Reactions of Alcohols Oxidation RX Ether and Ester













- Slides: 60

Reactions of Alcohols Oxidation R-X, Ether, and Ester Preparation Protection of Alcohols Synthesis The Logic of Mechanisms

Alcohols are Synthetically Versatile

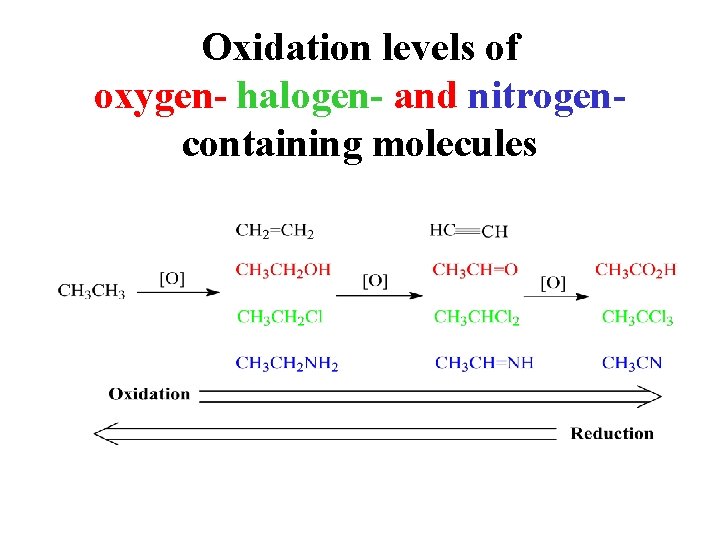
Oxidation levels of oxygen- halogen- and nitrogencontaining molecules

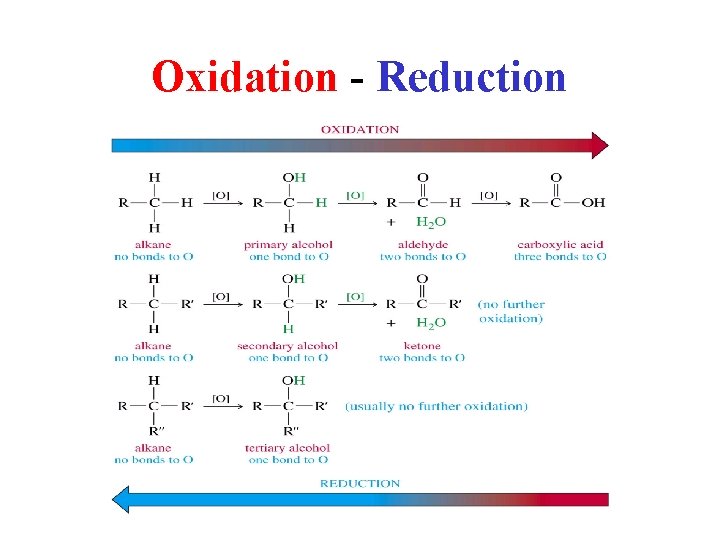
Oxidation - Reduction

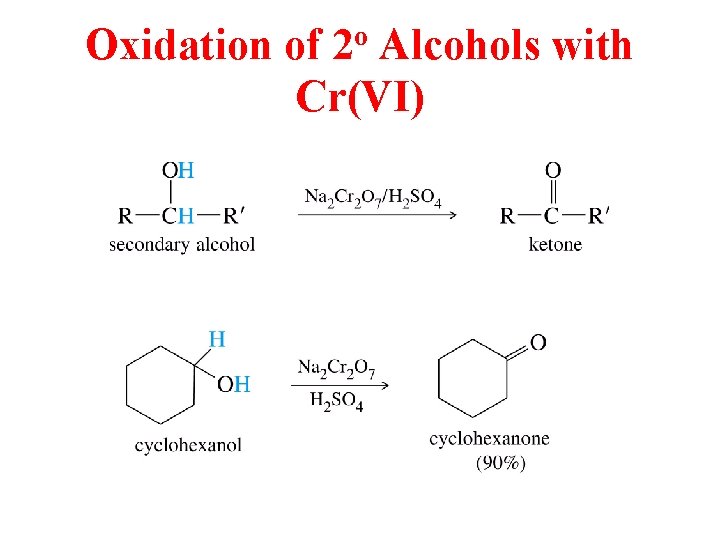
Oxidation of 2 o Alcohols with Cr(VI)

