Alcohols Ethers and Thiols Bettelheim Brown Campbell and

Alcohols, Ethers and Thiols Bettelheim, Brown, Campbell and Farrell Chapter 14

Review of Carbon Classification Type of C Attached to 1 o (primary) C one other carbon 2 o (secondary) C two other carbons 3 o (tertiary) C three other carbons 4 o (quaternary) C four other carbons
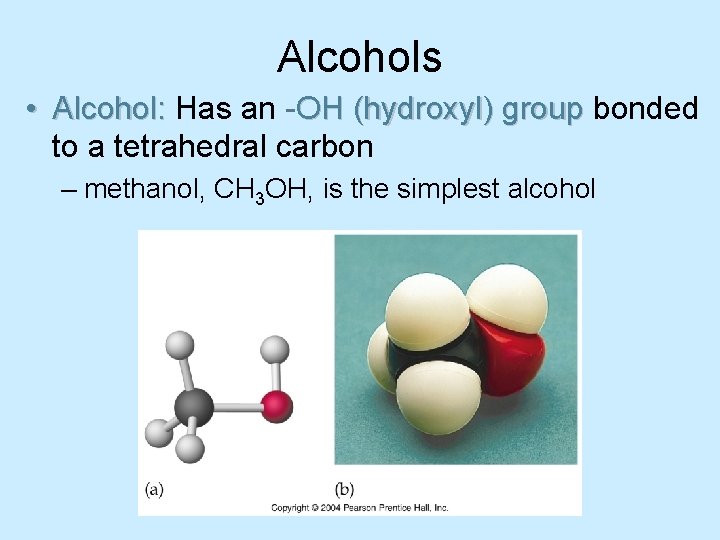
Alcohols • Alcohol: Has an -OH (hydroxyl) group bonded to a tetrahedral carbon – methanol, CH 3 OH, is the simplest alcohol
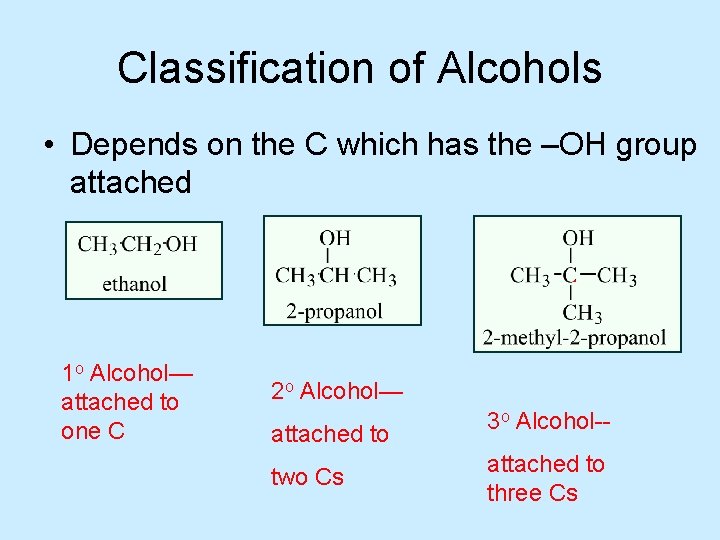
Classification of Alcohols • Depends on the C which has the –OH group attached 1 o Alcohol— attached to one C 2 o Alcohol— attached to two Cs 3 o Alcohol-attached to three Cs

