ALCOHOLS and PHENOLS Classification of hydroxylic compounds sp






















- Slides: 56

ALCOHOLS and PHENOLS

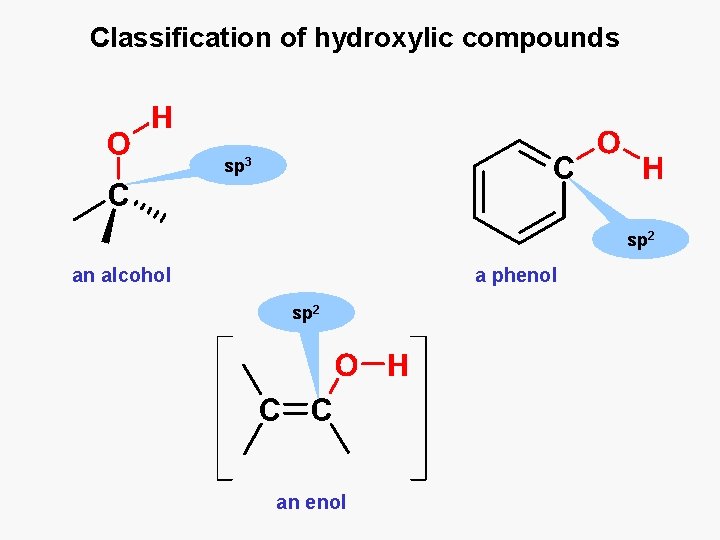
Classification of hydroxylic compounds sp 3 sp 2 an alcohol a phenol sp 2 an enol

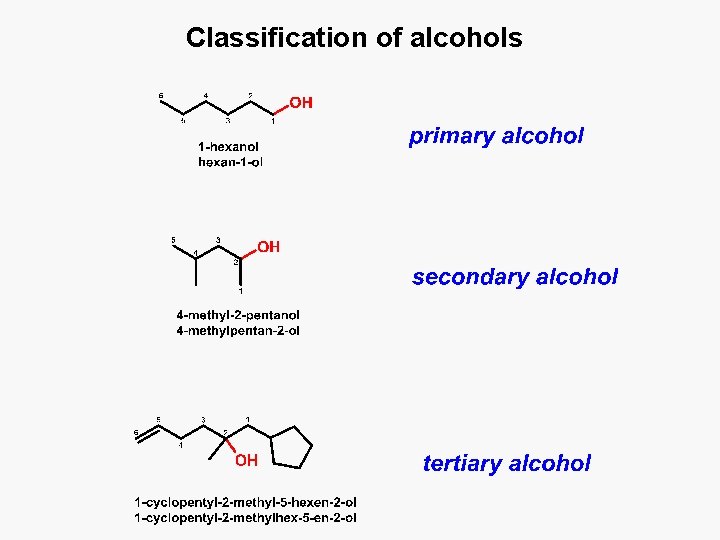
Classification of alcohols

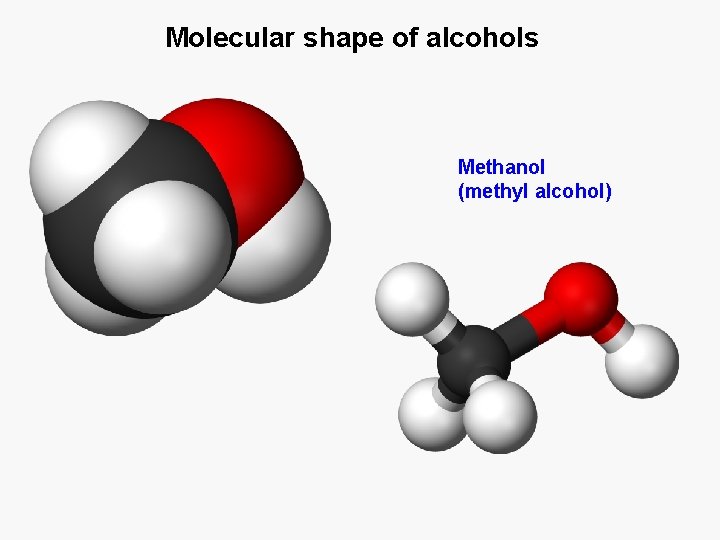
Molecular shape of alcohols Methanol (methyl alcohol)

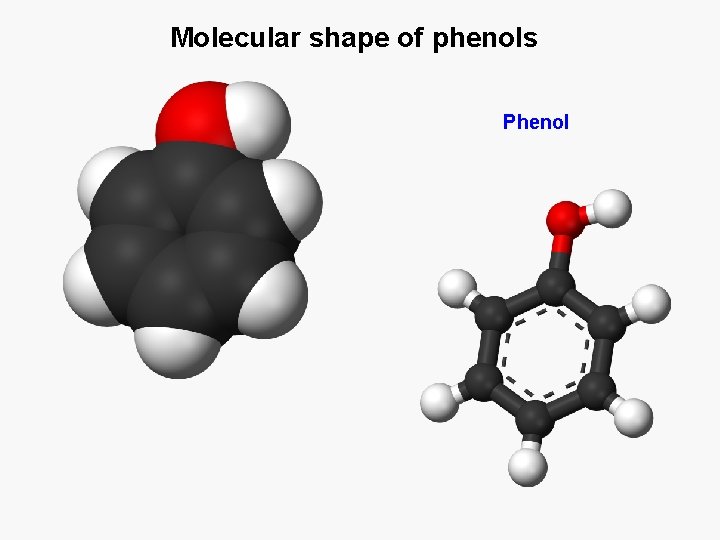
Molecular shape of phenols Phenol

Properties of hydroxyl group • Polarity • Hydrogen bonding • Solubility in water • Basicity • Acidity

