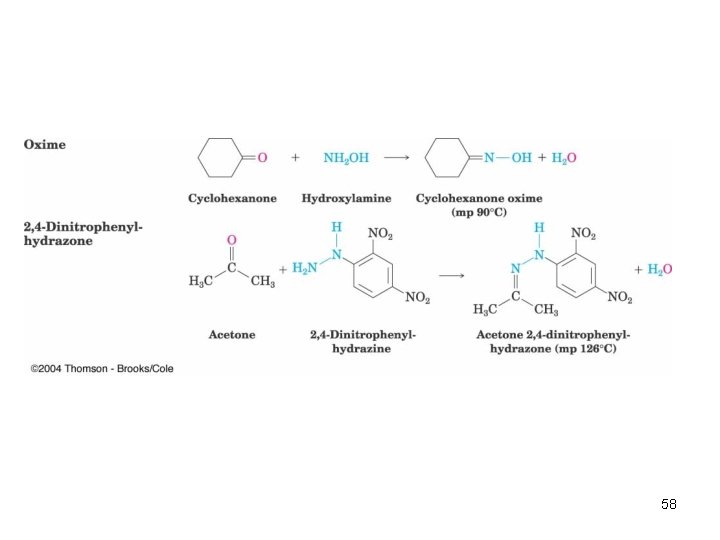
Imine Derivatives • Addition of amines with an atom containing a lone pair of electrons on the adjacent atom occurs very readily, giving useful, stable imines • For example, hydroxylamine forms oximes and 2, 4 -dinitrophenylhydrazine readily forms 2, 4 dinitrophenylhydrazones – These are usually solids and help in characterizing liquid ketones or aldehydes by melting points 57
58
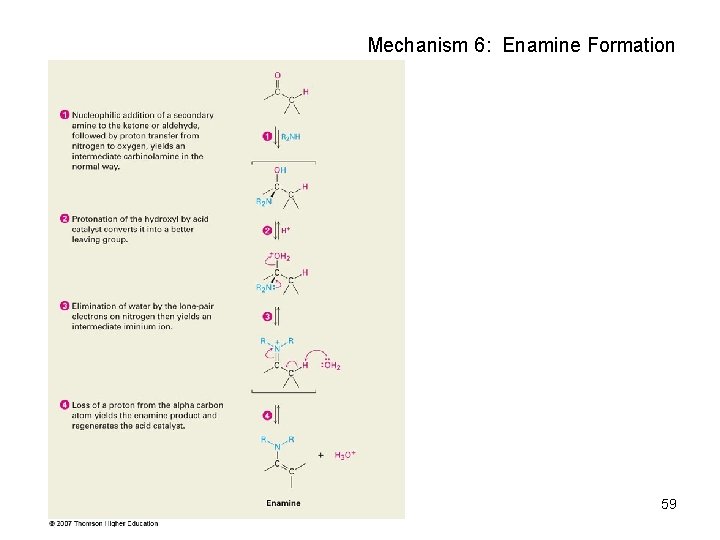
Mechanism 6: Enamine Formation 59

Nucleophilic Addition of Hydrazine: The Wolff–Kishner Reaction • Treatment of an aldehyde or ketone with hydrazine, H 2 NNH 2 and KOH converts the compound to an alkane • Originally carried out at high temperatures but with dimethyl sulfoxide as solvent takes place near room temperature 60