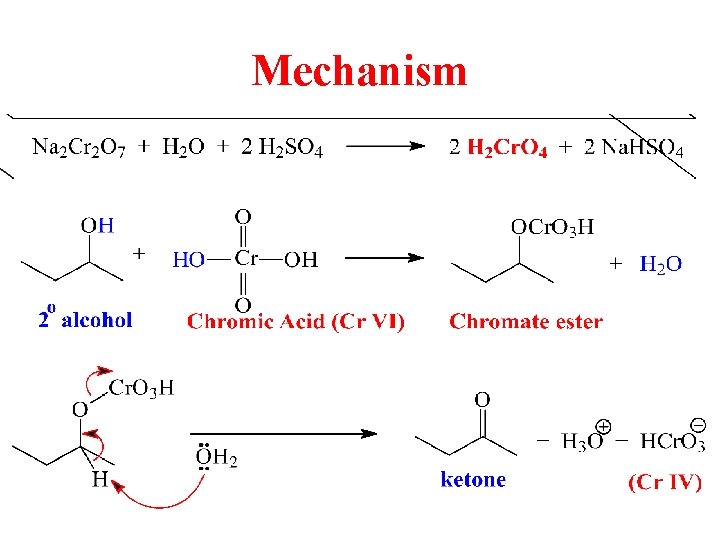
Mechanism

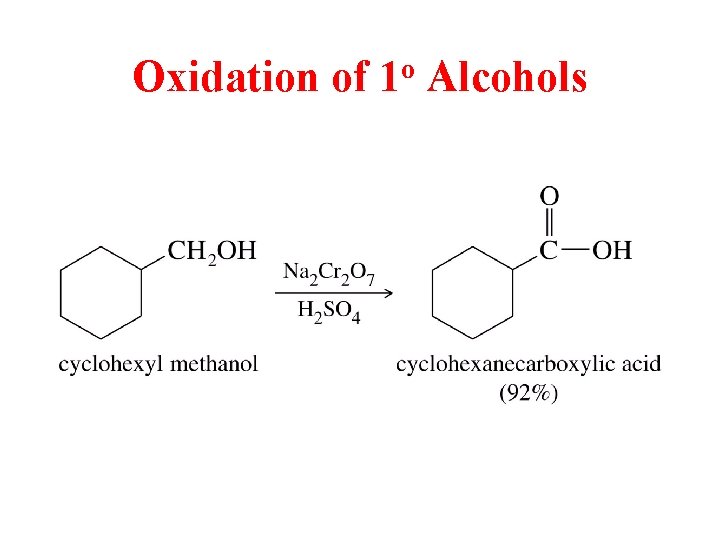
Oxidation of 1 o Alcohols

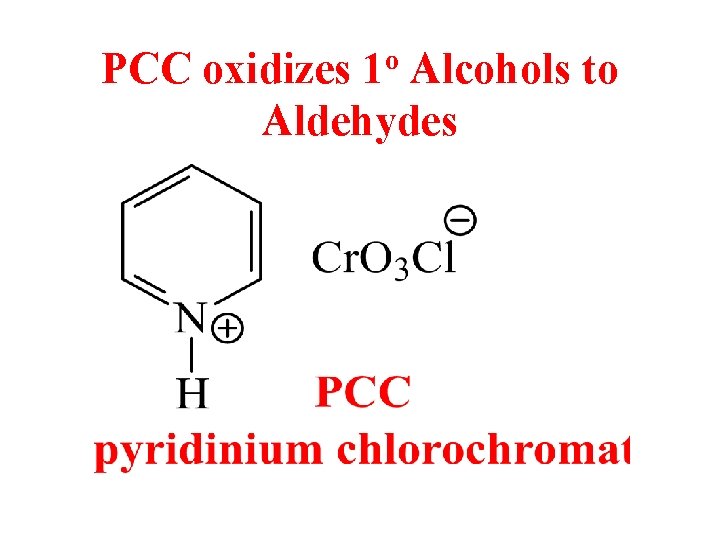
PCC oxidizes 1 o Alcohols to Aldehydes

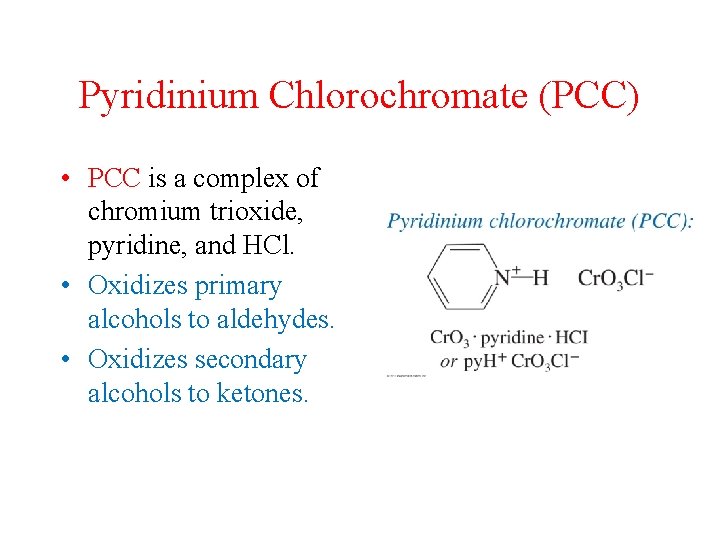
Pyridinium Chlorochromate (PCC) • PCC is a complex of chromium trioxide, pyridine, and HCl. • Oxidizes primary alcohols to aldehydes. • Oxidizes secondary alcohols to ketones.

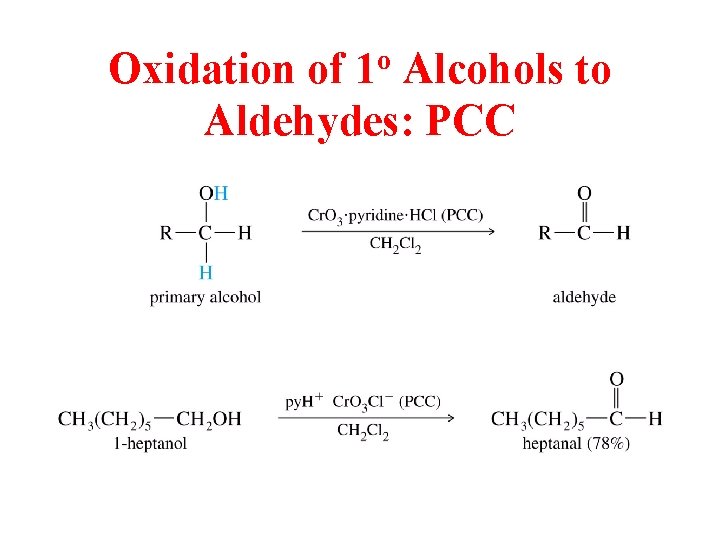
Oxidation of 1 o Alcohols to Aldehydes: PCC

3° Alcohols Cannot Be Oxidized • Carbon does not have hydrogen, so oxidation is difficult and involves the breakage of a C—C bond. • Chromic acid test is for primary and secondary alcohols because tertiary alcohols do not react. Orange color of Cr(VII) turns green - Cr(III); 3 o alcohol is not oxidized, therefore no color change.

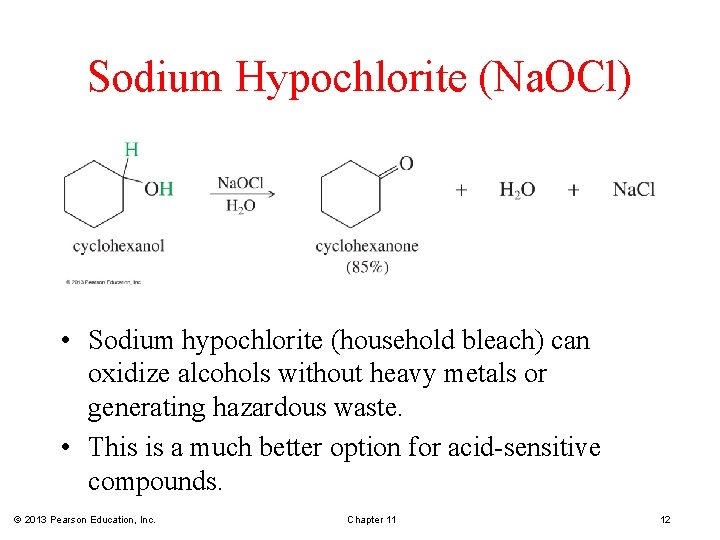
Sodium Hypochlorite (Na. OCl) • Sodium hypochlorite (household bleach) can oxidize alcohols without heavy metals or generating hazardous waste. • This is a much better option for acid-sensitive compounds. © 2013 Pearson Education, Inc. Chapter 11 12

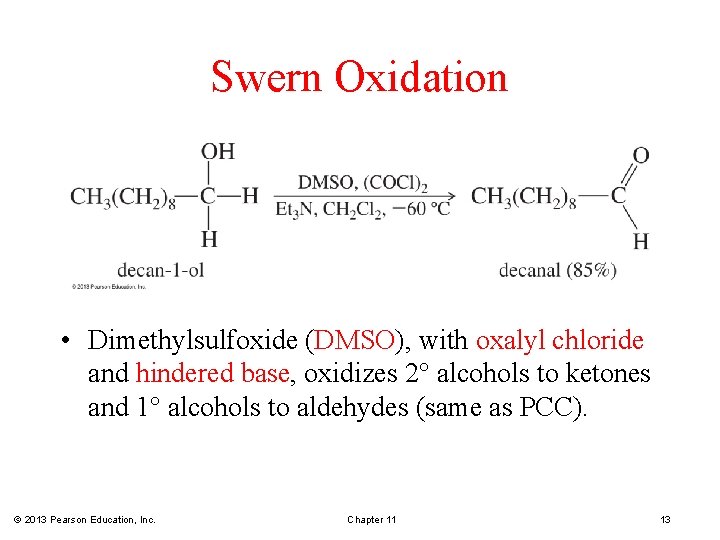
Swern Oxidation • Dimethylsulfoxide (DMSO), with oxalyl chloride and hindered base, oxidizes 2 alcohols to ketones and 1 alcohols to aldehydes (same as PCC). © 2013 Pearson Education, Inc. Chapter 11 13

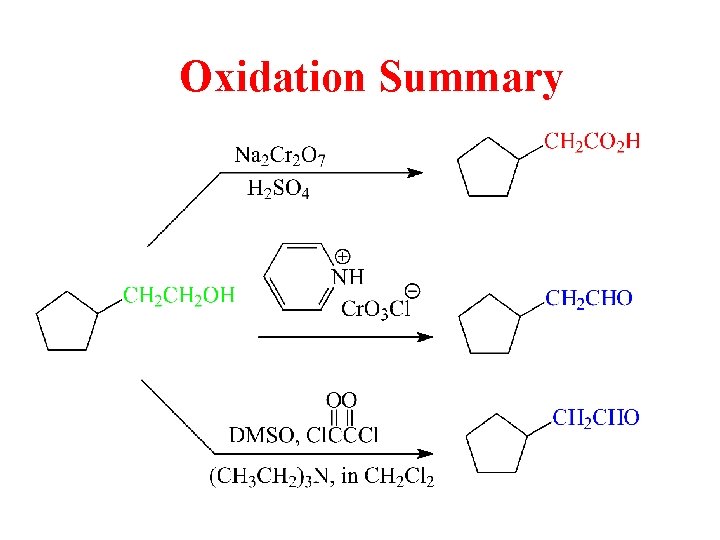
Oxidation Summary

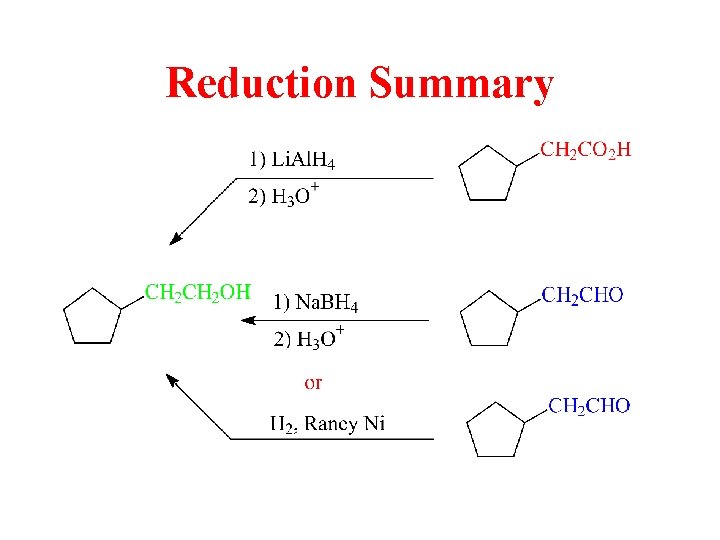
Reduction Summary

Conversion of Alcohol into a Leaving Group • Form Tosylate (p-Ts. Cl, pyridine) • Use strong acid (H 3 O+) • Convert to Alkyl Halide (HX, SOCl 2, PBr 3)

