CARBONYL COMPOUNDS WORKSHEET 1 Draw and name the
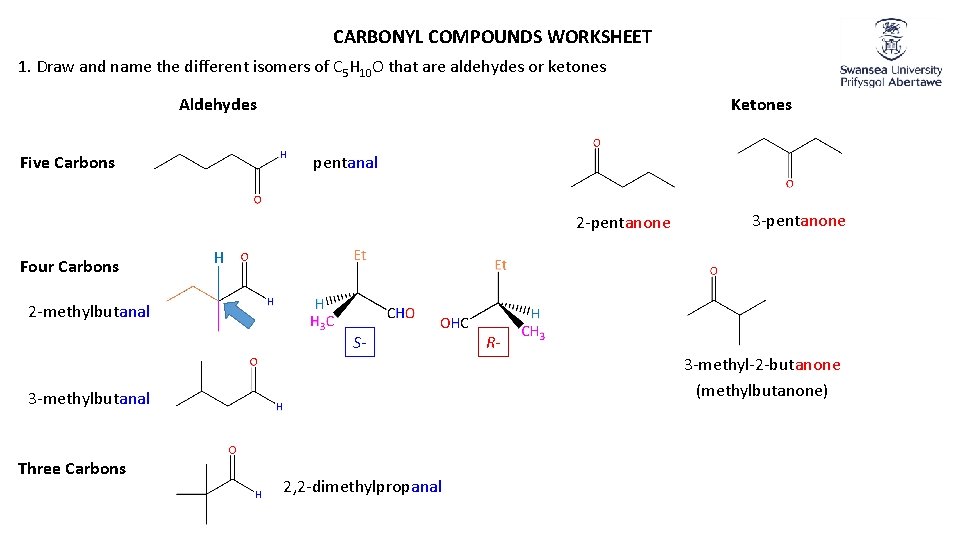
CARBONYL COMPOUNDS WORKSHEET 1. Draw and name the different isomers of C 5 H 10 O that are aldehydes or ketones Aldehydes Ketones pentanal Five Carbons 2 -pentanone Four Carbons 3 -pentanone H 2 -methylbutanal S- 3 -methyl-2 -butanone (methylbutanone) 3 -methylbutanal Three Carbons R- 2, 2 -dimethylpropanal
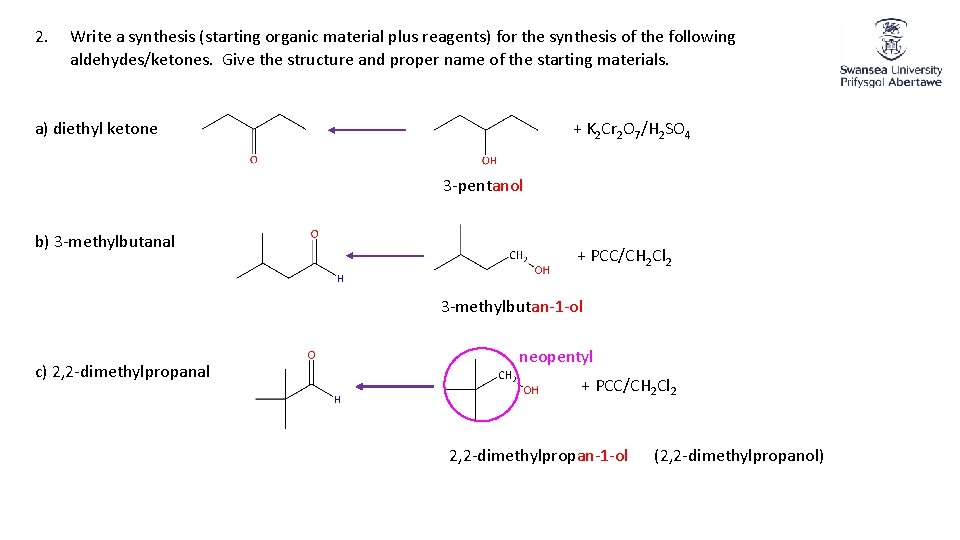
2. Write a synthesis (starting organic material plus reagents) for the synthesis of the following aldehydes/ketones. Give the structure and proper name of the starting materials. a) diethyl ketone + K 2 Cr 2 O 7/H 2 SO 4 3 -pentanol b) 3 -methylbutanal + PCC/CH 2 Cl 2 3 -methylbutan-1 -ol c) 2, 2 -dimethylpropanal neopentyl + PCC/CH 2 Cl 2 2, 2 -dimethylpropan-1 -ol (2, 2 -dimethylpropanol)
