Synthesis of 2 Alcohols Grignard aldehyde yields a




- Slides: 18

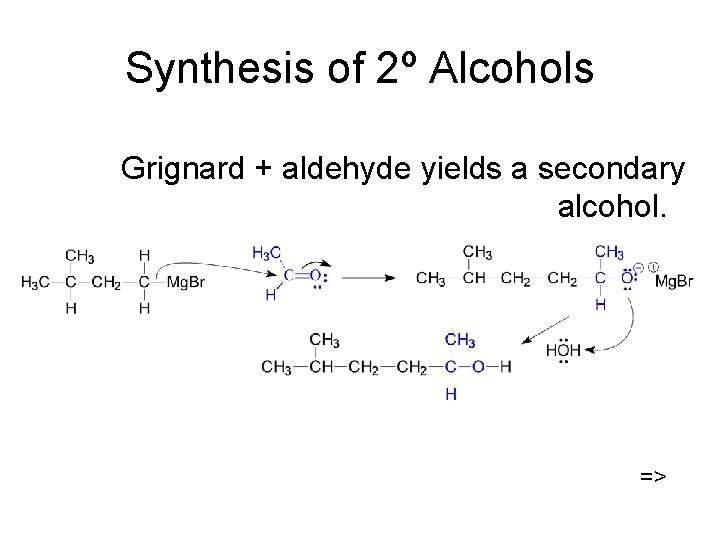
Synthesis of 2º Alcohols Grignard + aldehyde yields a secondary alcohol. =>

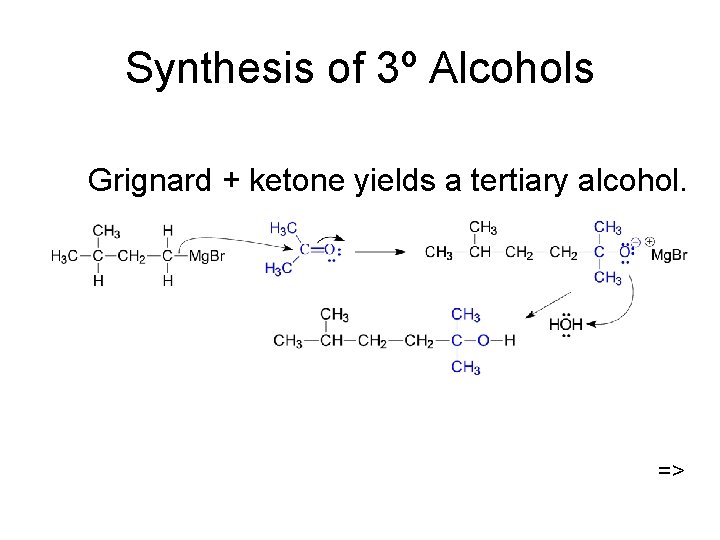
Synthesis of 3º Alcohols Grignard + ketone yields a tertiary alcohol. =>

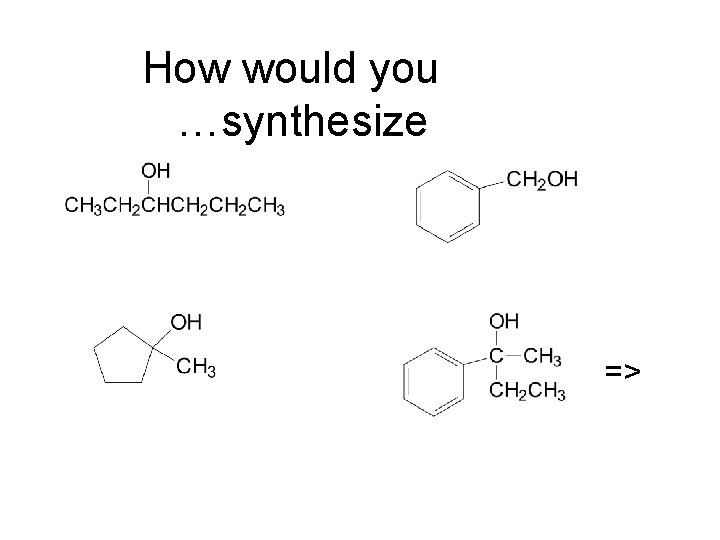
How would you …synthesize =>

Grignard Reactions with Acid Chlorides and Esters • Use two moles of Grignard reagent. • The product is a tertiary alcohol with two identical alkyl groups. • Reaction with one mole of Grignard reagent produces a ketone intermediate, which reacts with the second mole of Grignard reagent. =>