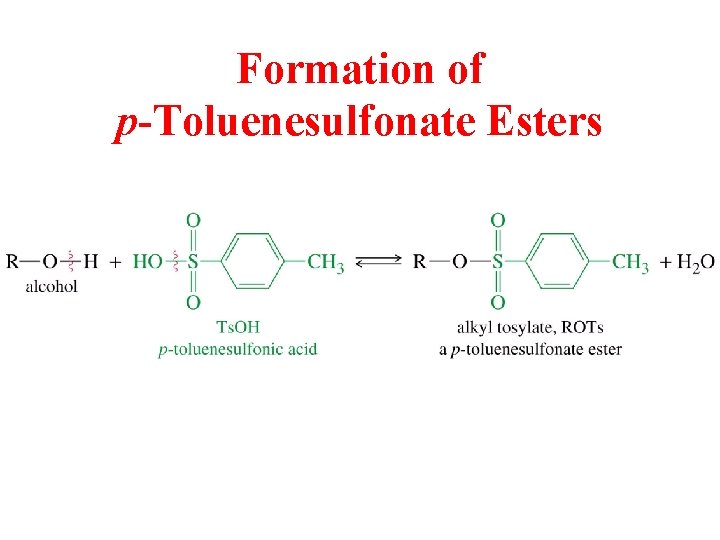
Formation of p-Toluenesulfonate Esters

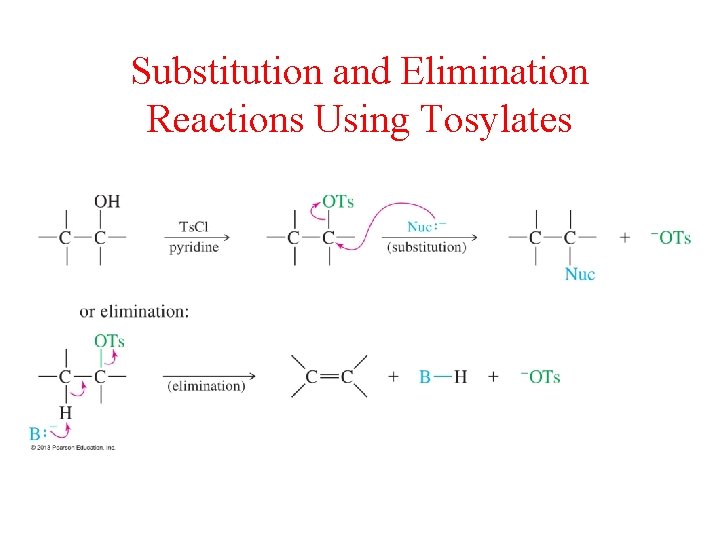
Substitution and Elimination Reactions Using Tosylates

Summary of Tosylate Reactions

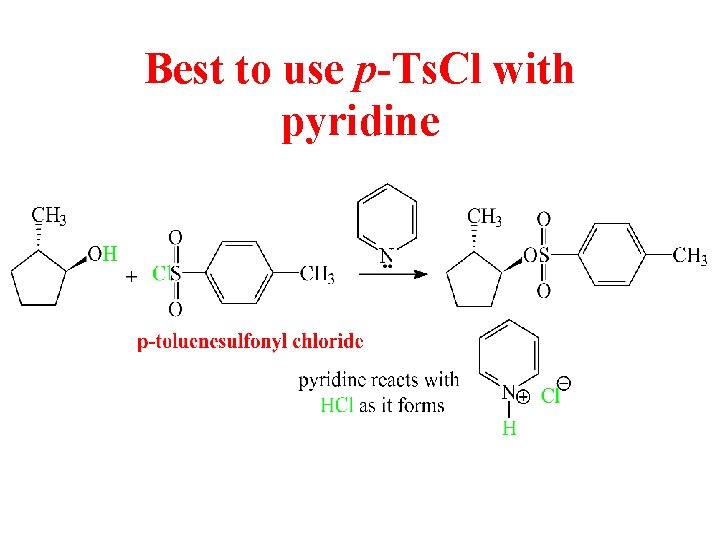
Best to use p-Ts. Cl with pyridine

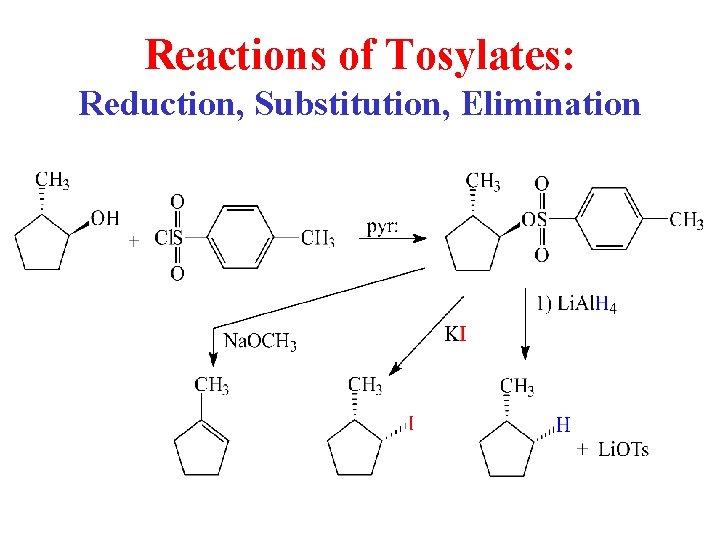
Reactions of Tosylates: Reduction, Substitution, Elimination

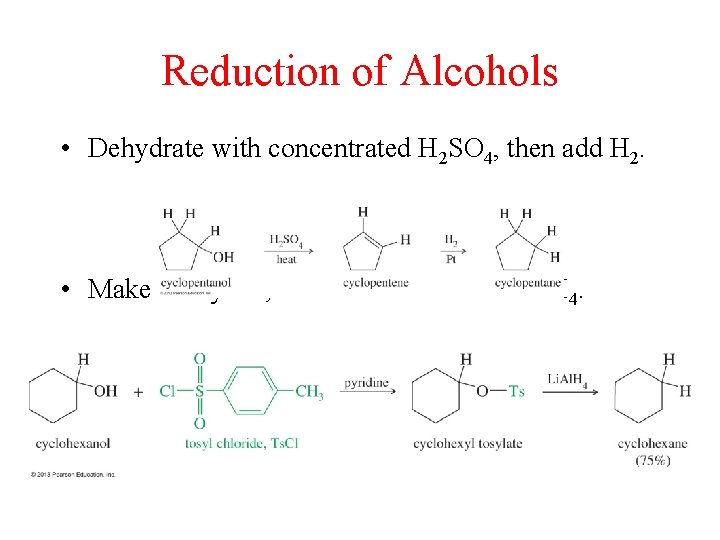
Reduction of Alcohols • Dehydrate with concentrated H 2 SO 4, then add H 2. • Make a tosylate, then reduce it with Li. Al. H 4.