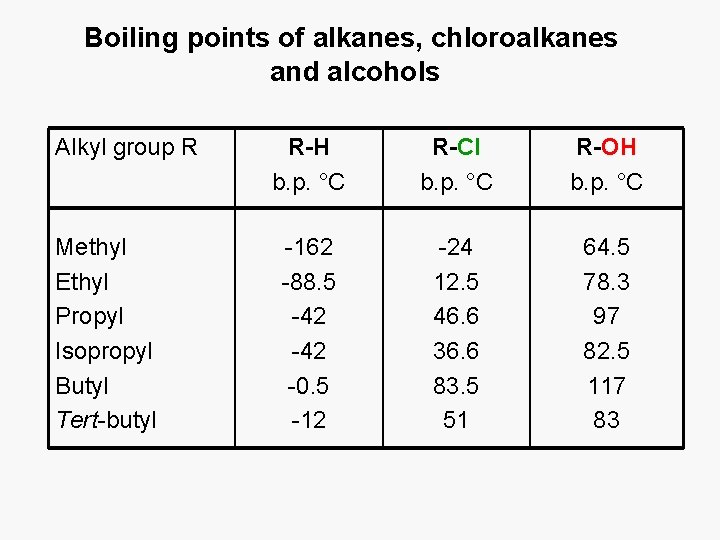
Boiling points of alkanes, chloroalkanes and alcohols Alkyl group R Methyl Ethyl Propyl Isopropyl Butyl Tert-butyl R-H b. p. °C R-Cl b. p. °C R-OH b. p. °C -162 -88. 5 -42 -0. 5 -12 -24 12. 5 46. 6 36. 6 83. 5 51 64. 5 78. 3 97 82. 5 117 83

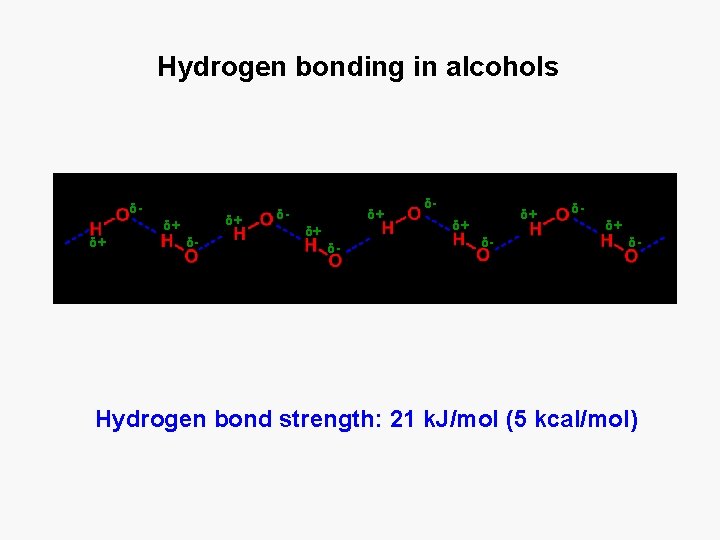
Hydrogen bonding in alcohols δ- δ+ δ+ δ+ δ- δ- δ+ δ+ δ- δδ+ δ- Hydrogen bond strength: 21 k. J/mol (5 kcal/mol)

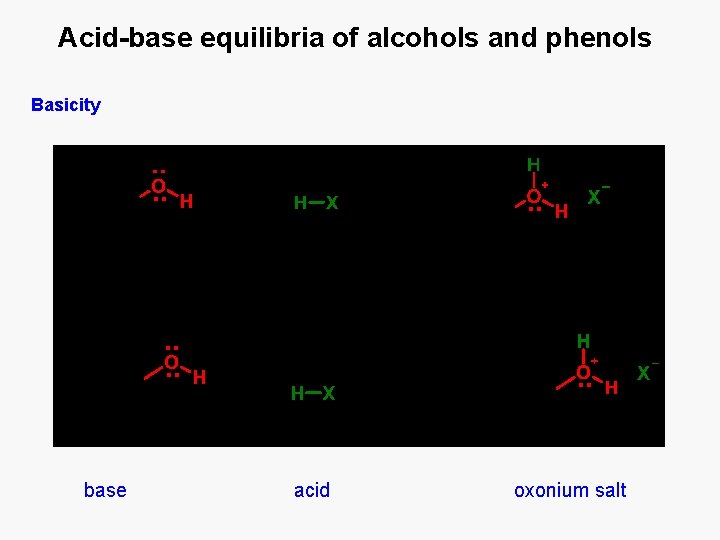
Acid-base equilibria of alcohols and phenols Basicity base acid oxonium salt

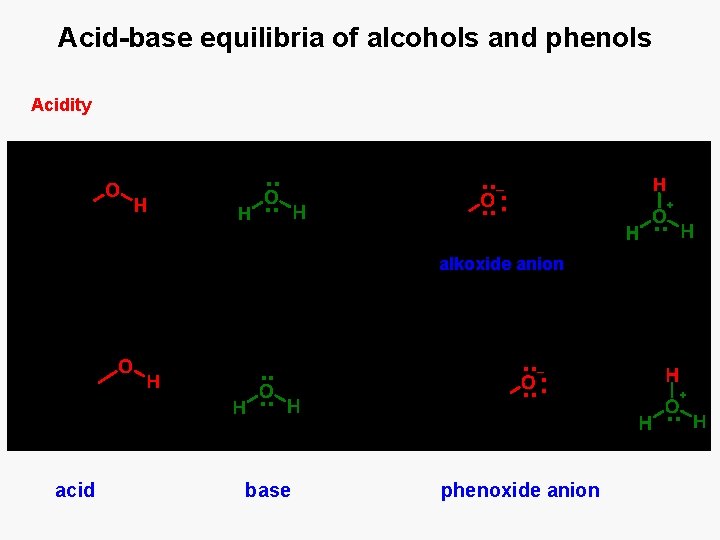
Acid-base equilibria of alcohols and phenols Acidity alkoxide anion acid base phenoxide anion

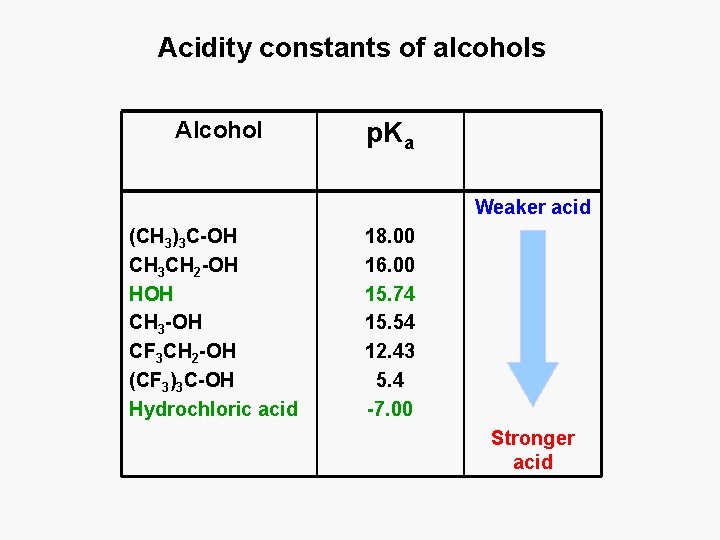
Acidity constants of alcohols Alcohol p. Ka Weaker acid (CH 3)3 C-OH CH 3 CH 2 -OH HOH CH 3 -OH CF 3 CH 2 -OH (CF 3)3 C-OH Hydrochloric acid 18. 00 16. 00 15. 74 15. 54 12. 43 5. 4 -7. 00 Stronger acid