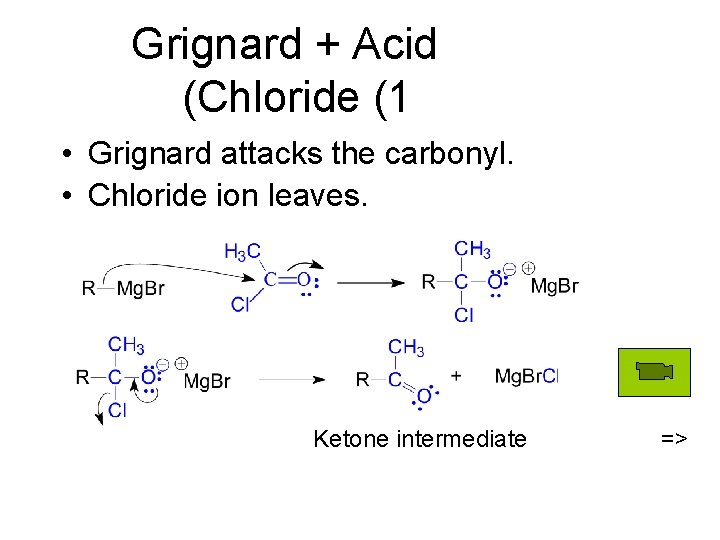
Grignard + Acid (Chloride (1 • Grignard attacks the carbonyl. • Chloride ion leaves. Ketone intermediate =>

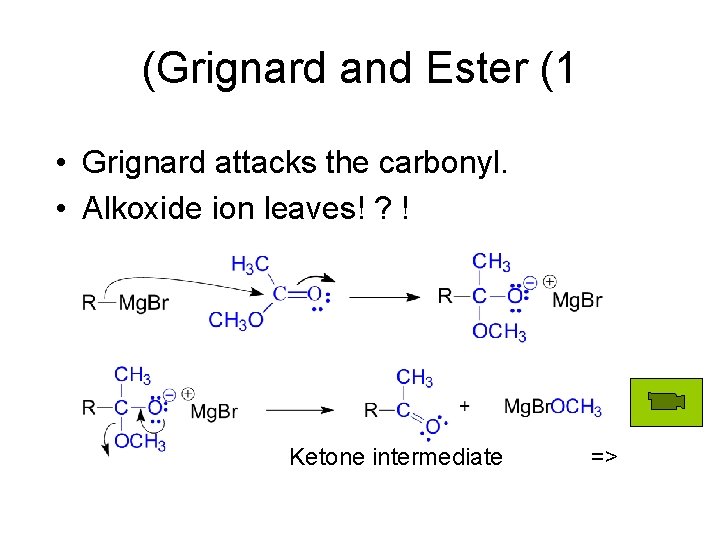
(Grignard and Ester (1 • Grignard attacks the carbonyl. • Alkoxide ion leaves! ? ! Ketone intermediate =>

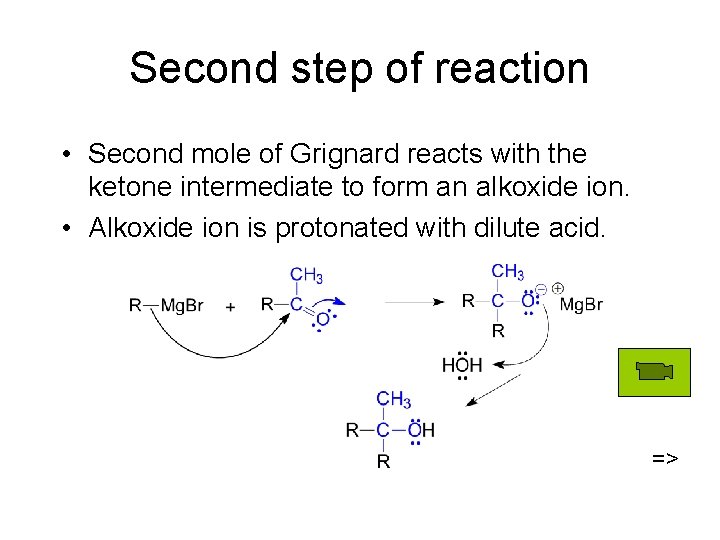
Second step of reaction • Second mole of Grignard reacts with the ketone intermediate to form an alkoxide ion. • Alkoxide ion is protonated with dilute acid. =>