Mechanism 7: The Wolff–Kishner Reaction 61
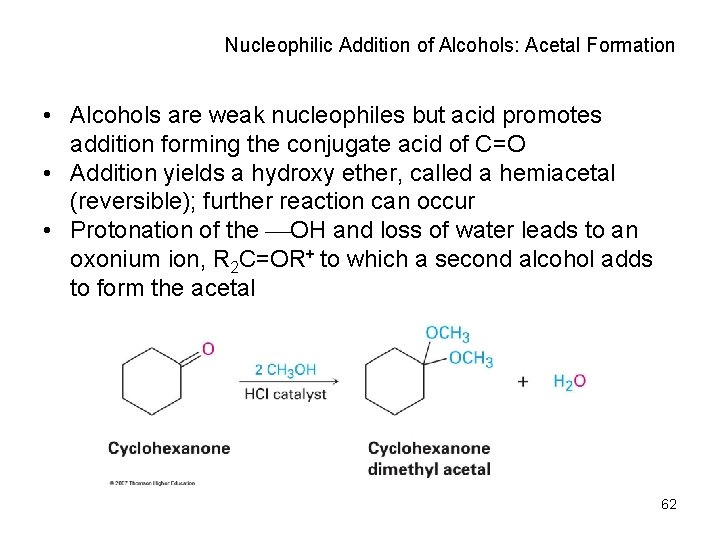
Nucleophilic Addition of Alcohols: Acetal Formation • Alcohols are weak nucleophiles but acid promotes addition forming the conjugate acid of C=O • Addition yields a hydroxy ether, called a hemiacetal (reversible); further reaction can occur • Protonation of the OH and loss of water leads to an oxonium ion, R 2 C=OR+ to which a second alcohol adds to form the acetal 62
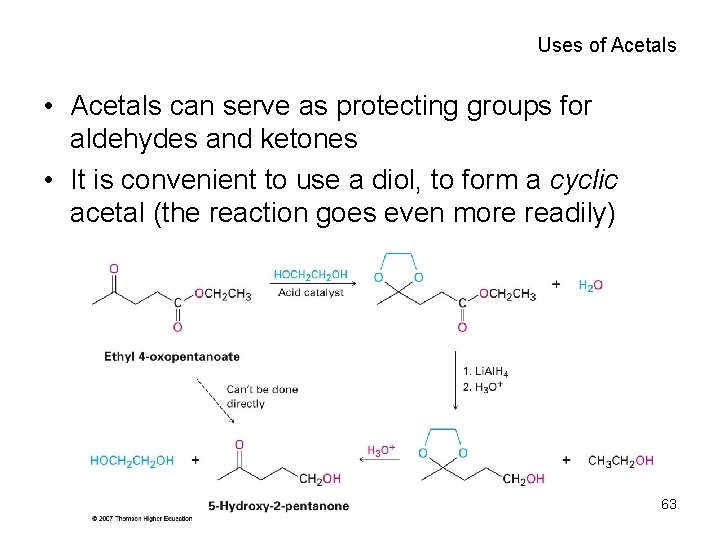
Uses of Acetals • Acetals can serve as protecting groups for aldehydes and ketones • It is convenient to use a diol, to form a cyclic acetal (the reaction goes even more readily) 63
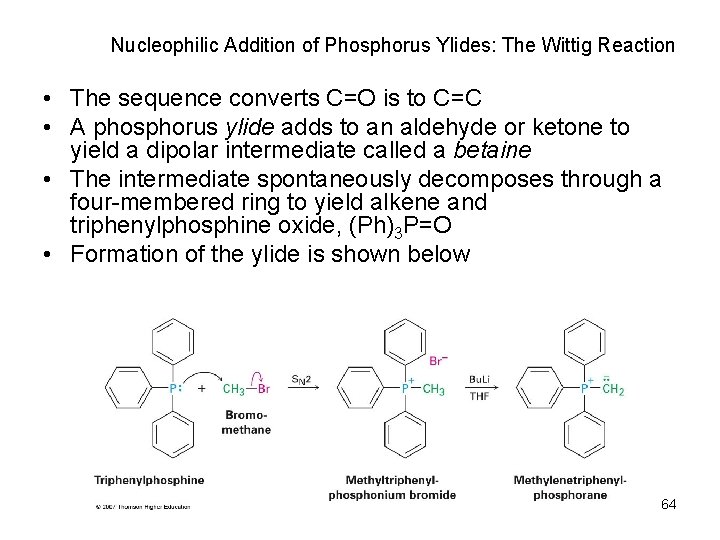