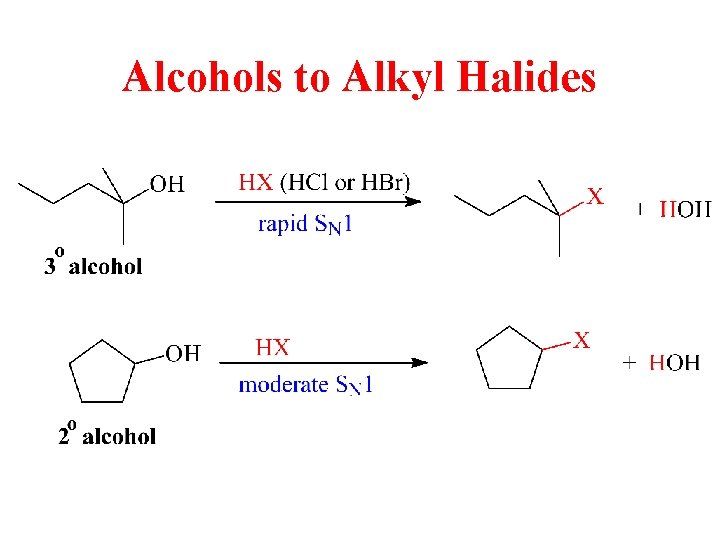
Alcohols to Alkyl Halides

Reaction of Alcohols with Acids • The hydroxyl group is protonated by an acid to convert it into a good leaving group (H 2 O). • Once the alcohol is protonated, a substitution or elimination reaction can take place. © 2013 Pearson Education, Inc. Chapter 11 24

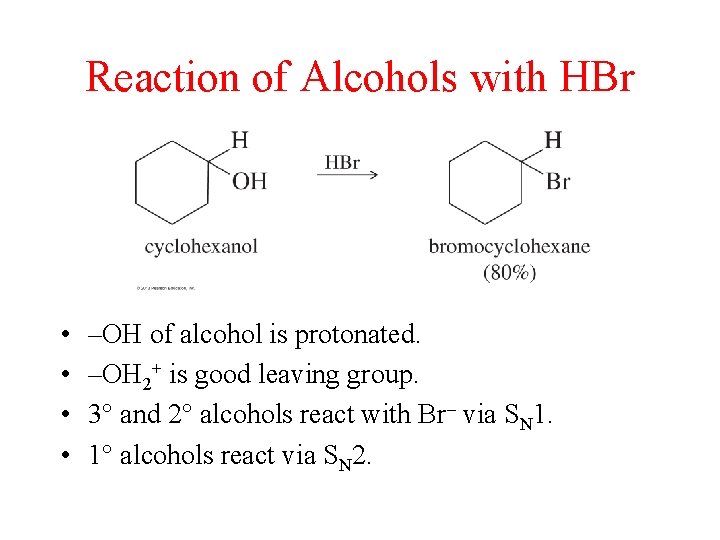
Reaction of Alcohols with HBr • • –OH of alcohol is protonated. –OH 2+ is good leaving group. 3° and 2° alcohols react with Br– via SN 1. 1° alcohols react via SN 2.

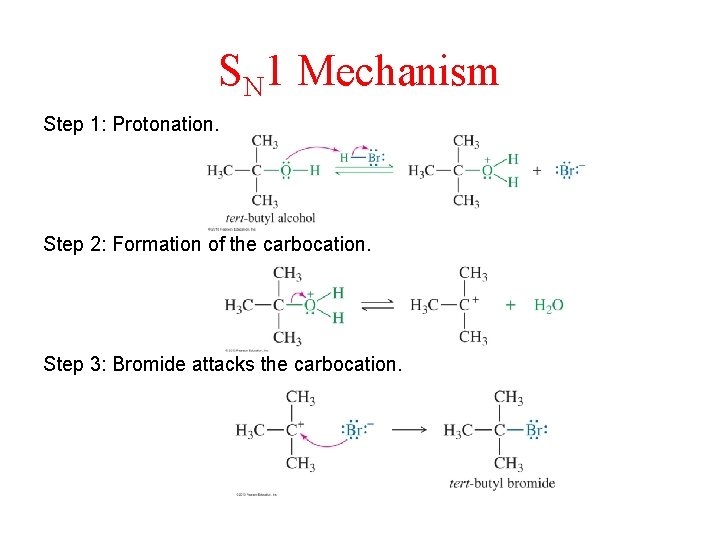
SN 1 Mechanism Step 1: Protonation. Step 2: Formation of the carbocation. Step 3: Bromide attacks the carbocation.

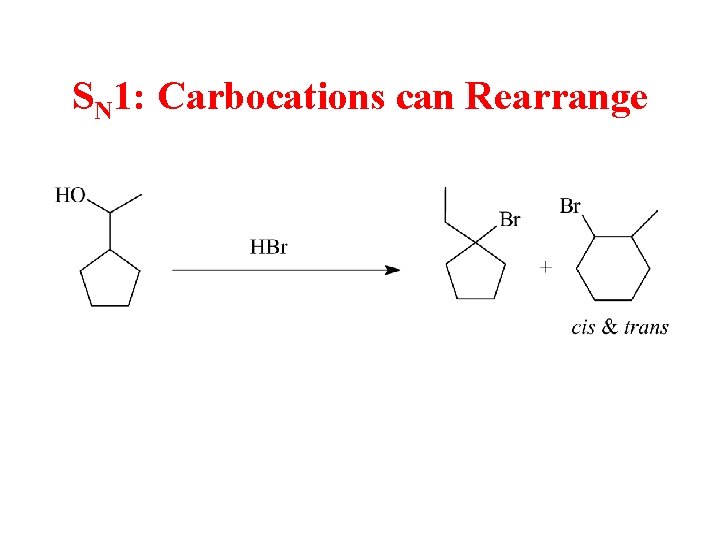
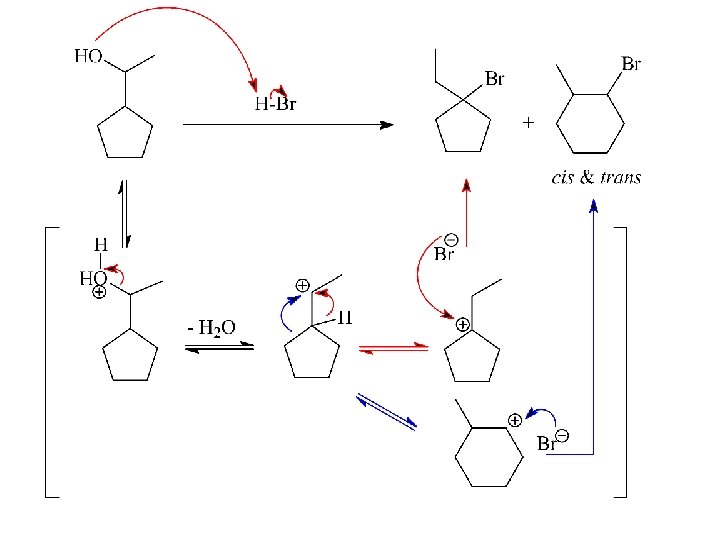
SN 1: Carbocations can Rearrange


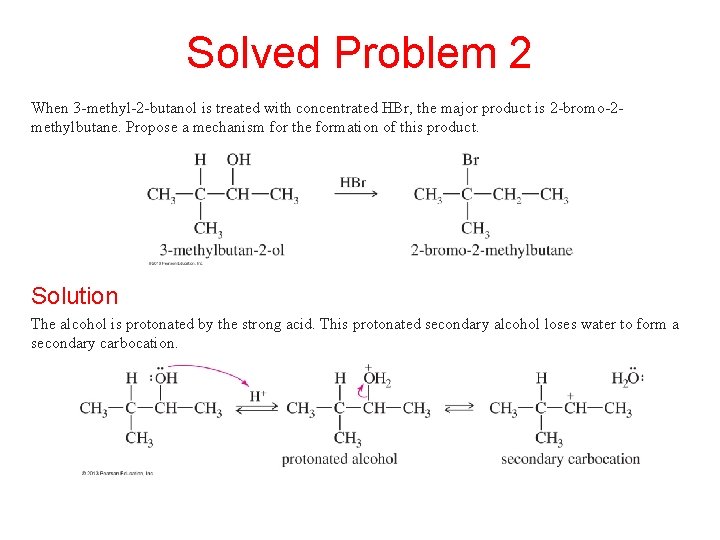
Solved Problem 2 When 3 -methyl-2 -butanol is treated with concentrated HBr, the major product is 2 -bromo-2 methylbutane. Propose a mechanism for the formation of this product. Solution The alcohol is protonated by the strong acid. This protonated secondary alcohol loses water to form a secondary carbocation.

Solved Problem 2 (Continued) Solution (Continued) A hydride shift transforms the secondary carbocation into a more stable tertiary cation. Attack by bromide leads to the observed product.