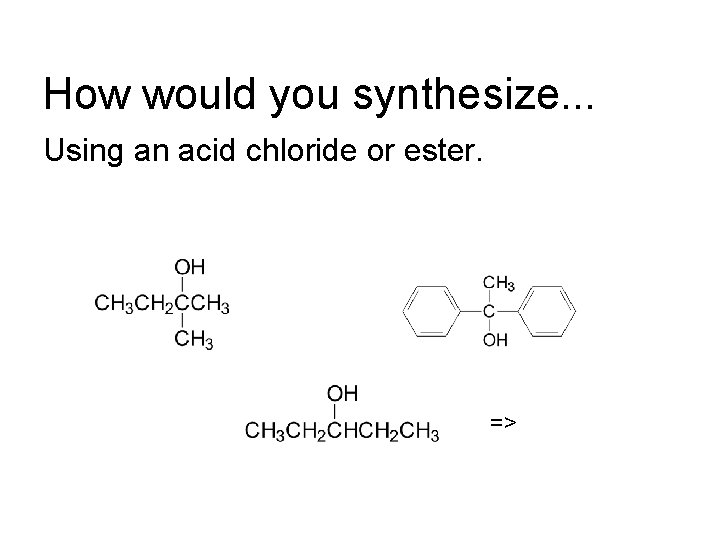
How would you synthesize. . . Using an acid chloride or ester. =>

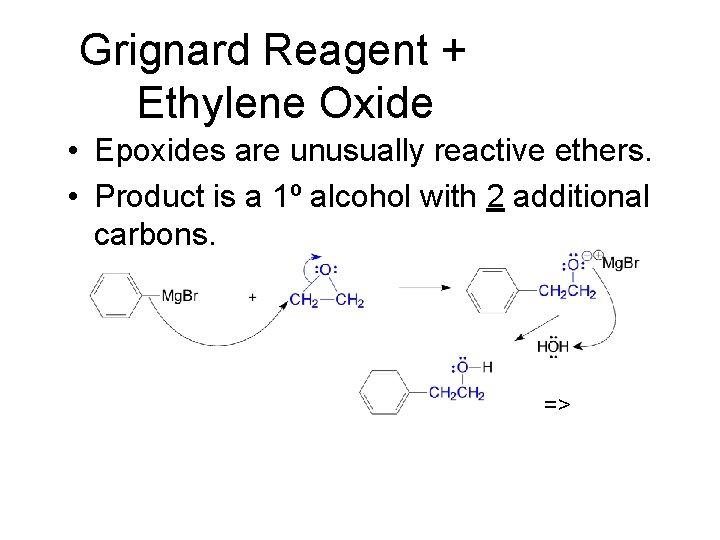
Grignard Reagent + Ethylene Oxide • Epoxides are unusually reactive ethers. • Product is a 1º alcohol with 2 additional carbons. =>

Limitations of Grignard • No water or other acidic protons like O-H, N-H, S-H, or -C—C-H. Grignard reagent is destroyed, becomes an alkane. • No other electrophilic multiple bonds, like C=N, C—N, S=O, or N=O. =>

Reduction of Carbonyl • Reduction of aldehyde yields 1º alcohol. • Reduction of ketone yields 2º alcohol. • Reagents: – Sodium borohydride, Na. BH 4 – Lithium aluminum hydride, Li. Al. H 4 – Raney nickel =>