Nucleophilic Addition of Phosphorus Ylides: The Wittig Reaction • The sequence converts C=O is to C=C • A phosphorus ylide adds to an aldehyde or ketone to yield a dipolar intermediate called a betaine • The intermediate spontaneously decomposes through a four-membered ring to yield alkene and triphenylphosphine oxide, (Ph)3 P=O • Formation of the ylide is shown below 64
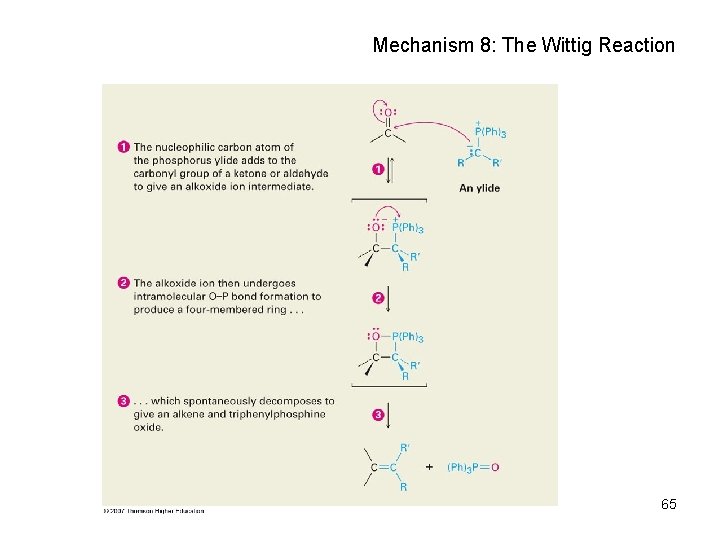
Mechanism 8: The Wittig Reaction 65
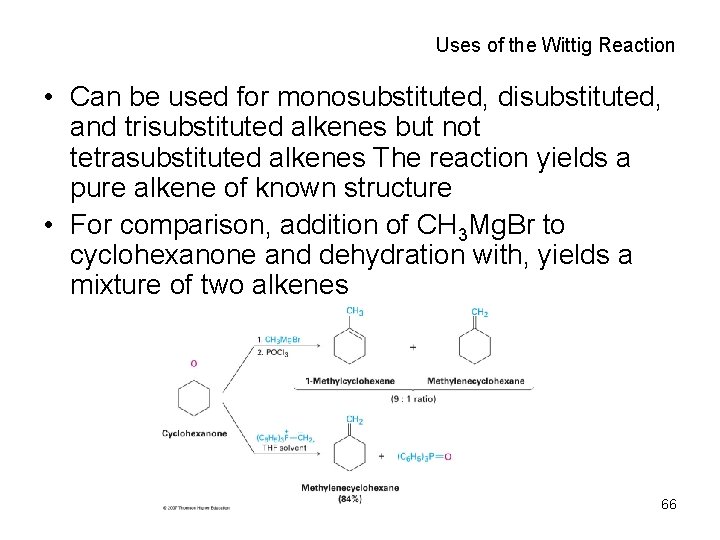
Uses of the Wittig Reaction • Can be used for monosubstituted, disubstituted, and trisubstituted alkenes but not tetrasubstituted alkenes The reaction yields a pure alkene of known structure • For comparison, addition of CH 3 Mg. Br to cyclohexanone and dehydration with, yields a mixture of two alkenes 66