Alcohol Nomenclature 1. Find longest carbon chain that contains the -OH group (parent chain) 2. Number chain from end that gives the -OH the lower number 3. Change the ending -e to -ol 4. Use a number to show the location of the -OH group For cyclic alcohols, the carbon with the -OH group is C-1 5. Name and number substituents and list them in alphabetical order
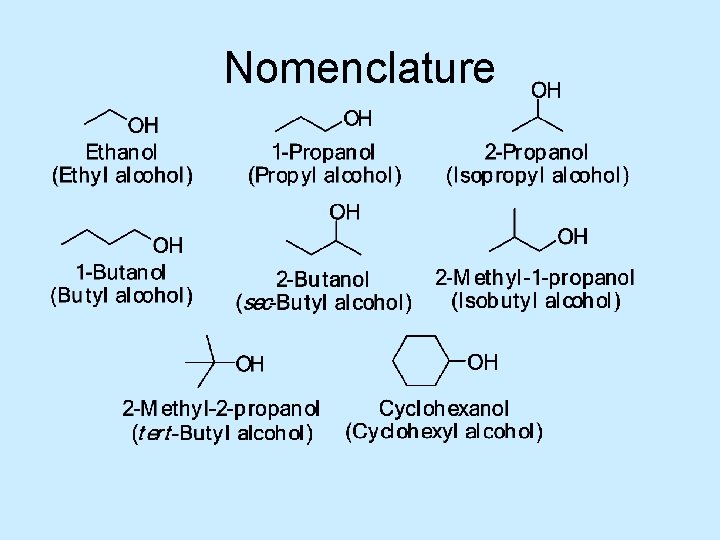
Nomenclature
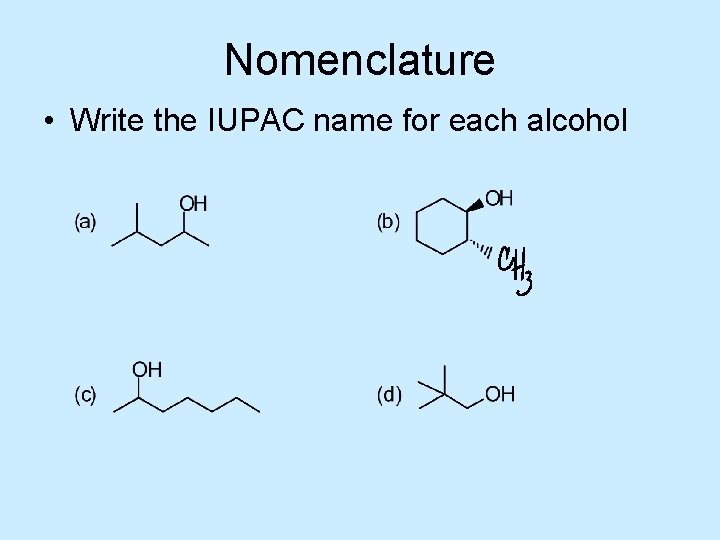
Nomenclature • Write the IUPAC name for each alcohol
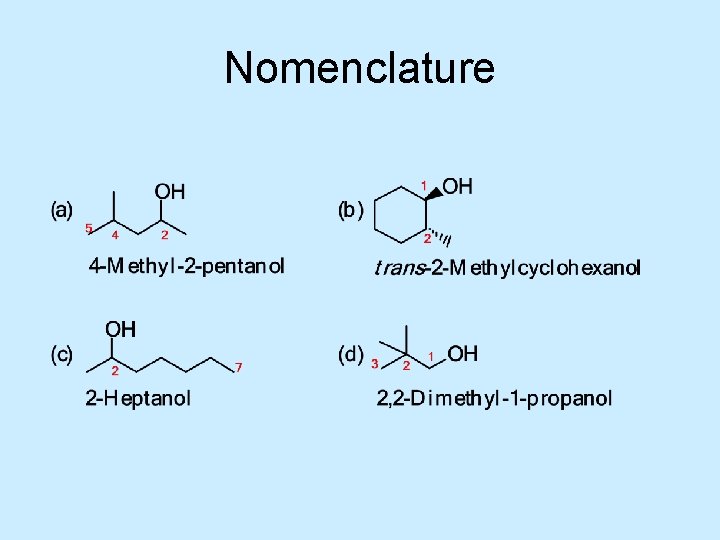
Nomenclature
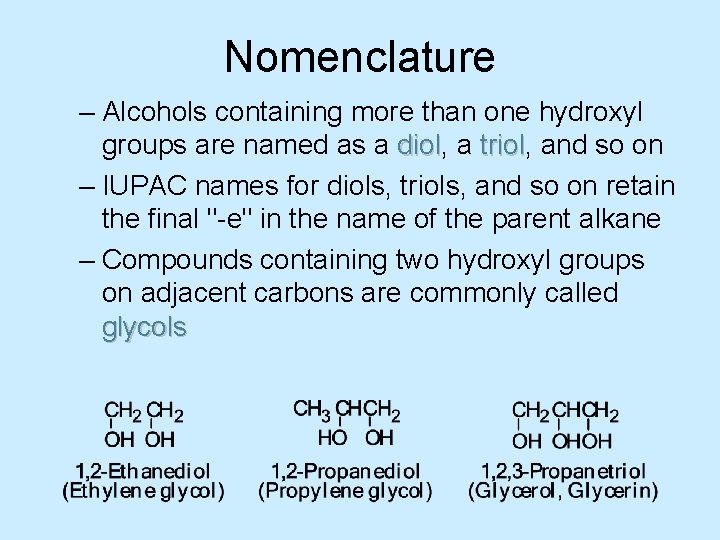
Nomenclature – Alcohols containing more than one hydroxyl groups are named as a diol, diol a triol, triol and so on – IUPAC names for diols, triols, and so on retain