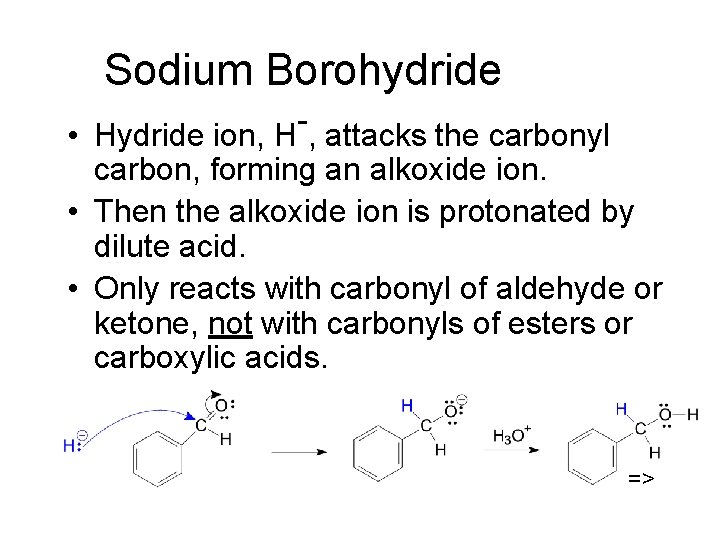
Sodium Borohydride • Hydride ion, H , attacks the carbonyl carbon, forming an alkoxide ion. • Then the alkoxide ion is protonated by dilute acid. • Only reacts with carbonyl of aldehyde or ketone, not with carbonyls of esters or carboxylic acids. =>

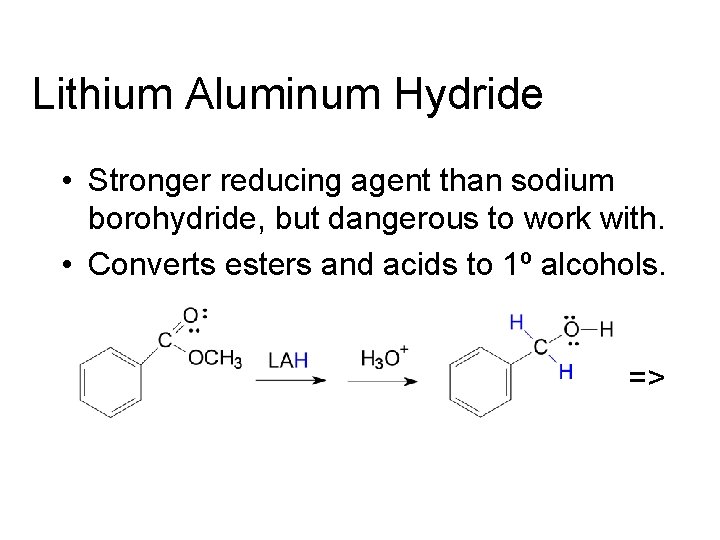
Lithium Aluminum Hydride • Stronger reducing agent than sodium borohydride, but dangerous to work with. • Converts esters and acids to 1º alcohols. =>

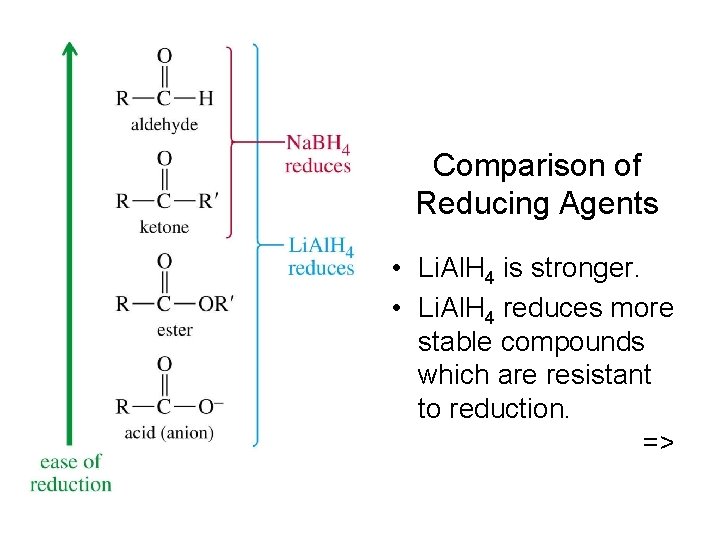
Comparison of Reducing Agents • Li. Al. H 4 is stronger. • Li. Al. H 4 reduces more stable compounds which are resistant to reduction. =>