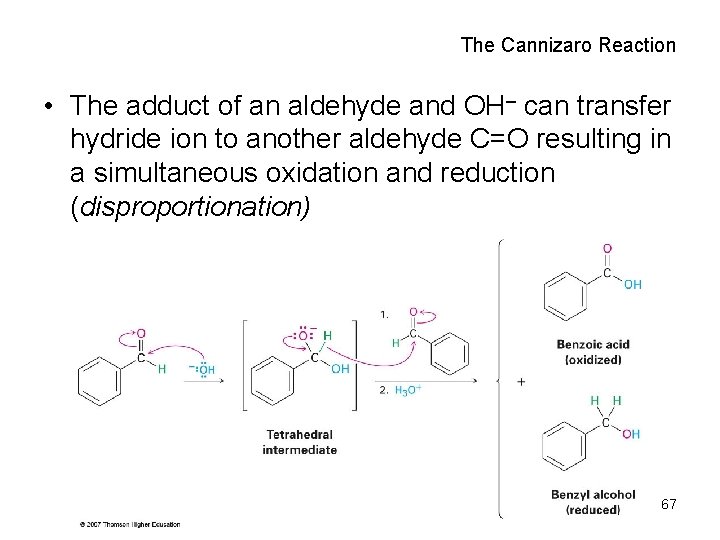
The Cannizaro Reaction • The adduct of an aldehyde and OH can transfer hydride ion to another aldehyde C=O resulting in a simultaneous oxidation and reduction (disproportionation) 67

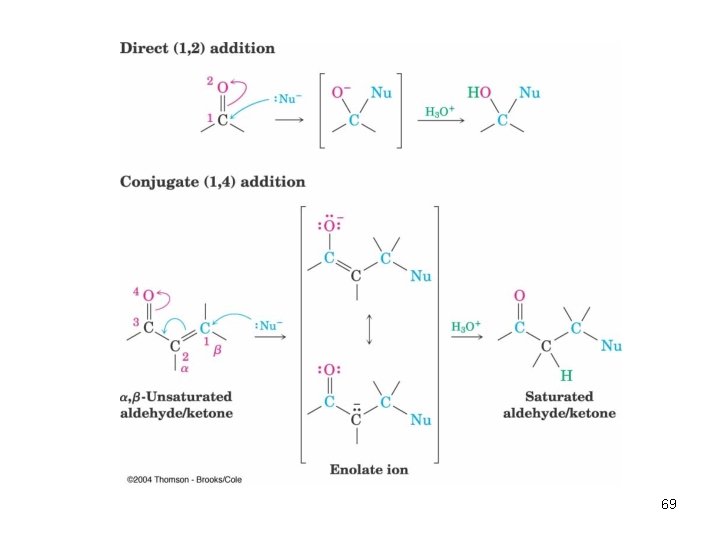
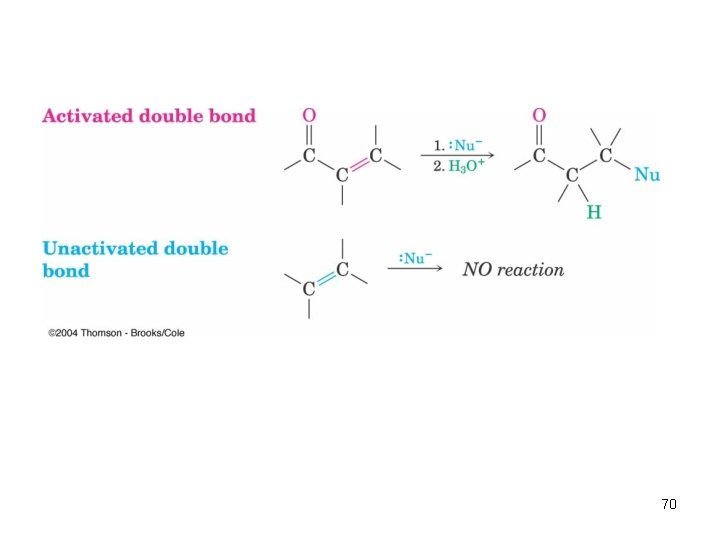
Conjugate Nucleophilic Addition to , b-Unsaturated Aldehydes and Ketones • A nucleophile can add to the C=C double bond of an , b-unsaturated aldehyde or ketone (conjugate addition, or 1, 4 addition) • The initial product is a resonance-stabilized enolate ion, which is then protonated 68
69
70
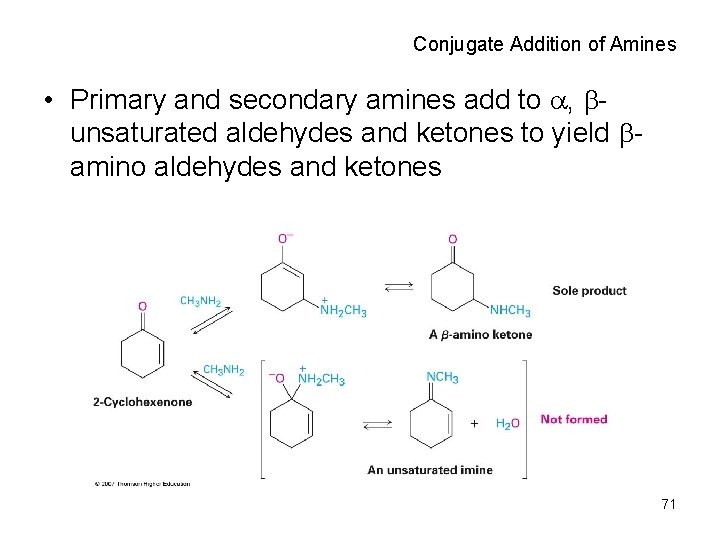
Conjugate Addition of Amines • Primary and secondary amines add to , bunsaturated aldehydes and ketones to yield bamino aldehydes and ketones 71