the final "-e" in the name of the parent alkane – Compounds containing two hydroxyl groups on adjacent carbons are commonly called glycols
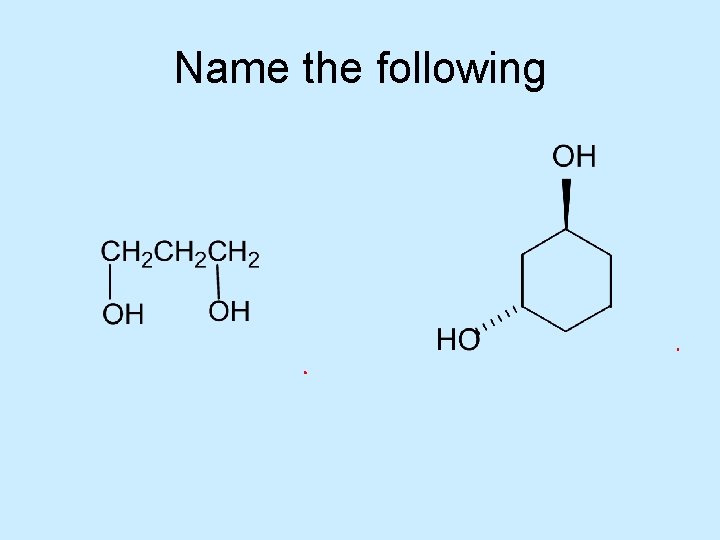
Name the following
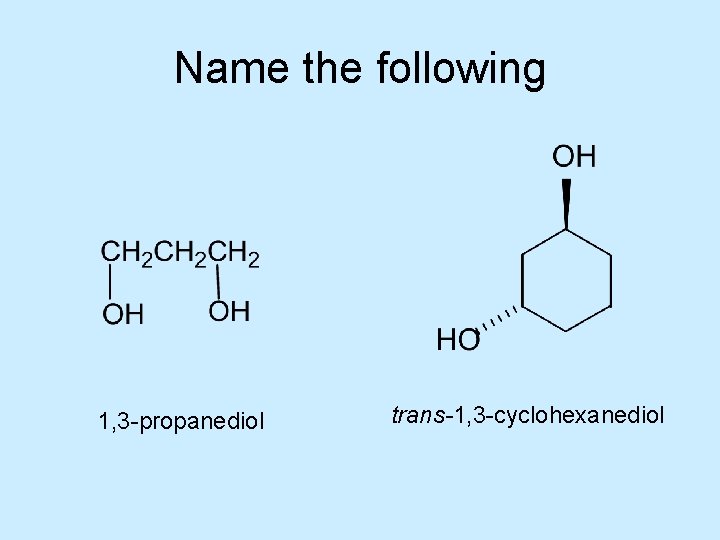
Name the following 1, 3 -propanediol trans-1, 3 -cyclohexanediol
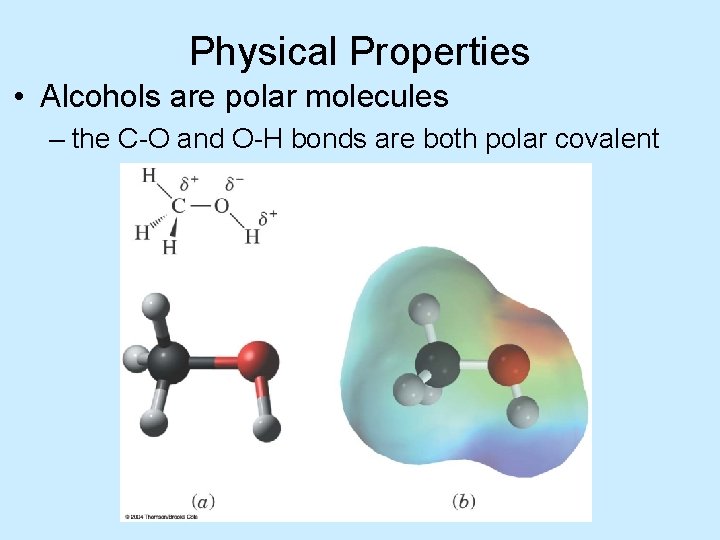
Physical Properties • Alcohols are polar molecules – the C-O and O-H bonds are both polar covalent
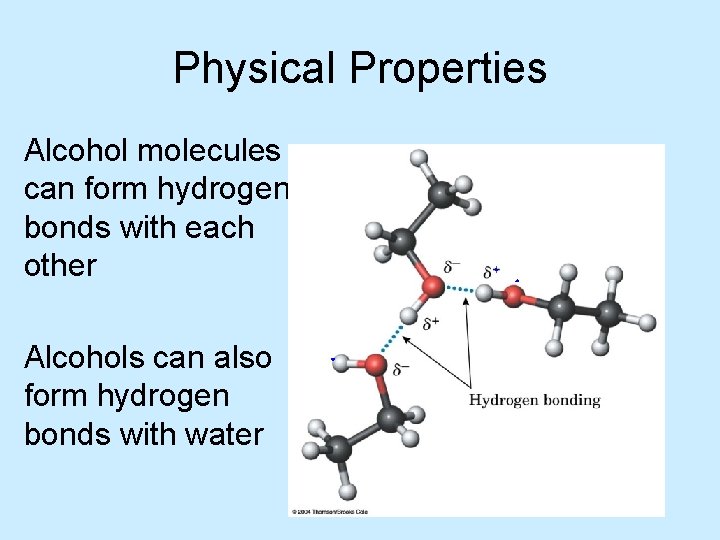
Physical Properties Alcohol molecules can form hydrogen bonds with each other Alcohols can also form hydrogen bonds with water
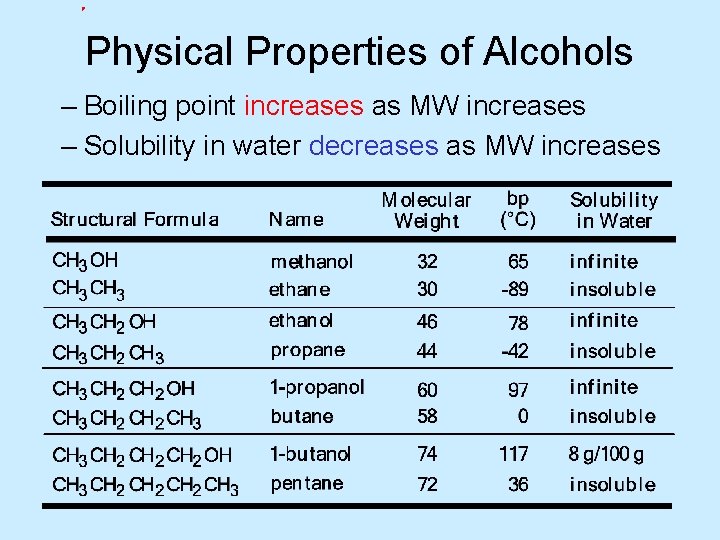
Physical Properties of Alcohols – Boiling point increases as MW increases – Solubility in water decreases as MW increases