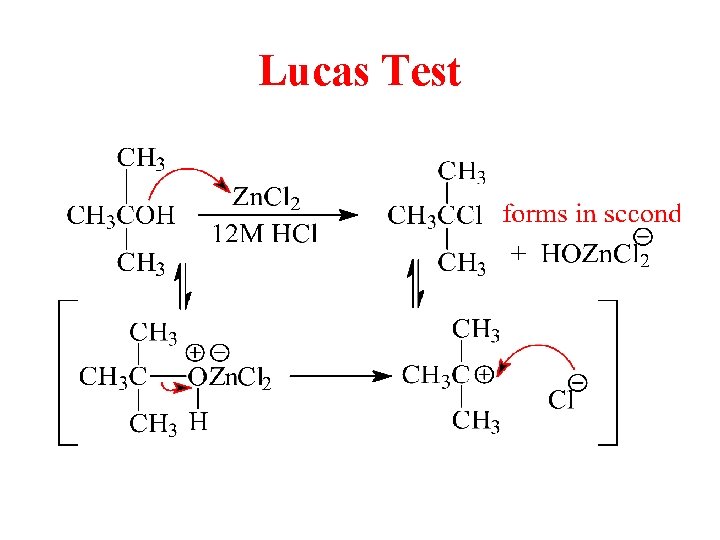
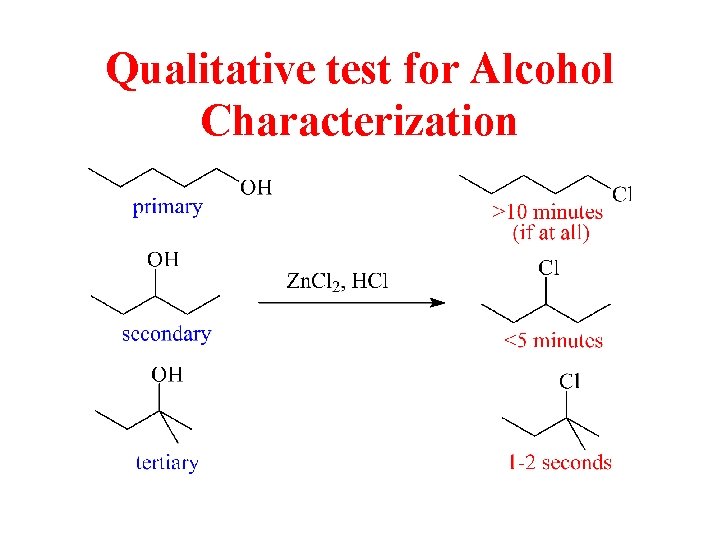
Lucas Test

SN 2 Reaction with the Lucas Reagent • Primary alcohols react with the Lucas reagent (HCl and Zn. Cl 2) by the SN 2 mechanism. • Reaction is very slow. The reaction can take from several minutes to several days. © 2013 Pearson Education, Inc. Chapter 11 32

Qualitative test for Alcohol Characterization

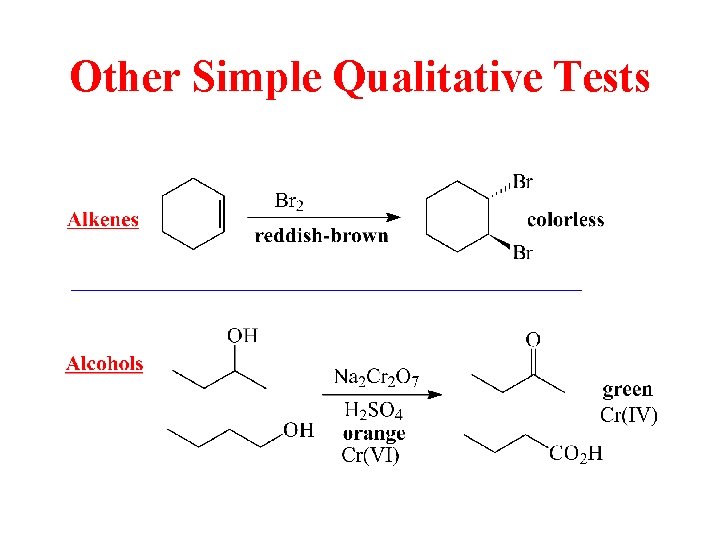
Other Simple Qualitative Tests

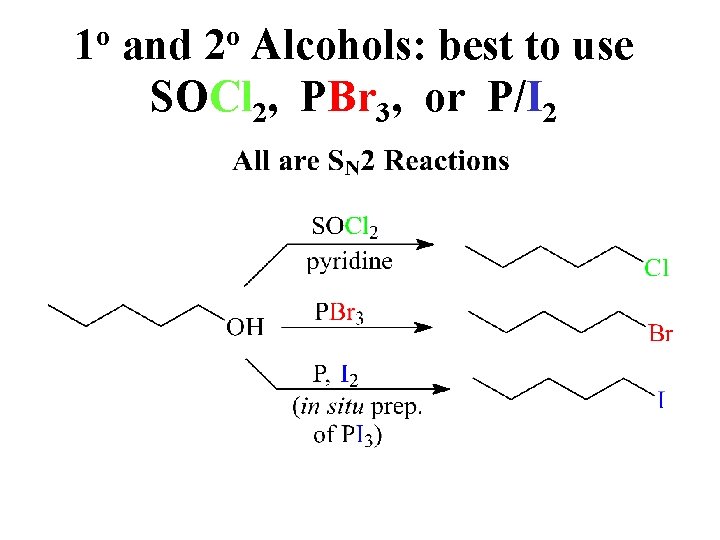
1 o and 2 o Alcohols: best to use SOCl 2, PBr 3, or P/I 2

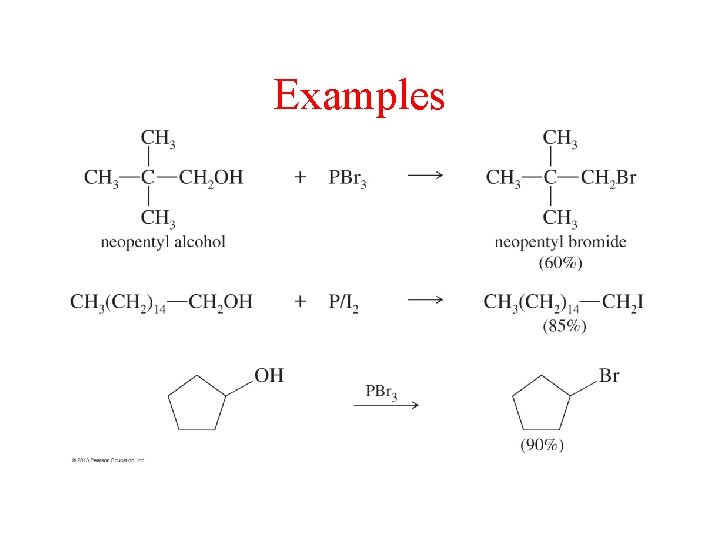
Examples

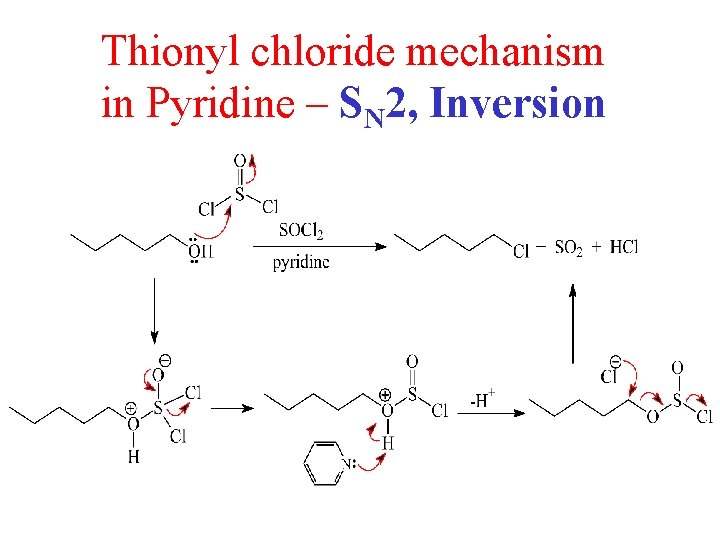
Thionyl chloride mechanism in Pyridine – SN 2, Inversion