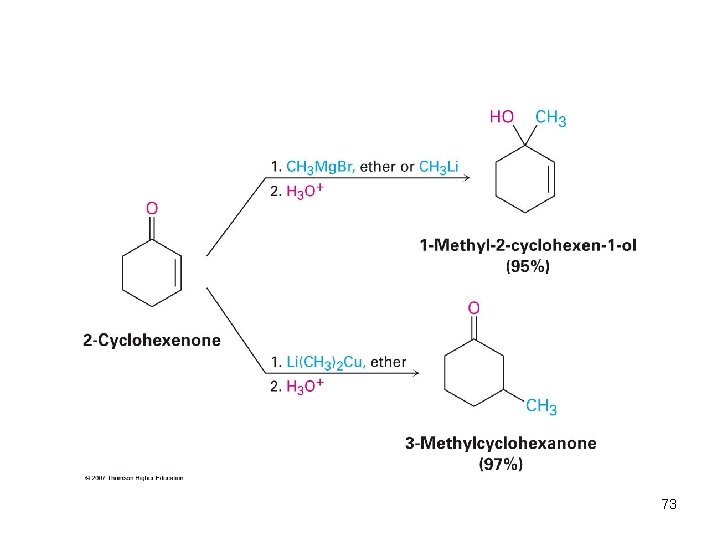
Conjugate Addition of Alkyl Groups: Organocopper Reactions • Reaction of an , b-unsaturated ketone with a lithium diorganocopper reagent • Diorganocopper (Gilman) reagents from by reaction of 1 equivalent of cuprous iodide and 2 equivalents of organolithium • 1 , 2 , 3 alkyl, aryl and alkenyl groups react but not alkynyl groups 72
73
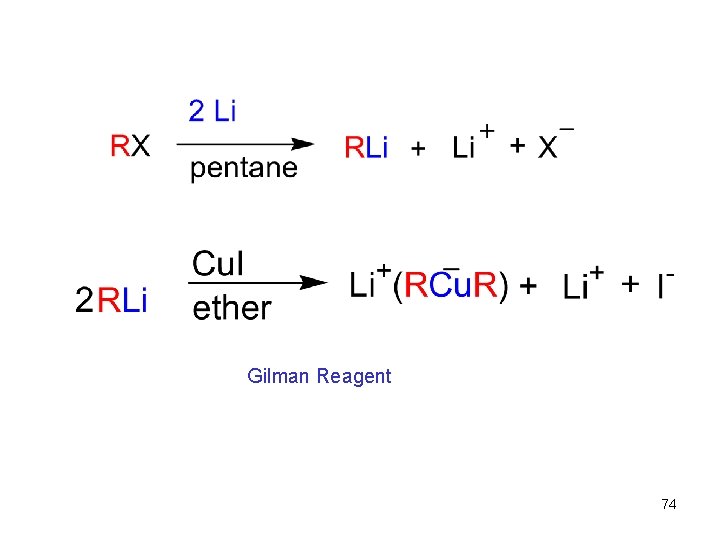
Gilman Reagent 74
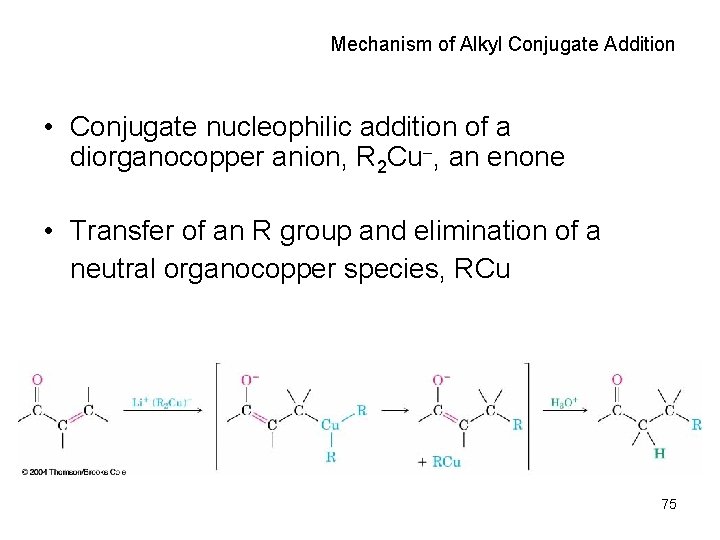
Mechanism of Alkyl Conjugate Addition • Conjugate nucleophilic addition of a diorganocopper anion, R 2 Cu , an enone • Transfer of an R group and elimination of a neutral organocopper species, RCu 75
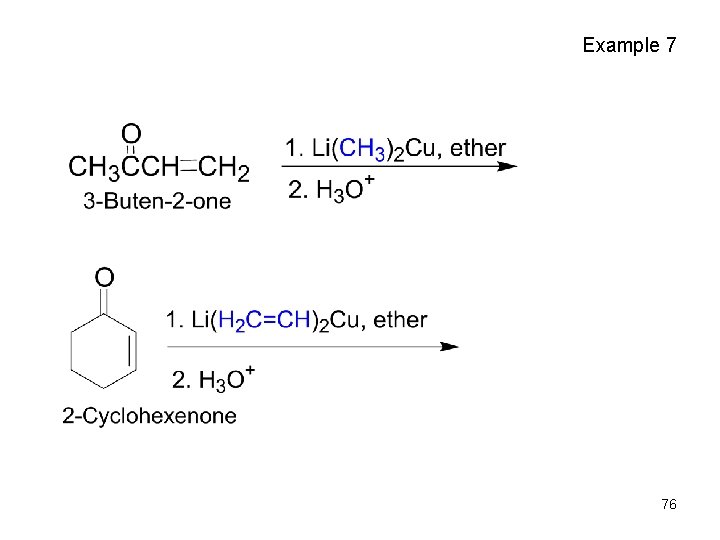
Example 7 76
- Slides: 76