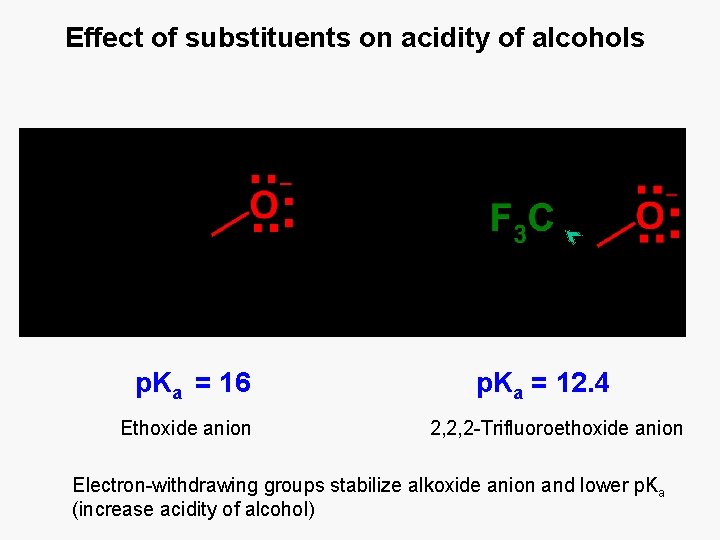
Effect of substituents on acidity of alcohols p. Ka = 16 Ethoxide anion p. Ka = 12. 4 2, 2, 2 -Trifluoroethoxide anion Electron-withdrawing groups stabilize alkoxide anion and lower p. Ka (increase acidity of alcohol)

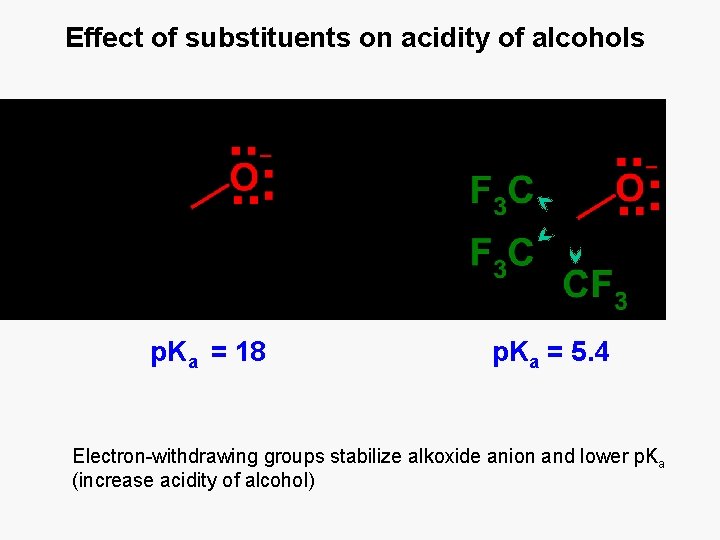
Effect of substituents on acidity of alcohols p. Ka = 18 p. Ka = 5. 4 Electron-withdrawing groups stabilize alkoxide anion and lower p. Ka (increase acidity of alcohol)

Effect of anion size on acidity of alcohols p. Ka = 15. 58 Small anion, sterically accessible Easy solvation Weaker basicity of anion Greater acidity of alcohol p. Ka = 18. 00 Large anion, sterically hindered Difficult solvation Stronger basicity of anion Lower acidity of alcohol

Acidity of phenols vs alcohols Phenols are stronger acids than alcohols Because of resonance stabilization of phenolate anion:

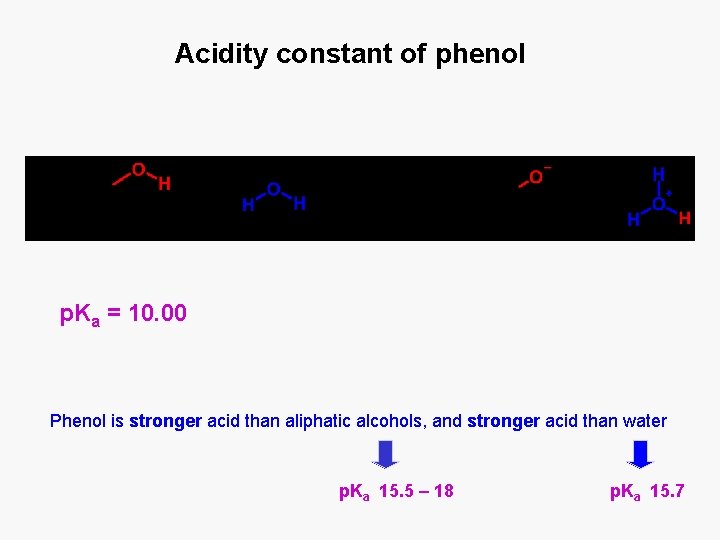
Acidity constant of phenol p. Ka = 10. 00 Phenol is stronger acid than aliphatic alcohols, and stronger acid than water p. Ka 15. 5 – 18 p. Ka 15. 7

Acidity constants of some phenols Phenol p. Ka Weaker acid p-aminophenol p-methoxyphenol p-methylphenol p-chlorophenol p-nitrophenol 2, 4, 6 -trinitrophenol 10. 46 10. 21 10. 17 10. 00 9. 38 7. 15 0. 60 HOH Hydrochloric acid 15. 74 -7. 00 Stronger acid

Effect of substituents on acidity of phenols EWG –Electron Withdrawing Group EDG –Electron Donating Group Phenoxide anion stabilized by substituent Phenoxide anion destabilized by substituent Greater acidity of phenol Lower acidity of phenol p. Ka < 10 p. Ka > 10 EWG: -NO 2, -COOH, -CN, -Cl, -Br, EDG: -OCH 3, -NH 2, alkyl (CH 3)