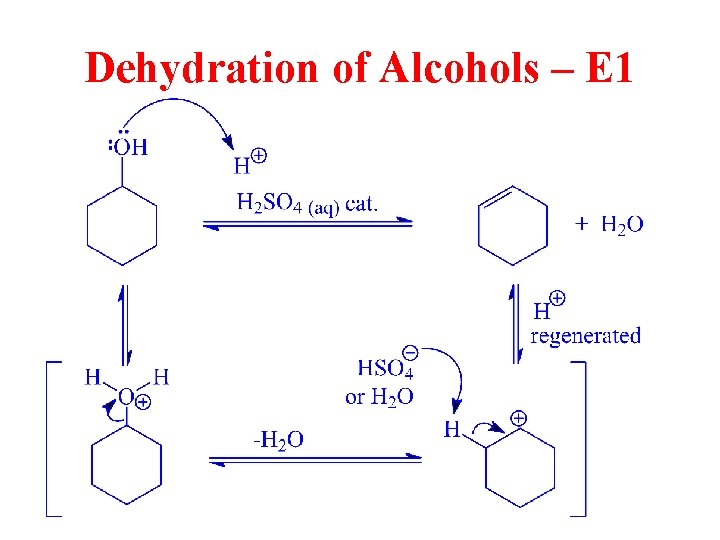
Dehydration of Alcohols • Alcohol dehydration generally takes place through the E 1 mechanism. • Rearrangements are possible. • The rate of the reaction follows the same rate as the ease of formation of carbocations: 3 o > 2 o > 1 o. • Primary alcohols rearrange, so this is not a good reaction for converting 1° alcohols into alkenes. © 2013 Pearson Education, Inc. Chapter 11 38

Dehydration of Alcohols – E 1

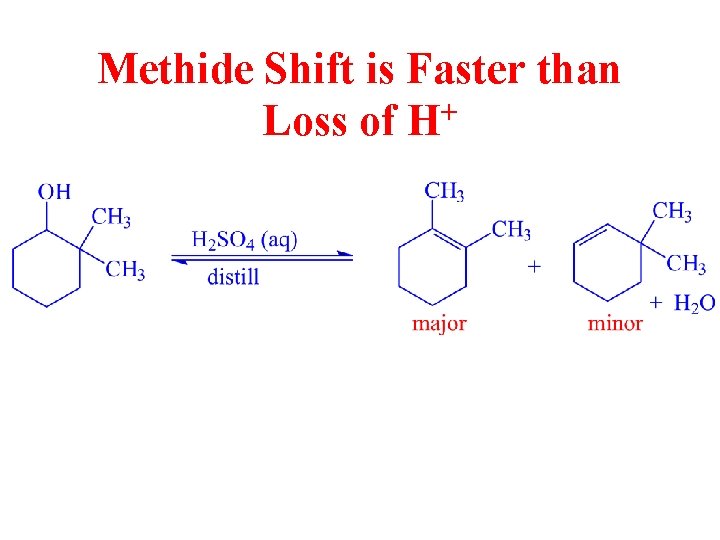
Methide Shift is Faster than Loss of H+

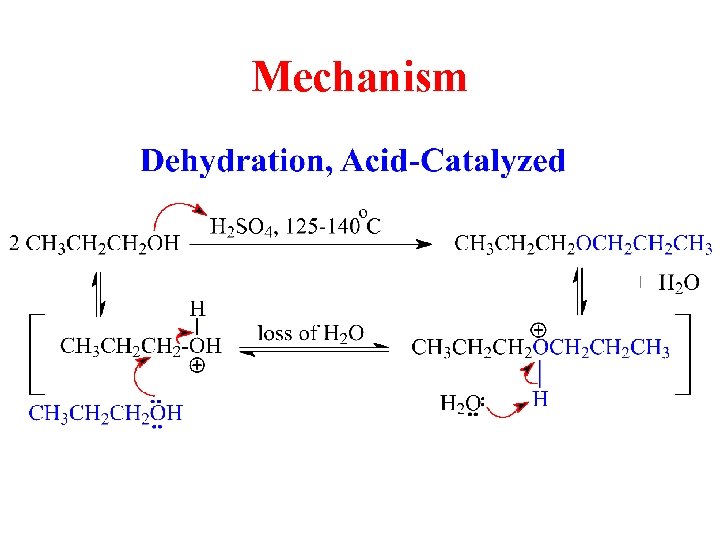
Dimerization of Alcohols: Symmetrical Ethers

Mechanism

Esterification • • • Fischer: Alcohol + carboxylic acid Tosylate esters Sulfate esters Nitrate esters Phosphate esters

Fischer Esterification • Reaction of an alcohol and a carboxylic acid produces an ester. • Sulfuric acid is a catalyst. • The reaction is an equilibrium between starting materials and products, and for this reason the Fischer esterification is seldom used to prepare esters. © 2013 Pearson Education, Inc. Chapter 11 44

Nitrate Esters • The best-known nitrate ester is nitroglycerine, whose systematic name is glyceryl trinitrate. • Glyceryl nitrate results from the reaction of glycerol (1, 2, 3 propanetriol) with three molecules of nitric acid. © 2013 Pearson Education, Inc. Chapter 11 45