Chemical Properties of Alcohols • Combustion Reactions (they burn in O 2) • Acid-Base Chemistry • Dehydration • Oxidation

Acidity of Alcohols • Alcohols have about the same p. Ka values as water – Aqueous solutions of alcohols have the same p. H as that of pure water – Phenols have lower p. Ka values than normal alcohols do (are more acidic)
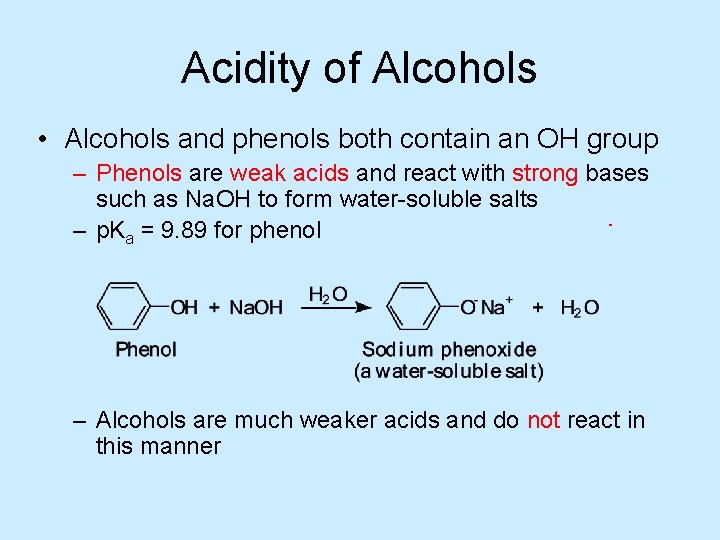
Acidity of Alcohols • Alcohols and phenols both contain an OH group – Phenols are weak acids and react with strong bases such as Na. OH to form water-soluble salts – p. Ka = 9. 89 for phenol – Alcohols are much weaker acids and do not react in this manner
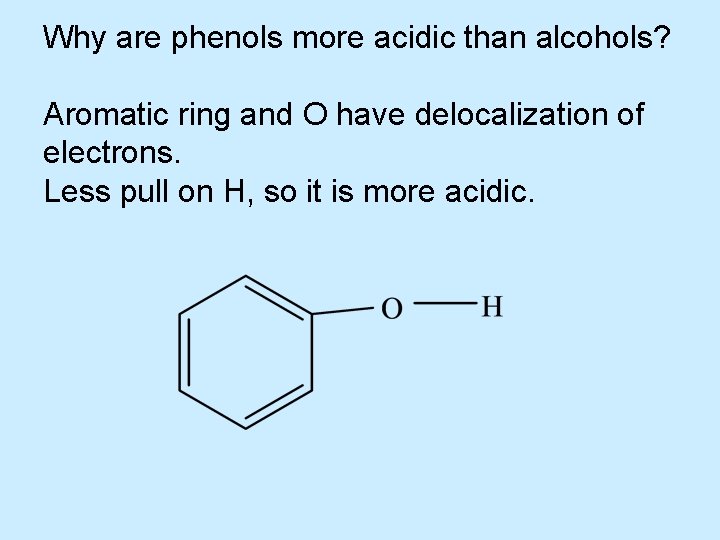
Why are phenols more acidic than alcohols? Aromatic ring and O have delocalization of electrons. Less pull on H, so it is more acidic.
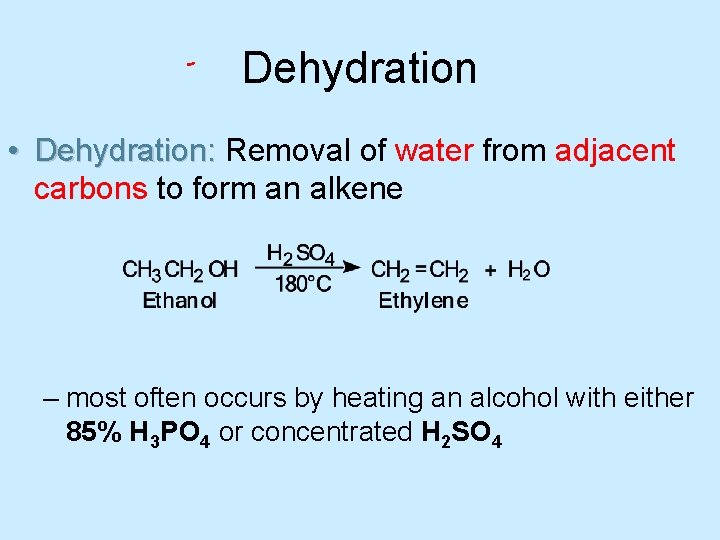
Dehydration • Dehydration: Removal of water from adjacent carbons to form an alkene – most often occurs by heating an alcohol with either 85% H 3 PO 4 or concentrated H 2 SO 4