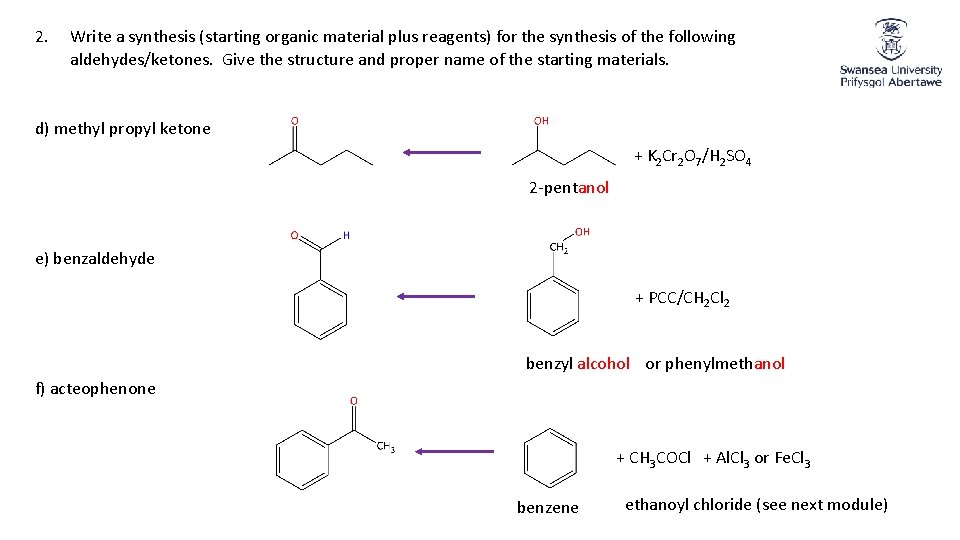
2. Write a synthesis (starting organic material plus reagents) for the synthesis of the following aldehydes/ketones. Give the structure and proper name of the starting materials. d) methyl propyl ketone + K 2 Cr 2 O 7/H 2 SO 4 2 -pentanol e) benzaldehyde + PCC/CH 2 Cl 2 benzyl alcohol or phenylmethanol f) acteophenone + CH 3 COCl + Al. Cl 3 or Fe. Cl 3 benzene ethanoyl chloride (see next module)
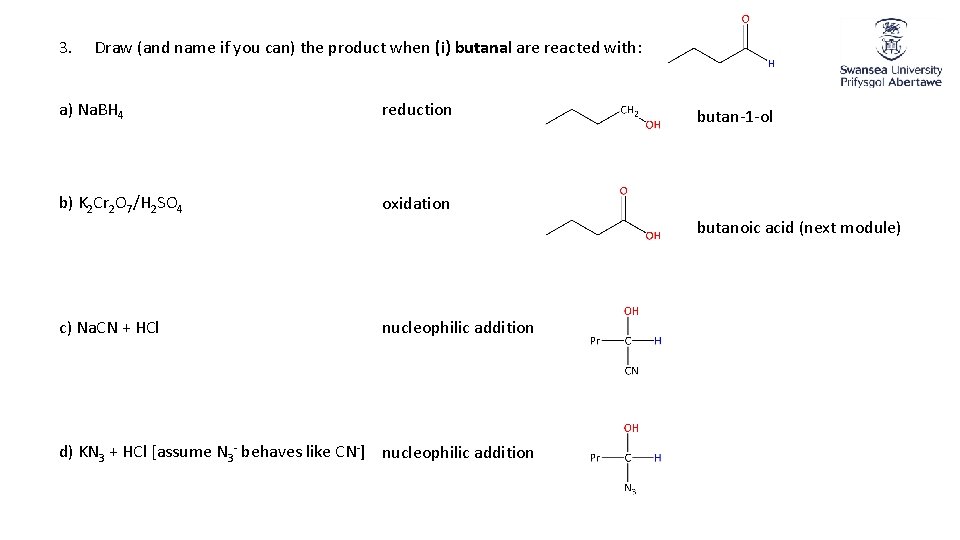
3. Draw (and name if you can) the product when (i) butanal are reacted with: a) Na. BH 4 reduction b) K 2 Cr 2 O 7/H 2 SO 4 oxidation butan-1 -ol butanoic acid (next module) c) Na. CN + HCl nucleophilic addition d) KN 3 + HCl [assume N 3 - behaves like CN-] nucleophilic addition