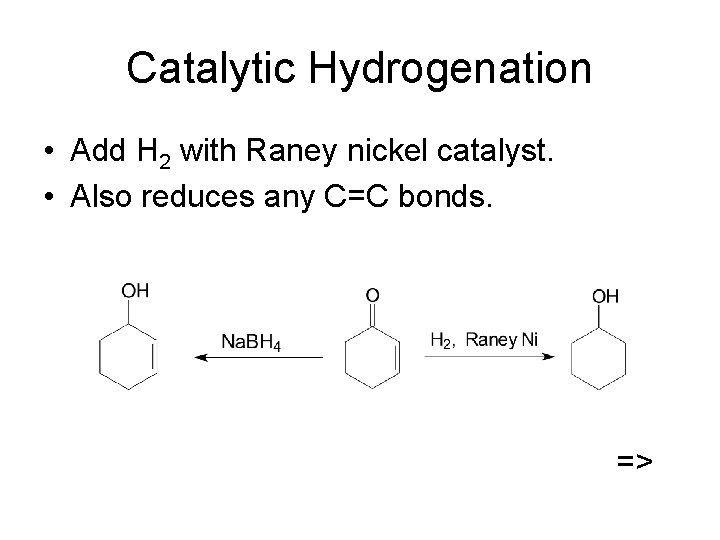
Catalytic Hydrogenation • Add H 2 with Raney nickel catalyst. • Also reduces any C=C bonds. =>

(Thiols (Mercaptans • • • Sulfur analogues of alcohols, -SH. Named by adding -thiol to alkane name. The -SH group is called mercapto. Complex with heavy metals: Hg, As, Au. More acidic than alcohols, react with Na. OH to form thiolate ion. Stinks! => •

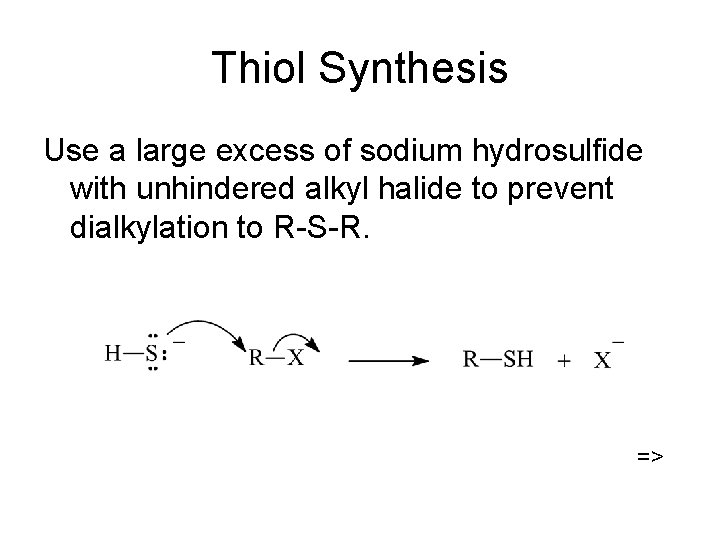
Thiol Synthesis Use a large excess of sodium hydrosulfide with unhindered alkyl halide to prevent dialkylation to R-S-R. =>

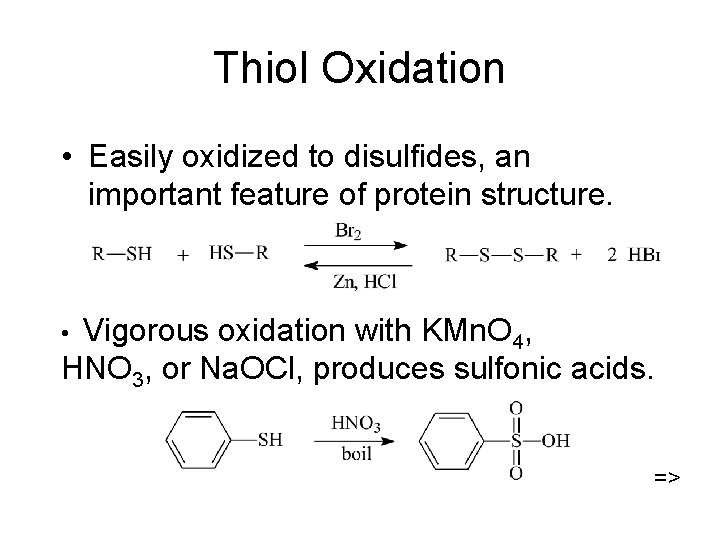
Thiol Oxidation • Easily oxidized to disulfides, an important feature of protein structure. Vigorous oxidation with KMn. O 4, HNO 3, or Na. OCl, produces sulfonic acids. • =>