• Dehydration essentially the reverse of hydration (addition of water to double bond) • Conditions required depend on kind of alcohol
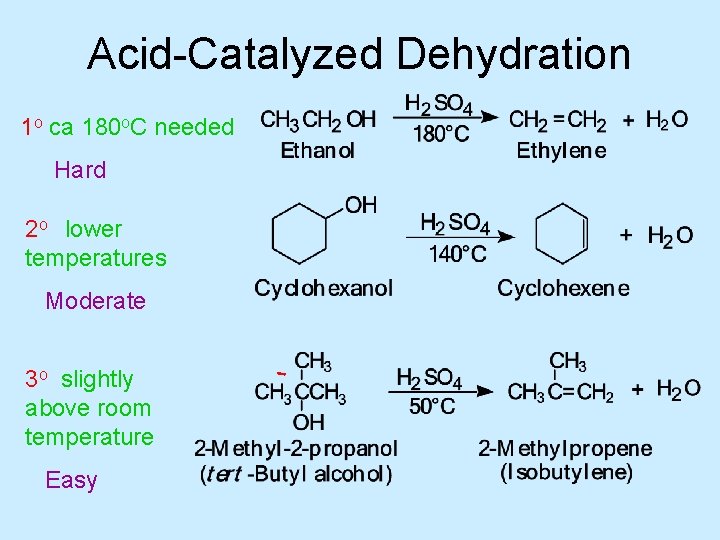
Acid-Catalyzed Dehydration 1 o ca 180 o. C needed Hard 2 o lower temperatures Moderate 3 o slightly above room temperature Easy
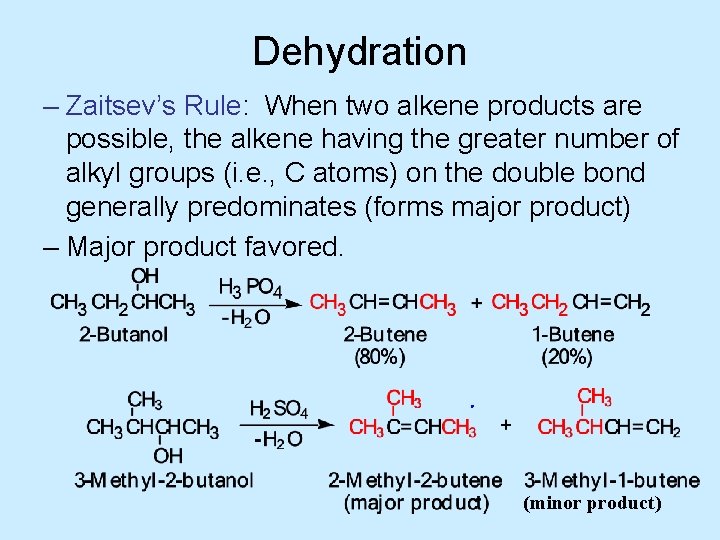
Dehydration – Zaitsev’s Rule: When two alkene products are possible, the alkene having the greater number of alkyl groups (i. e. , C atoms) on the double bond generally predominates (forms major product) – Major product favored. (minor product)
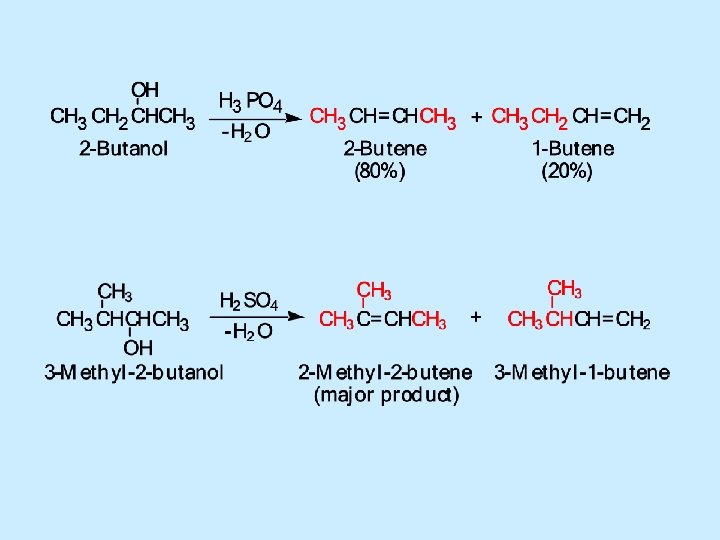