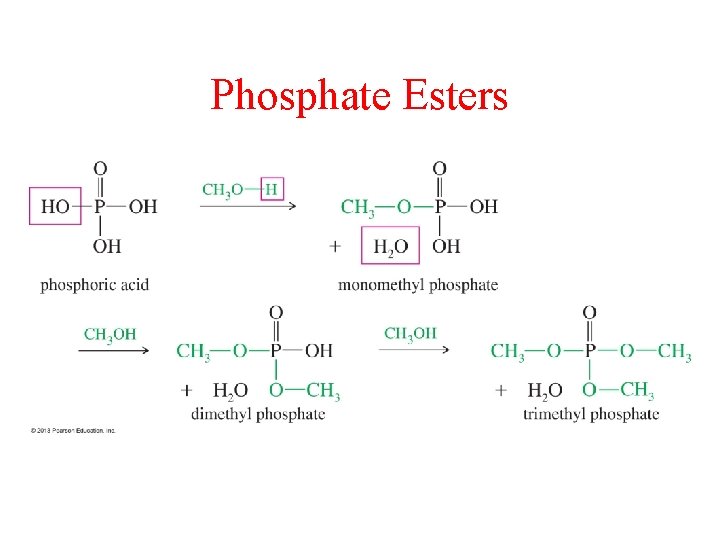
Phosphate Esters

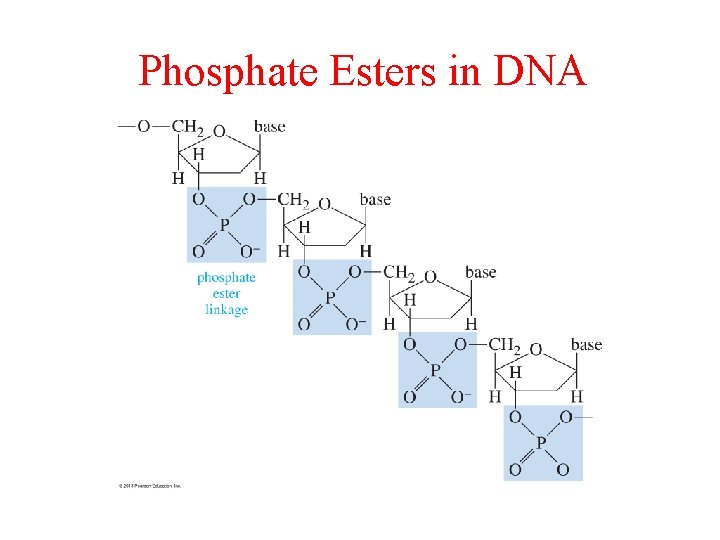
Phosphate Esters in DNA

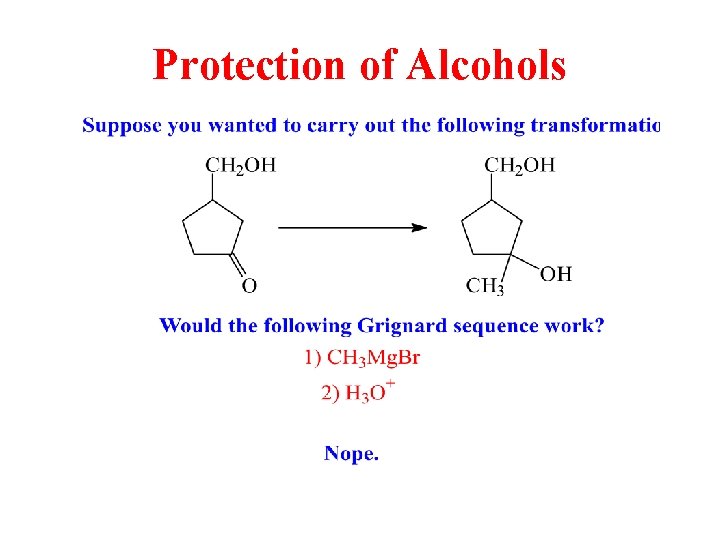
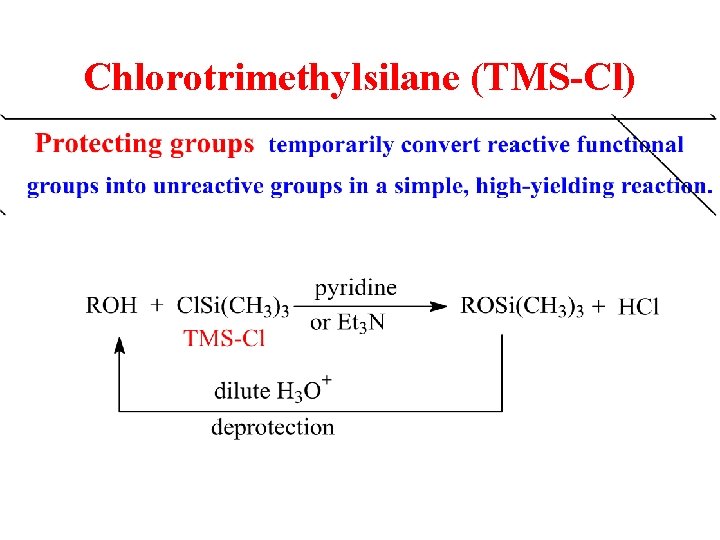
Protection of Alcohols

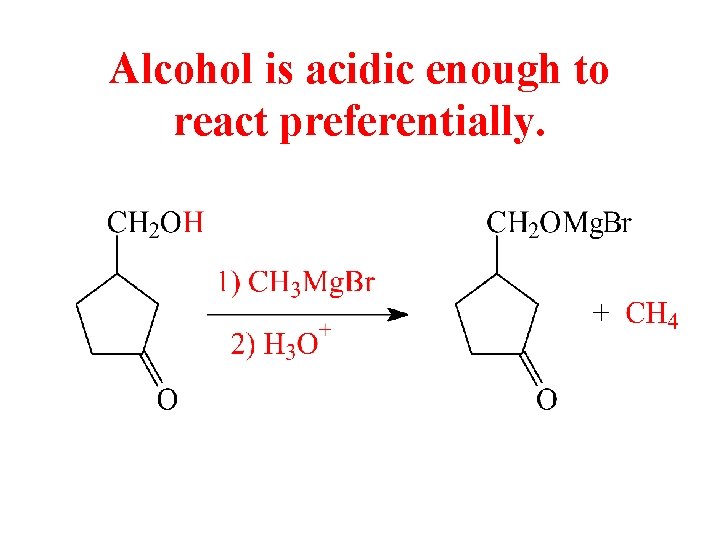
Alcohol is acidic enough to react preferentially.

Chlorotrimethylsilane (TMS-Cl)