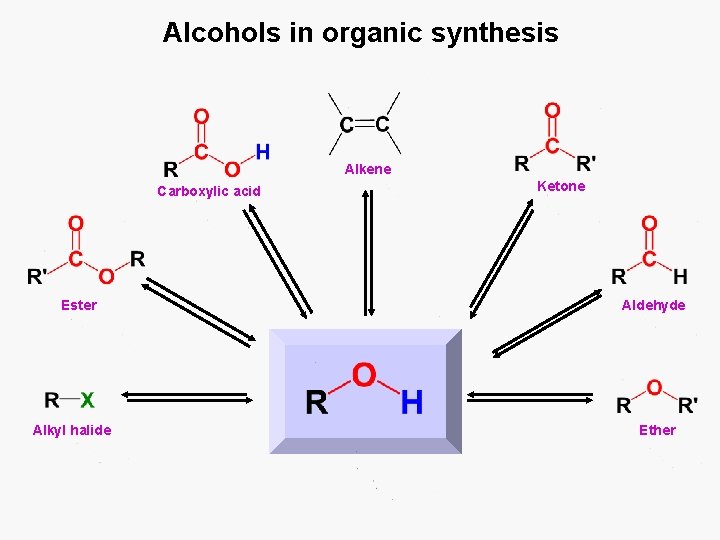
Alcohols in organic synthesis Alkene Carboxylic acid Ester Alkyl halide Ketone Aldehyde Ether

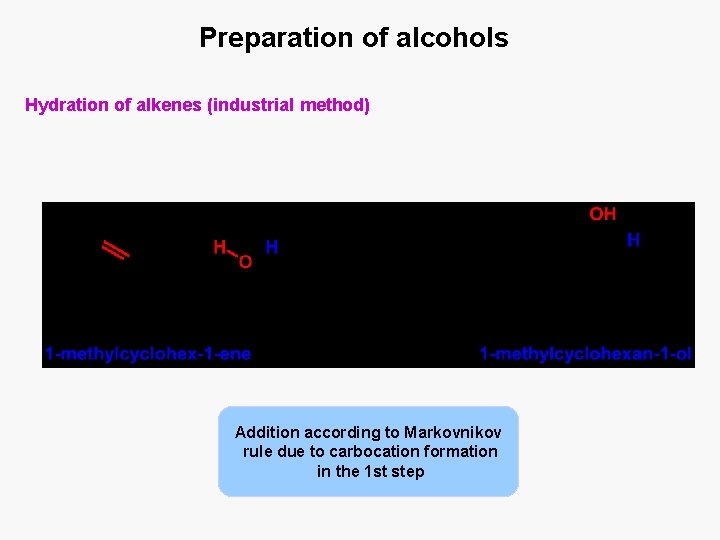
Preparation of alcohols Hydration of alkenes (industrial method) Addition according to Markovnikov rule due to carbocation formation in the 1 st step

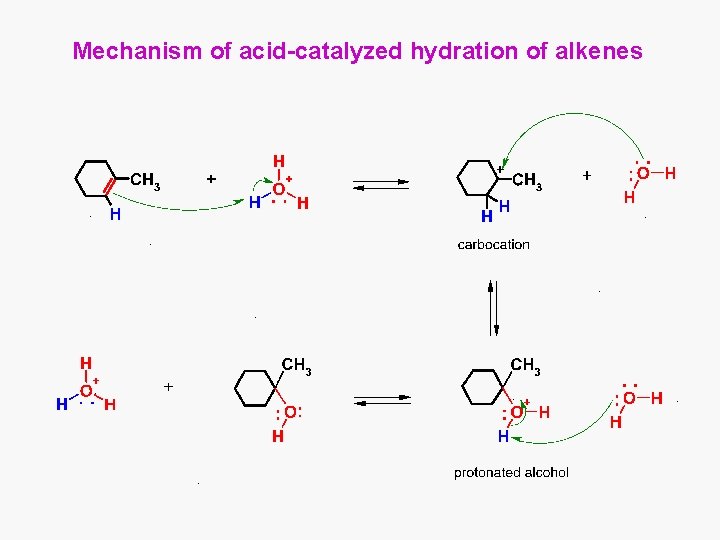
Mechanism of acid-catalyzed hydration of alkenes

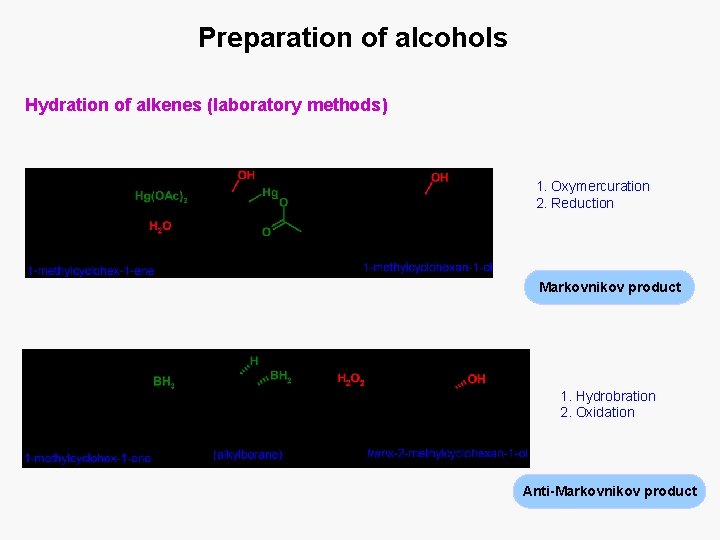
Preparation of alcohols Hydration of alkenes (laboratory methods) 1. Oxymercuration 2. Reduction Markovnikov product 1. Hydrobration 2. Oxidation Anti-Markovnikov product

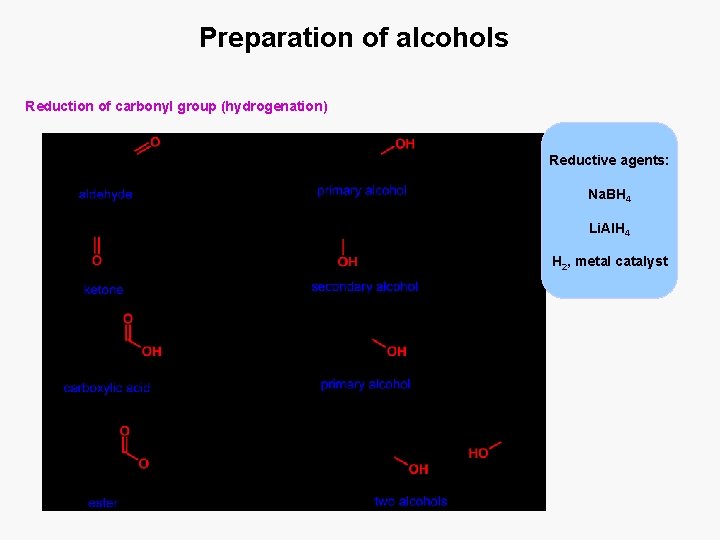
Preparation of alcohols Reduction of carbonyl group (hydrogenation) Reductive agents: Na. BH 4 Li. Al. H 4 H 2, metal catalyst