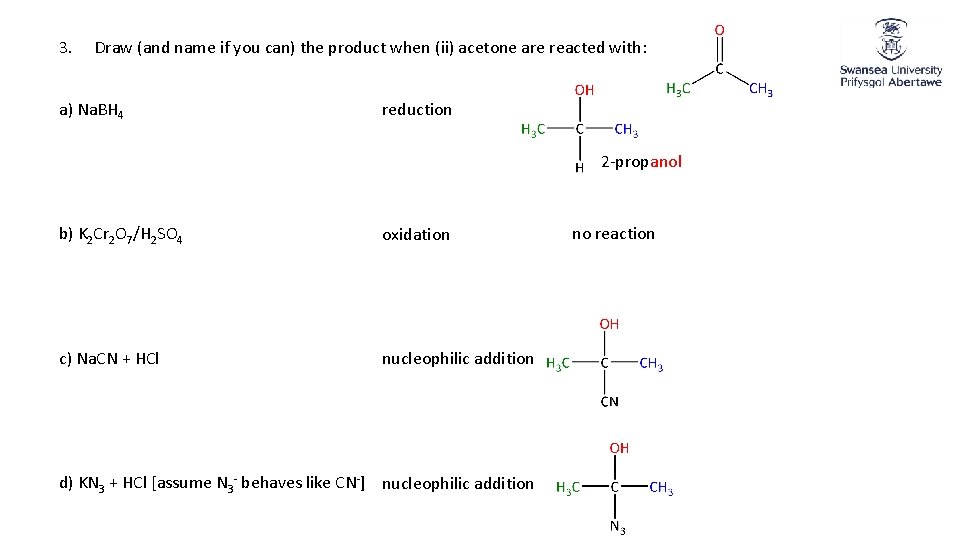
3. Draw (and name if you can) the product when (ii) acetone are reacted with: a) Na. BH 4 reduction 2 -propanol b) K 2 Cr 2 O 7/H 2 SO 4 oxidation c) Na. CN + HCl nucleophilic addition d) KN 3 + HCl [assume N 3 - behaves like CN-] nucleophilic addition no reaction
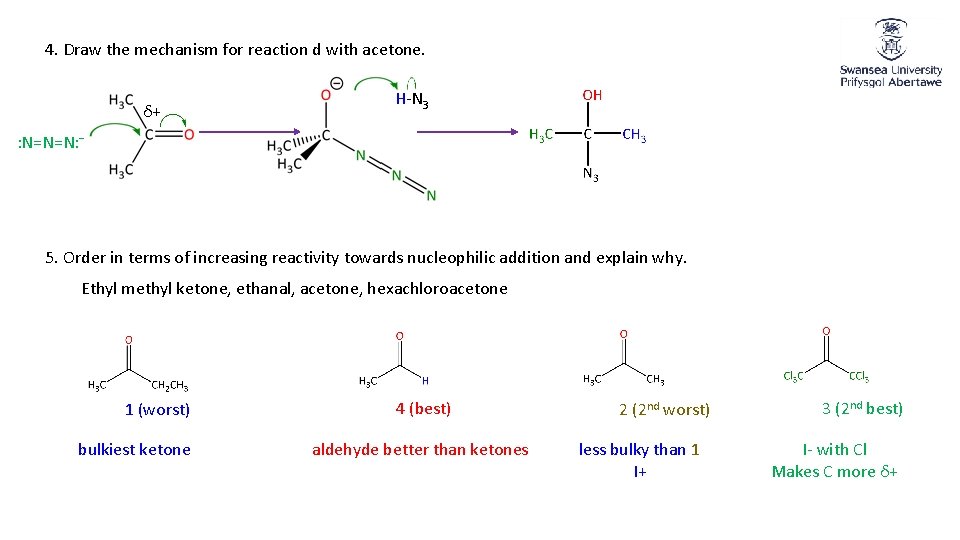
4. Draw the mechanism for reaction d with acetone. δ+ H-N 3 : N=N=N: − 5. Order in terms of increasing reactivity towards nucleophilic addition and explain why. Ethyl methyl ketone, ethanal, acetone, hexachloroacetone 1 (worst) bulkiest ketone 4 (best) aldehyde better than ketones 2 (2 nd worst) less bulky than 1 I+ 3 (2 nd best) I- with Cl Makes C more δ+
- Slides: 6