Dehydration-Hydration Related • Acid-catalyzed hydration of alkenes and acidcatalyzed dehydration of alcohols are competing reactions • Equilibrium between alkene and alcohol exists – Large amounts of water favor alcohol formation, while removal of water from the equilibrium mixture favors alkene formation (Le Chatelier's principle)

Dehydration vs Oxidation • Dehydration: Remove –OH and –H – OH from one C: H from adjacent C • Oxidation: Remove two –H atoms – One H from OH group: Other H from C that has the –OH attached

Oxidation of Alcohols • Oxidation of a 1° alcohol gives an aldehyde or a carboxylic acid, depending on the experimental conditions – Oxidation of a 1° alcohol to a carboxylic acid is carried out using an oxidizing agent such as potassium dichromate, K 2 Cr 2 O 7, in aqueous sulfuric acid
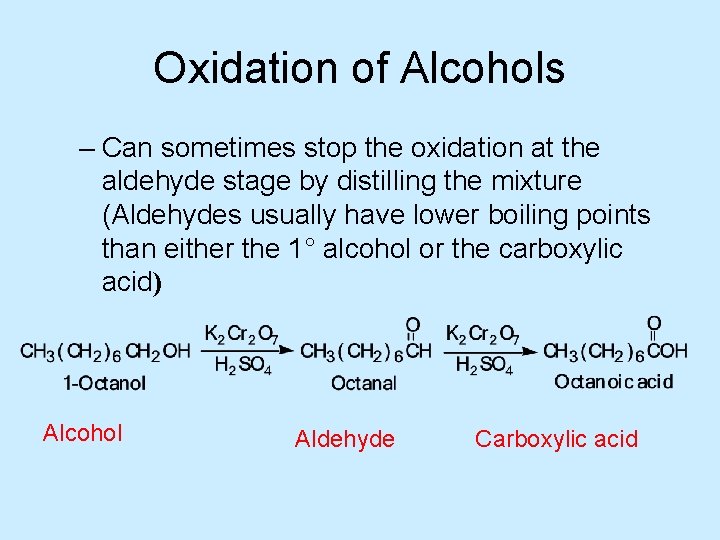
Oxidation of Alcohols – Can sometimes stop the oxidation at the aldehyde stage by distilling the mixture (Aldehydes usually have lower boiling points than either the 1° alcohol or the carboxylic acid) Alcohol Aldehyde Carboxylic acid
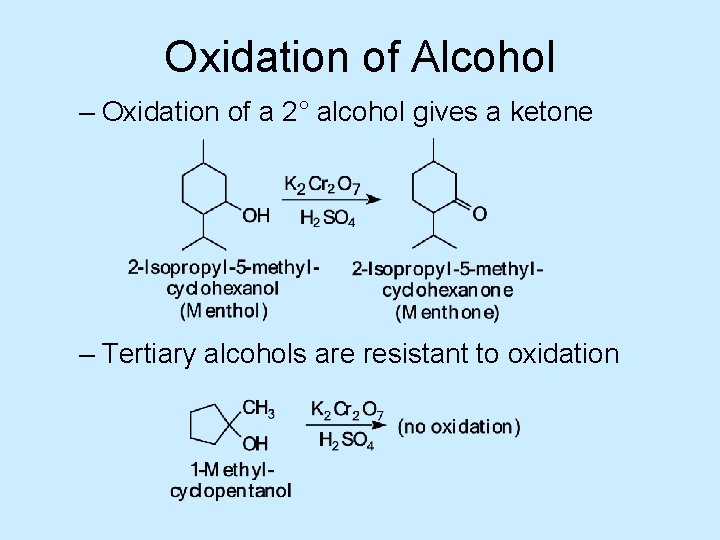
Oxidation of Alcohol – Oxidation of a 2° alcohol gives a ketone – Tertiary alcohols are resistant to oxidation

Oxidation of Alcohols • Draw a structural formula for the product formed by oxidation of each of the following alcohols with potassium dichromate. • a) 1 -Hexanol b) 2 -Hexanol • Strategy • Oxidation of 1 -hexanol, a primary alcohol, gives either an aldehyde or a carboxylic acid, depending on the experimental conditions. Oxidation of 2 -hexanol, a secondary alcohol, gives a ketone.
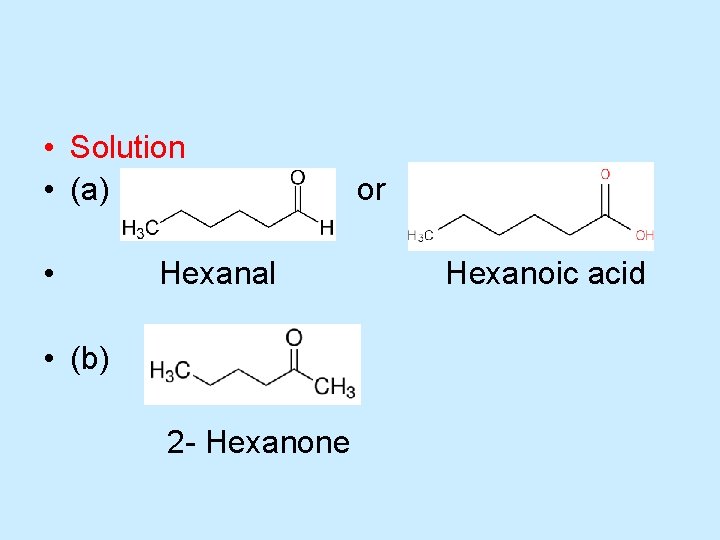
• Solution • (a) • Hexanal • (b) 2 - Hexanone or Hexanoic acid