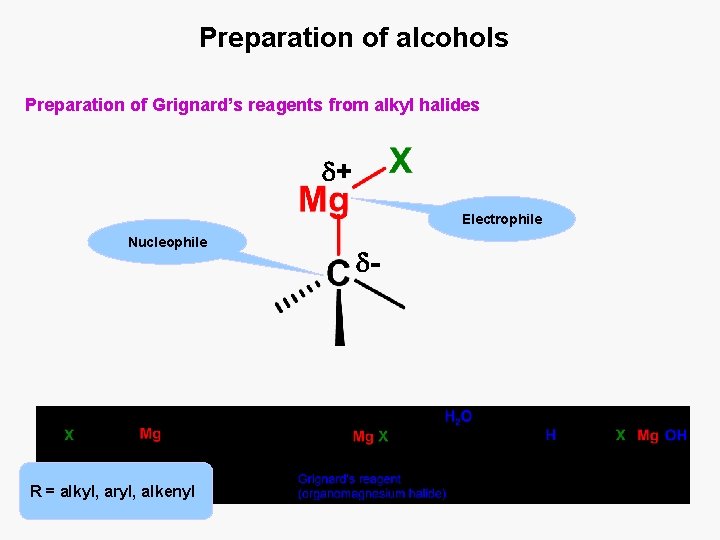
Preparation of alcohols Preparation of Grignard’s reagents from alkyl halides + Electrophile Nucleophile R = alkyl, aryl, alkenyl -

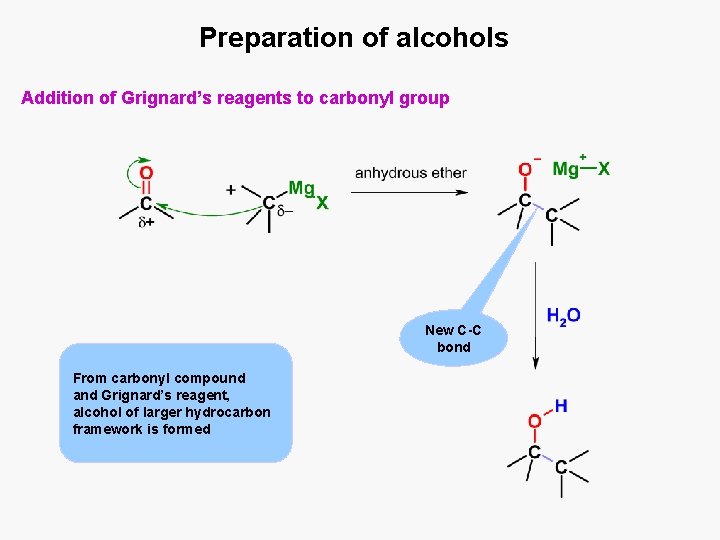
Preparation of alcohols Addition of Grignard’s reagents to carbonyl group New C-C bond From carbonyl compound and Grignard’s reagent, alcohol of larger hydrocarbon framework is formed

Preparation of alcohols Addition of Grignard’s reagent to FORMALDEHYDE Primary alcohol is formed

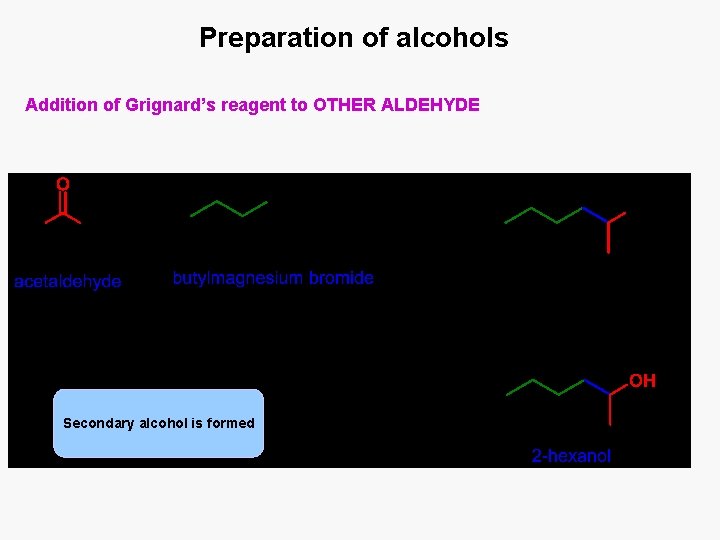
Preparation of alcohols Addition of Grignard’s reagent to OTHER ALDEHYDE Secondary alcohol is formed

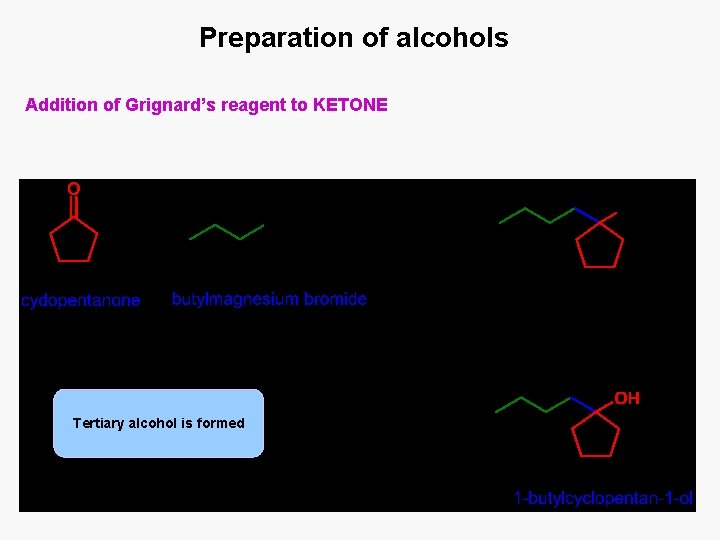
Preparation of alcohols Addition of Grignard’s reagent to KETONE Tertiary alcohol is formed