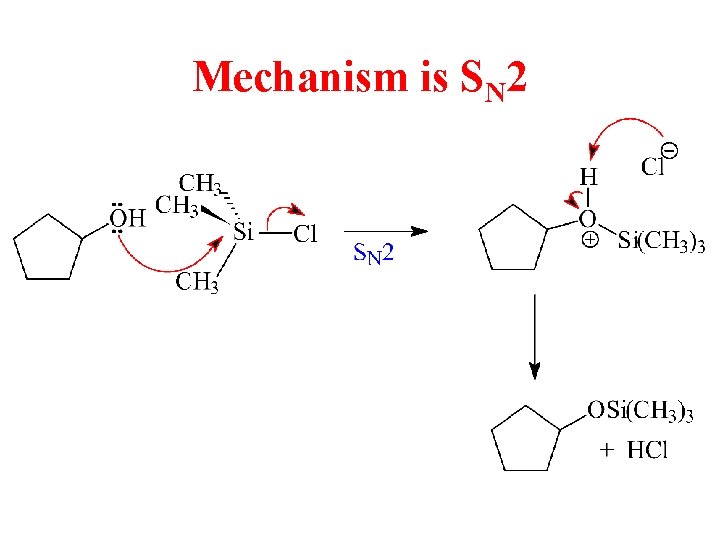
Mechanism is SN 2

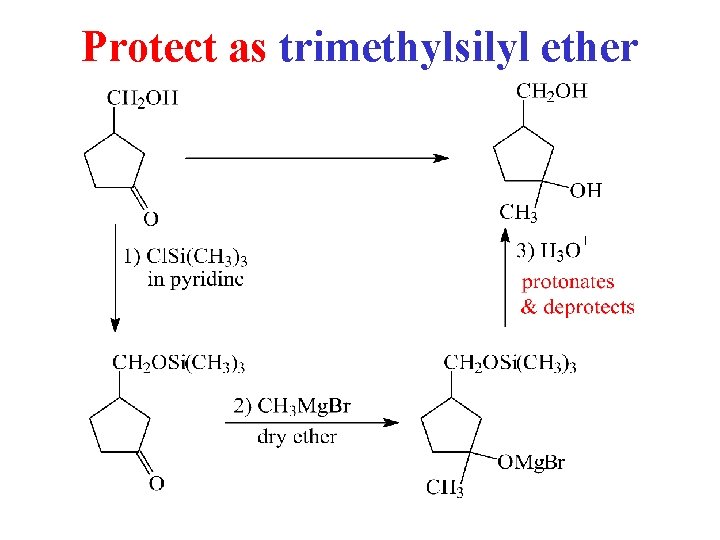
Protect as trimethylsilyl ether

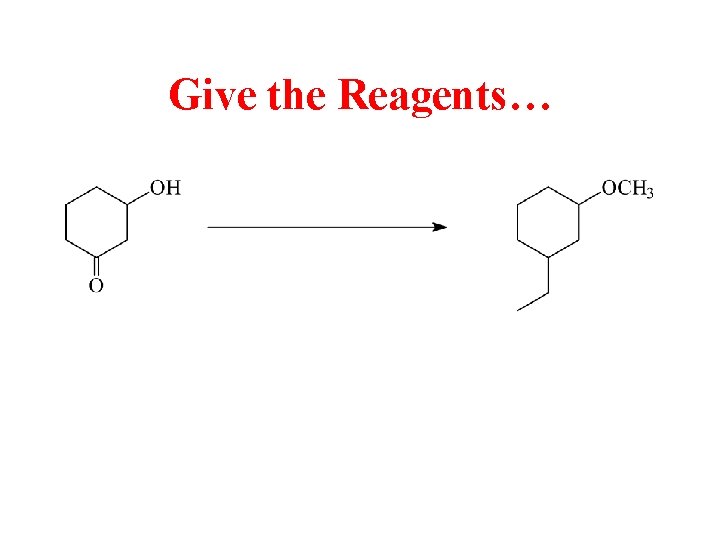
Give the Reagents…

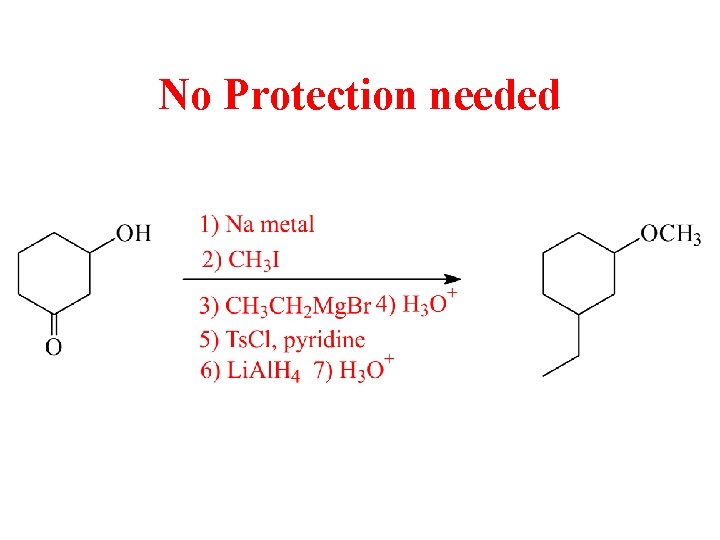
No Protection needed

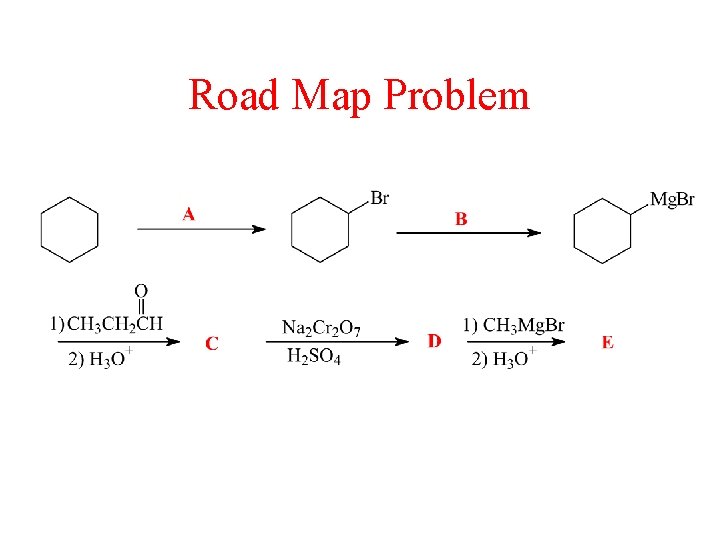
Road Map Problem

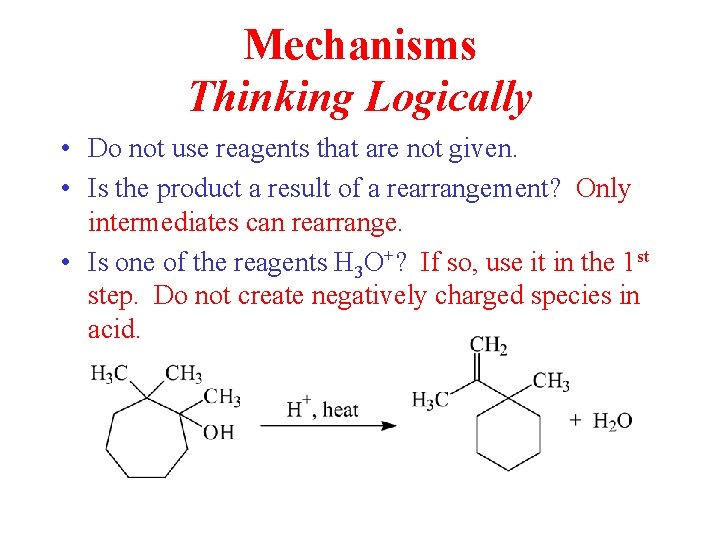
Mechanisms Thinking Logically • Do not use reagents that are not given. • Is the product a result of a rearrangement? Only intermediates can rearrange. • Is one of the reagents H 3 O+? If so, use it in the 1 st step. Do not create negatively charged species in acid.

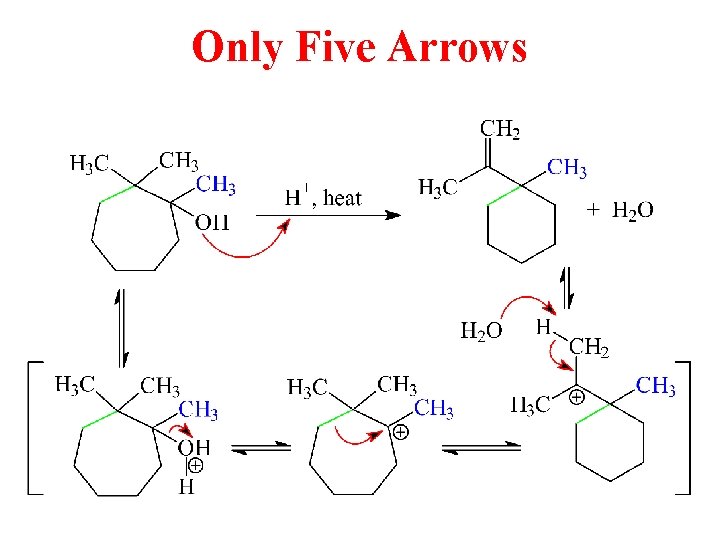
Only Five Arrows

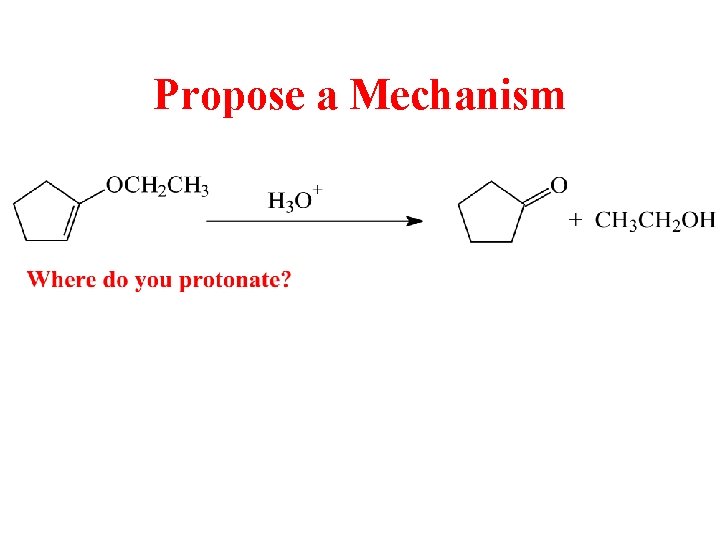
Propose a Mechanism

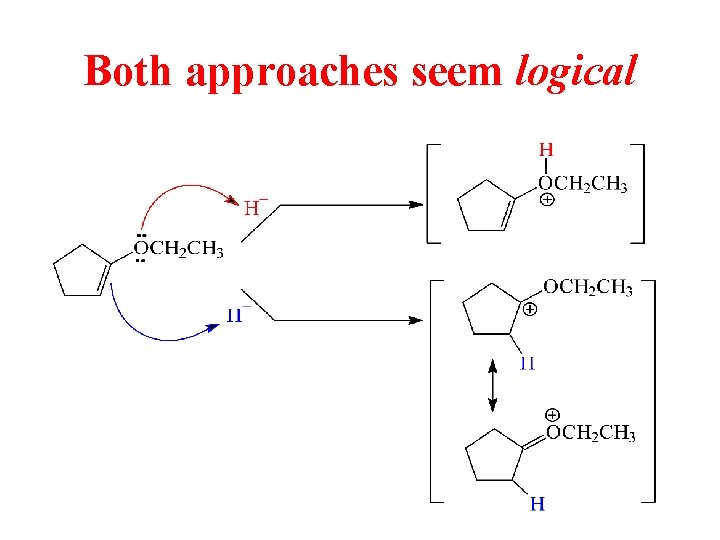
Both approaches seem logical

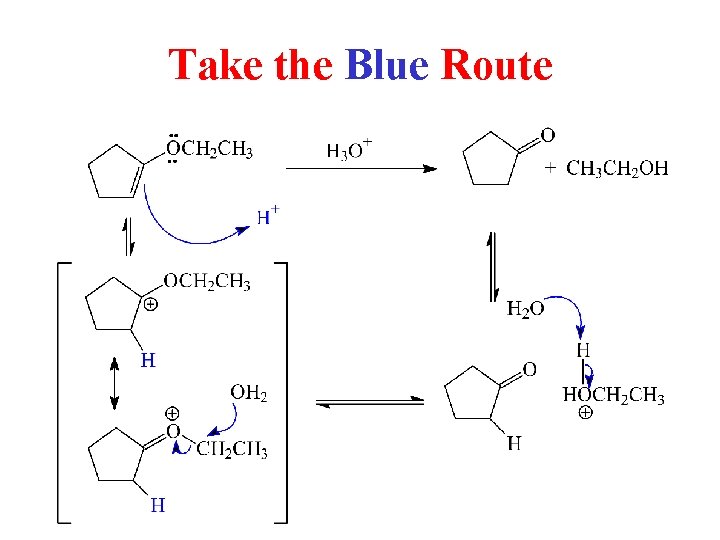
Take the Blue Route