Common Alcohols • Ethanol – Ethyl alcohol, “grain alcohol” – Produced by fermentation – Natural fermentation of sugars and starches in grains, corn, molasses, fruits, vegetables – Found in beer, wine, and other “spirits” C 6 H 12 O 6 → 2 CH 3 CH 2 OH + 2 CO 2 glucose ethanol carbon dioxide

Common Alcohols • Methanol – Methyl alcohol or “wood alcohol” – Distilled from wood pulp originally – Very toxic, even in small amounts – Reacts to make formaldehyde (a fixative) CH 3 OH methanol → CH 2=O formaldehyde

Common Alcohols • Isopropyl alcohol – 2 -propanol “rubbing alcohol” – Evaporates quickly—used to reduce fever – Solvent in many cosmetics, perfumes, etc.

Common Alcohols • 1, 2 -ethanediol – Ethylene glycol – Very soluble in water – Used as antifreeze • Glycerol – – 1, 2, 3 -propanetriol Very soluble in water Sweet taste Part of fats and oils
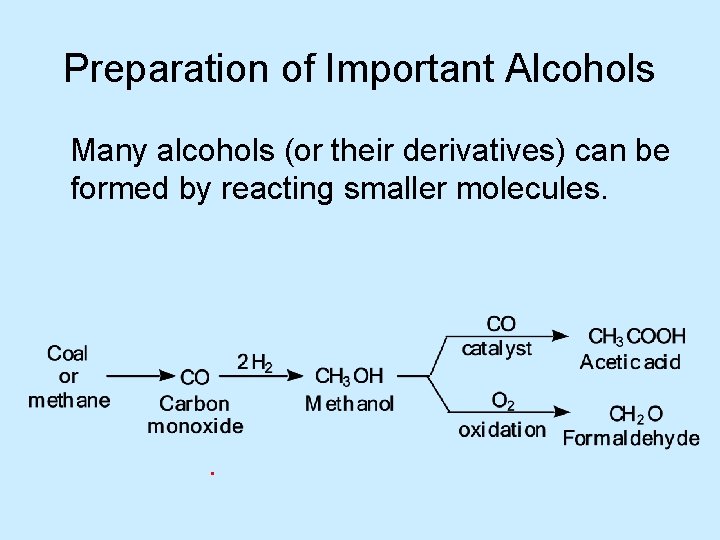
Preparation of Important Alcohols Many alcohols (or their derivatives) can be formed by reacting smaller molecules.
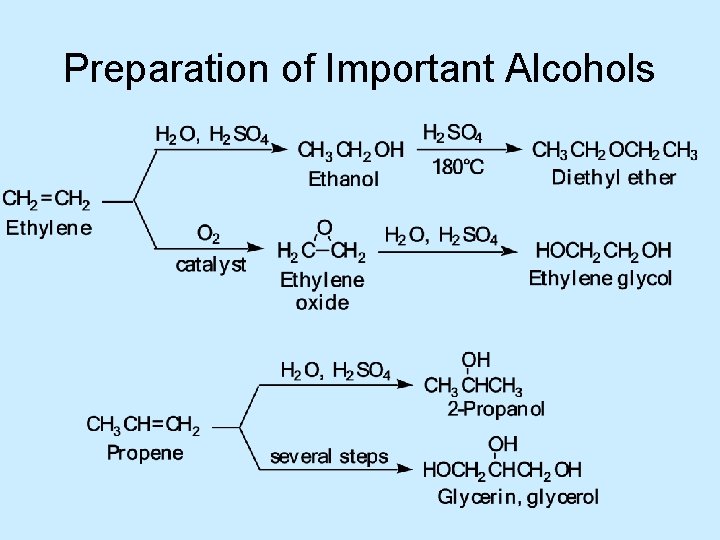
Preparation of Important Alcohols
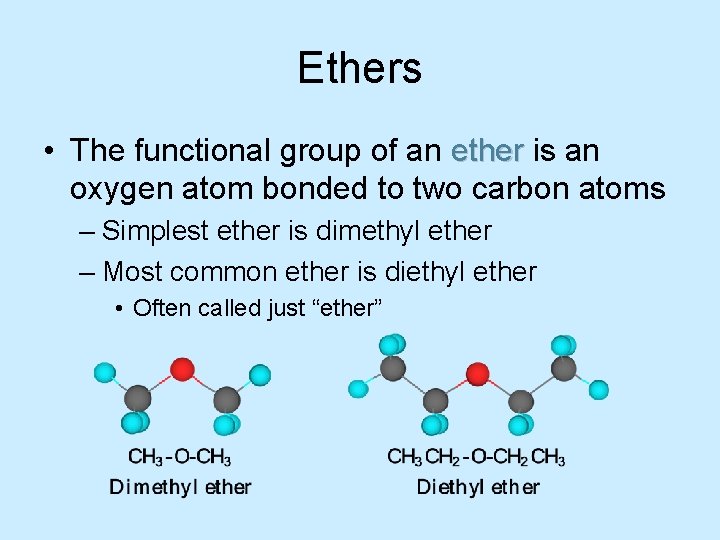
Ethers • The functional group of an ether is an oxygen atom bonded to two carbon atoms – Simplest ether is dimethyl ether – Most common ether is diethyl ether • Often called just “ether”
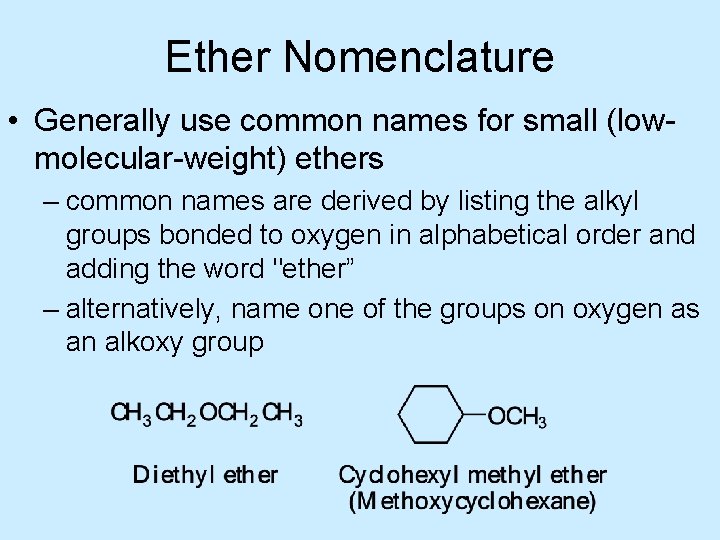
Ether Nomenclature • Generally use common names for small (lowmolecular-weight) ethers – common names are derived by listing the alkyl groups bonded to oxygen in alphabetical order and adding the word "ether” – alternatively, name one of the groups on oxygen as an alkoxy group