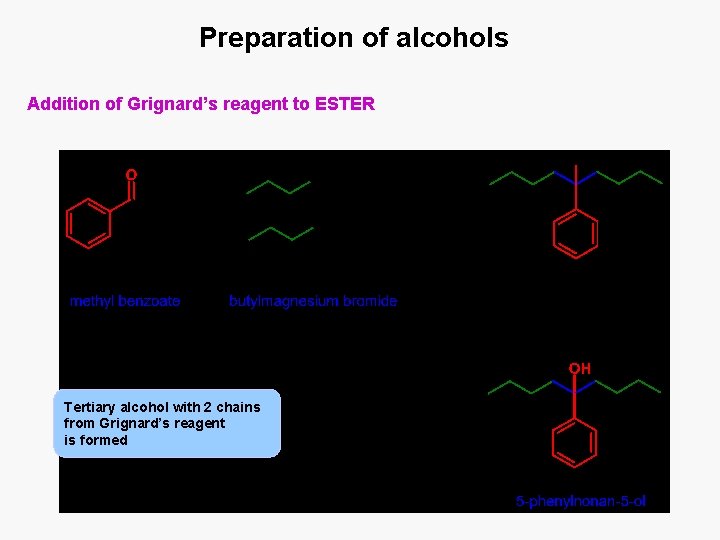
Preparation of alcohols Addition of Grignard’s reagent to ESTER Tertiary alcohol with 2 chains from Grignard’s reagent is formed

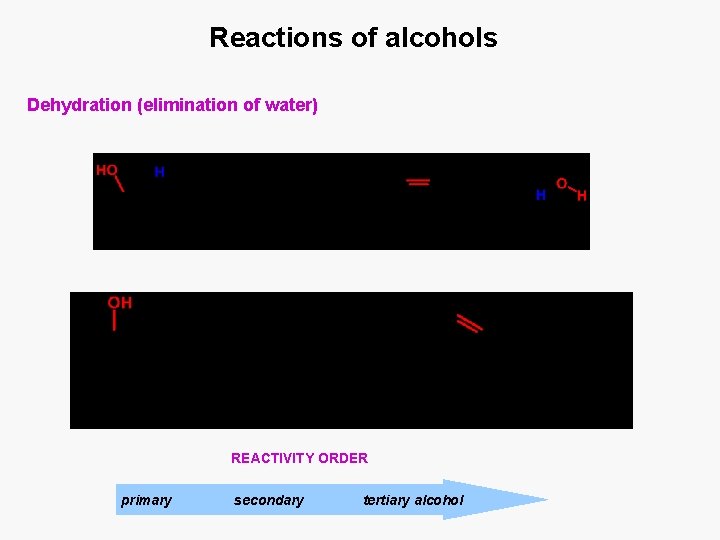
Reactions of alcohols Dehydration (elimination of water) REACTIVITY ORDER primary secondary tertiary alcohol

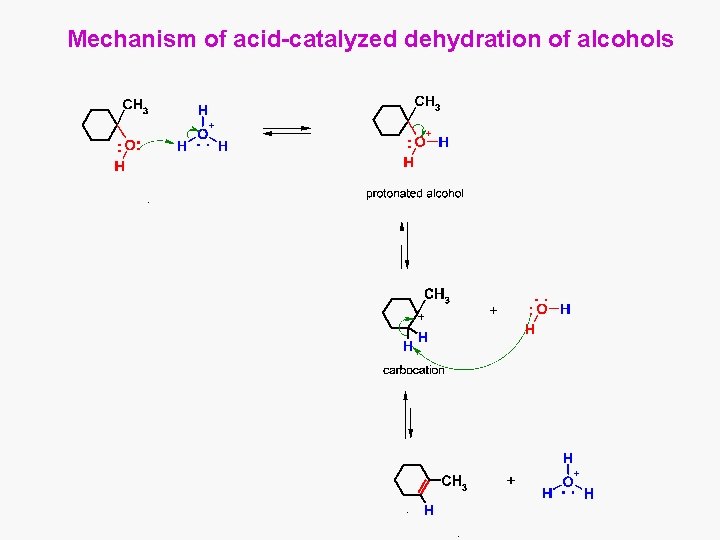
Mechanism of acid-catalyzed dehydration of alcohols

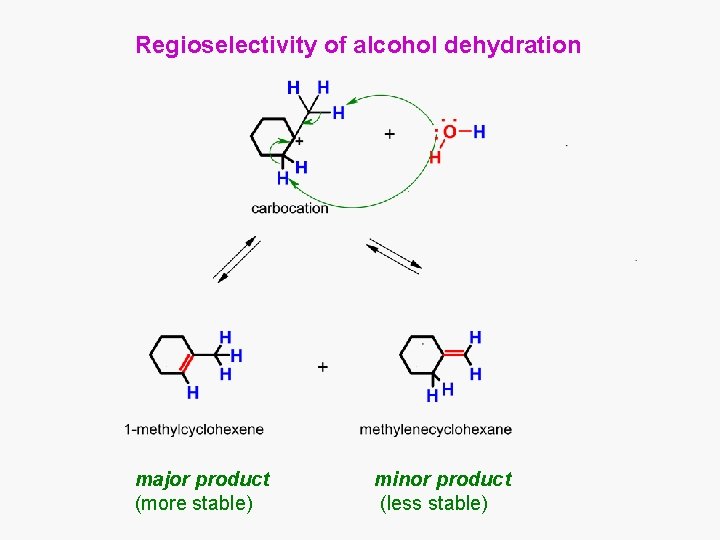
Regioselectivity of alcohol dehydration major product (more stable) minor product (less stable)

Reactions of alcohols Conversion to alkyl halides (nucleophilic substitution) SOCl 2 – thionyl chloride PBr 3 – phosphorus tribromide

Reactions of alcohols Oxidation of hydroxyl group (dehydrogenation) Oxidants: O 2, catalyst Cr. O 3, H 2 SO 4 (Jones reagent) K 2 Cr 2 O 7 (Na 2 Cr 2 O 7), H 2 SO 4 PCC (pyridinium chlorochromate) KMn. O 4

Preparation of phenols Oxidation of cumene – industrial method of phenol production

Preparation of phenols Thermal decomposition of arylsufonic acids

Reactivity of phenols • Aromatic ring susceptible for electrophilic substitution (alkylation, nitration, halogenation etc. ) • Hydroxyl group does not react in nucleophilic substitution • Hydroxyl proton more acidic than in aliphatic alcohols • Different way of oxidation

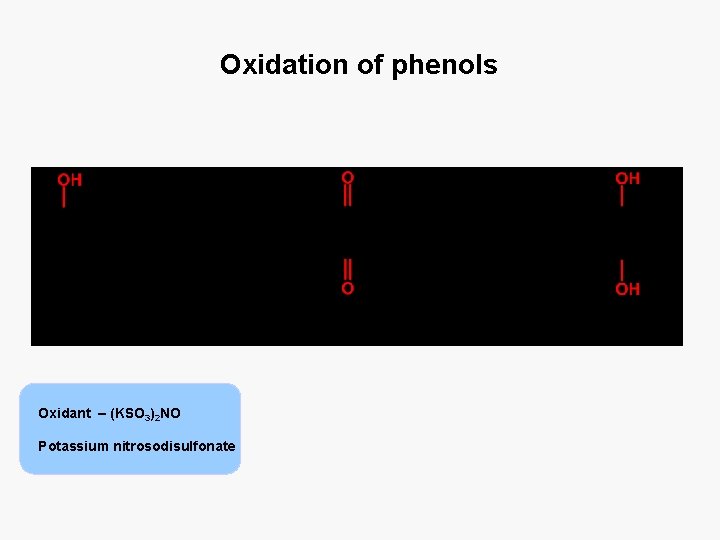
Oxidation of phenols Oxidant – (KSO 3)2 NO Potassium nitrosodisulfonate

Applications of some phenols

ETHERS

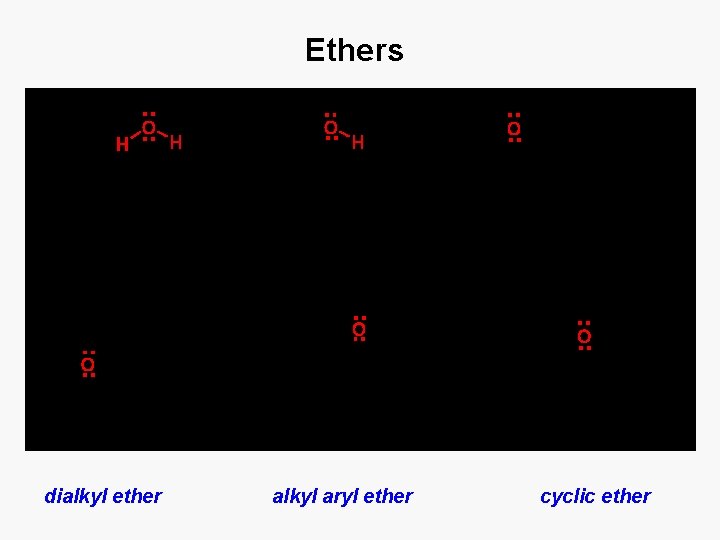
Ethers dialkyl ether alkyl aryl ether cyclic ether

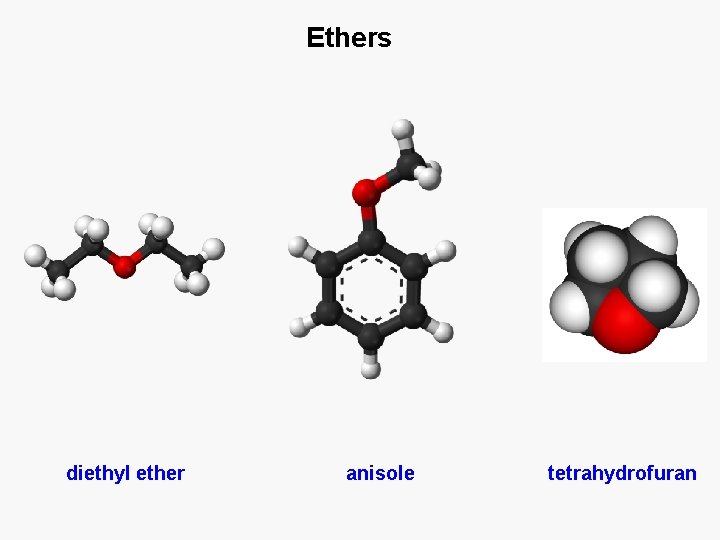
Ethers diethyl ether anisole tetrahydrofuran

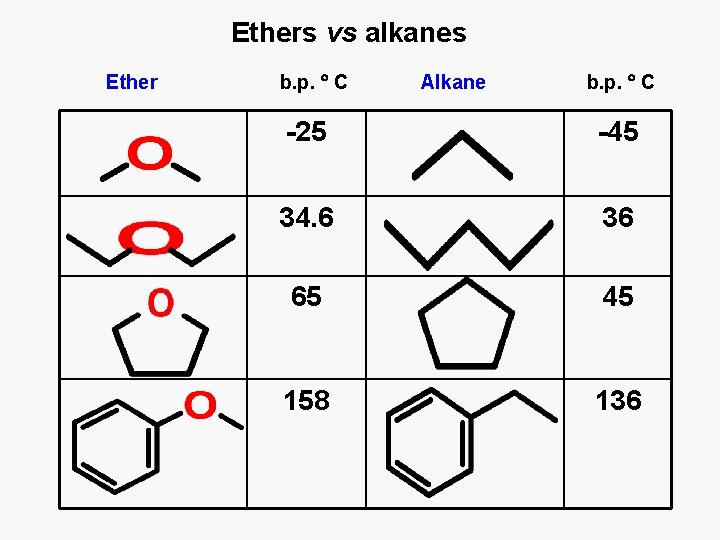
Ethers vs alkanes Ether b. p. C Alkane b. p. C -25 -45 34. 6 36 65 45 158 136

Properties of ethers Comparison to alcohols and alkanes • More polar than alkanes, less than alcohols • No hydrogen bonding • Boiling points – higher than for alkanes, lower than for alcohols • Solubility in water limited • Neutral - nor basic nor acidic • Used frequently as solvents

Preparation of ethers Dehydration of alcohols – symmetric ethers only Williamson’s synthesis (SN 2 reaction)