Ether Nomenclature • Cyclic ether: an ether in which one of the atoms in a ring is oxygen – Cyclic ethers also known by their common names – ethylene oxide is an important building block for the organic chemical industry; it is also used as a fumigant in foodstuffs and textiles, and in hospitals to sterilize surgical instruments – tetrahydrofuran is a useful laboratory and industrial solvent

Physical Properties of Ethers • Ethers are polar compounds – O has a partial negative charge – Each C bonded to it has a partial positive charge • Only weak forces of attraction exist between ether molecules in the pure liquid • Boiling points of ethers are close to those of hydrocarbons of similar molecular weight • Have lower boiling points than alcohols of the same molecular formula

Reactions of Ethers • Ethers not very reactive (like hydrocarbons) – Do not react with oxidizing agents such as potassium dichromate – Do not react with reducing agents such as H 2 in the presence of a transition metal catalyst – Not affected by most acids or bases at moderate temperatures • Good solvents for carrying out organic reactions, because of their general inertness and good solvent properties – Diethyl ether and THF (tetrahydrofuran)
Thiols • Thiol: a compound containing an -SH (sulfhydryl) group – Also called mercaptans – Low-molecular-weight thiols STINK – Responsible for smells such as those from rotten eggs and sewage – the scent of skunks is due primarily to these two thiols

Thiols - Nomenclature • IUPAC names are derived in the same manner as are the names of alcohols – Retain the final -e of the parent alkane and add the suffix -thiol – Common names for simple thiols are derived by naming the alkyl group bonded to -SH and adding the word "mercaptan" mercaptan

Thiol Physical Properties • S-H bonds are nonpolar covalent – Small difference in electronegativity between sulfur and hydrogen (2. 5 - 2. 1 = 0. 4) – No hydrogen bonding – Lower boiling points than comparable alcohols – Less soluble in water and other polar solvents than comparable alcohols

Reactions of Thiols • Thiols are weak acids (p. Ka~10), and are comparable in strength to phenols – thiols react with strong bases such as Na. OH to form water-soluble thiolate salts

Reactions of Thiols • The most important thiol reaction in biological systems is their oxidation to disulfides – Functional group is a disulfide (-S-S-) bond • Disulfide bonds important in protein structure – Thiols are readily oxidized to disulfides by O 2 – -SH group very susceptible to oxidation and must be protected from contact with air during storage – disulfides, in turn, are easily reduced to thiols by several reducing agents.

Reactions of Thiols Oxidation of –SH groups to form S-S bonds Reduction of S-S bonds to form –SH groups
- Slides: 47