Other example of Williamson’s synthesis

Reactions of ethers Acid cleavage

Claisen rearrangement of allyl ethers

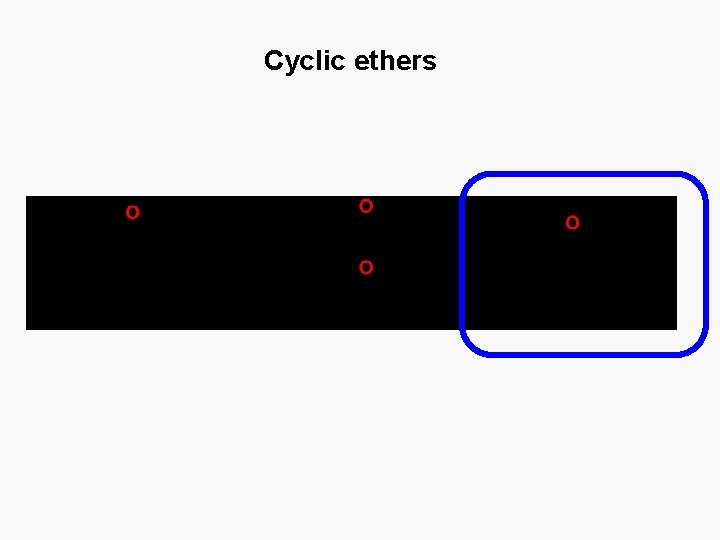
Cyclic ethers

Preparation of oxiranes Oxidation of alkenes

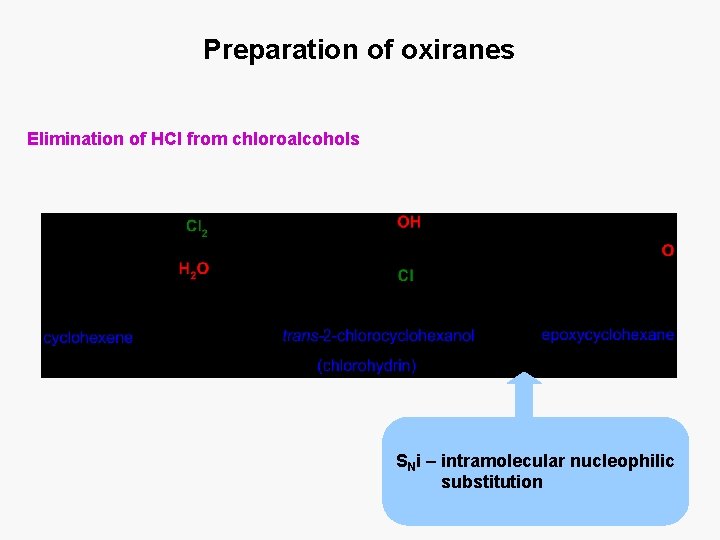
Preparation of oxiranes Elimination of HCl from chloroalcohols SNi – intramolecular nucleophilic substitution

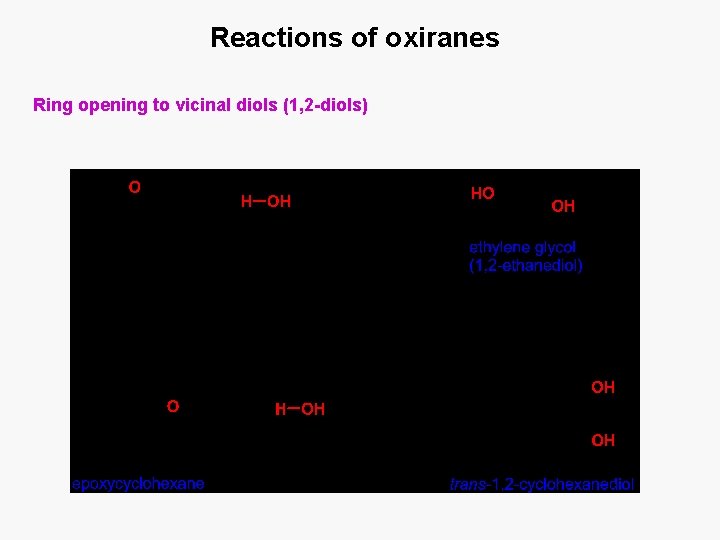
Reactions of oxiranes Ring opening to vicinal diols (1, 2 -diols)

Reactions of oxiranes Ring opening with other nucleophiles

Reactions of oxiranes Ring opening with Grignard reagents New carbon bond

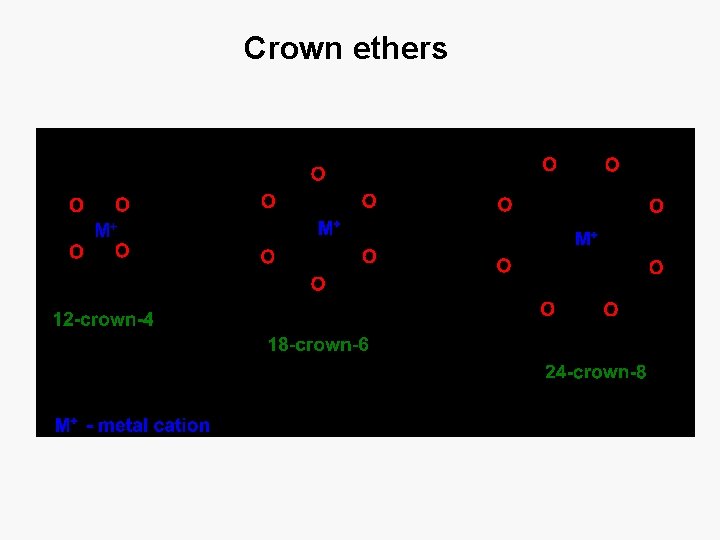
Crown ethers

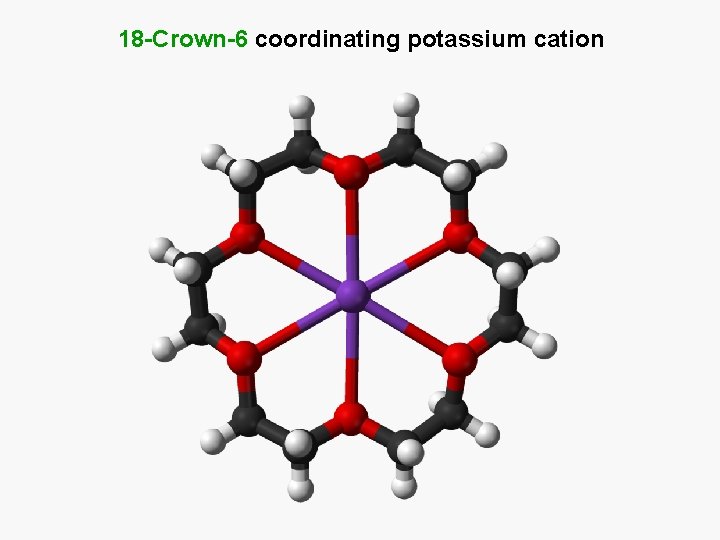
18 -Crown-6 coordinating potassium cation